| Clin Mol Hepatol > Volume 18(3); 2012 > Article |
ABSTRACT
Transcatheter arterial chemoembolization (TACE) has been used widely to treat patients with unresectable hepatocellular carcinoma. However, this method can induce various adverse events caused by necrosis of the tumor itself or damage to nontumor tissues. In particular, neurologic side effects such as cerebral infarction and paraplegia, although rare, may cause severe sequelae and permanent disability. Detailed information regarding the treatment process and prognosis associated with this procedure is not yet available. We experienced a case of paraplegia that occurred after conducting TACE through the intercostal artery to treat hepatocellular carcinoma that had metastasized to the rib. In this case, TACE was attempted to relieve severe bone pain, which had persisted even after palliative radiotherapy. A sudden impairment of sensory and motor functions after TACE developed in the trunk below the level of the sternum and in both lower extremities. The patient subsequently received steroid pulse therapy along with supportive care and continuous rehabilitation. At the time of discharge the patient had recovered sufficiently to enable him to walk by himself, although some paresthesia and spasticity remained.
Hepatocellular carcinoma (HCC) is ranked fifth on the list of cancer incidence globally,1 and various modalities have been developed for its treatment. Partial hepatectomy and liver transplantation are usually performed for complete cure, but they are restricted to less than 30% of patients due to number and size of HCC.2 Therefore, most patients with HCC are treated using non-surgical procedures like transcatheter arterial chemo-embolization (TACE), radiofrequency ablation, percutaneous ethanol injection, systemic chemotherapy, radiotherapy and so on. TACE is a palliative treatment method, which treats cancer by injecting an embolic material with anticancer agents through supplying arteries of tumors, and it has been reported to improve the survival of patients with advanced HCC.3 However, TACE demands special attention as it may cause not only a well-known post-infarction syndrome of high fever, right upper abdominal pain, and nausea in about 2.9% of cases,4 but also various other complications, resulting from unintended embolization of the normal liver or extrahepatic organs including the gall bladder, pancreas, lung, brain, and spinal cord.5 In particular, neurologic adverse events such as cerebral infarction or paraplegia, although being rare, might leave severe sequelae with permanent disability, and require an immediate and effective treatment.5-8 However, on their treatment process and prognosis, no well documented and detailed information is available. We experienced a case of paraplegia that occurred after conducting TACE through intercostal artery to treat hepatocellular carcinoma metastasized to the rib. We report it here together with a review of literature.
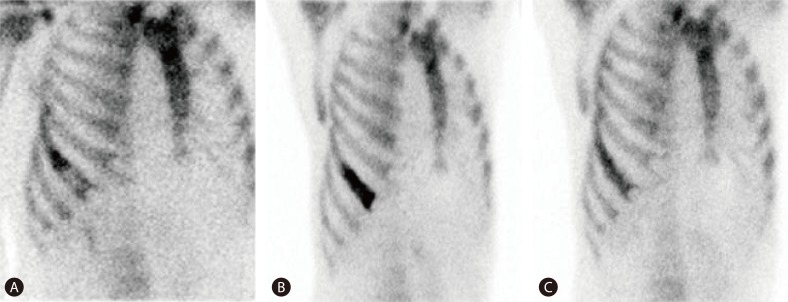
A 57-year-old man was hospitalized with a right flank pain, which was attributed to costal metastasis of HCC. The patient had received several sessions of TACE and radiofrequency ablation for HCCs of the liver since January 2006. About 10 months ago, he underwent a radiofrequency ablation on the small soft-tissue mass in the chest wall, measuring 2.2 cm, adjacent to segment 5 of the liver (Fig. 1A), which was suspected on computed tomography (CT) to be a metastatic lesion seeded from HCC during the process of radiofrequency ablation. Five months later, he felt pain around the rib neighboring the chest wall lesion treated last time, and CT revealed an osteolytic lesion in the right seventh rib (Fig. 1B) that was hypermetabolic on positron emission tomography (PET) (Fig. 2), which suggested a costal metastasis of HCC. He received a course of radiotherapy of total 39 Gy on that bone lesion. After the completion of radiotherapy, the pain was a bit relieved though no definite change was found on the image of the follow-up CT (Fig. 1C). However, it was suspected that some viable tumor mass was still remaining because the bone scan showed increased uptake in the treated region (Fig. 3A). The pain in costal metastasis was again intensified with an increased uptake in the latest bone scan (Fig. 3B).
The patient was taking lamivudine and adefovir dipivoxil due to hepatitis B virus induced cirrhosis with resistance to lamivudine. And he had smoked for 15 years but had no significant drinking history. Vital signs at the time of hospitalization were follows; blood pressure of 120/80 mmHg, heart rate of 66 per minute, respiratory rate of 20 per minute, and body temperature of 36.8Ōäā. There was no particular finding other than tenderness in the right chest wall on physical examination, and no abnormal finding in the neurologic examination as well. The results of blood test were as follows: hemoglobin of 10.9 g/dL, leukocyte of 11,800/mm3, platelet count of 31,000/mm3, aspartate aminotransferase of 27 IU/L, alanine aminotransferase of 25 IU/L, blood urea nitrogen of 16 mg/dL, creatinine of 1.1 mg/dL, Na of 139 mEq/L, K of 3.8 mEq/L, Cl of 10 mEq/L, prothrombin time of 17.4 seconds, positive hepatitis B surface antigen, positive hepatitis B e antigen, hepatitis B virus DNA level of 9,830 IU/mL on real time polymerase chain reaction, negative hepatitis C virus antibody, and alpha-fetoprotein of 4.4 ng/mL.
On the second hospital day, the patient received TACE using 20 mg of doxorubicin and 3 cc of lipiodol mixed with gelfoam through the right posterior intercostal artery which supplied the metastatic lesion of right seventh rib (Fig. 4). Immediately after TACE, the patient complained of sudden paraplegia and weakening of somatic senses in the trunk below the level of sternum and in both lower extremities along with difficult urination and dyschezia. On neurologic examination, it was found that muscle strength was severely decreased in both legs with Medical Research Council (MRC) scale 1 as compared to MRC scale 5 in both upper limbs. Somatic senses were prominently impaired below dermatome T6-7 on neurologic examination with the loss of proprioception. Deep tendon reflex of both legs was accelerated with positive sign of Babinski reflex and ankle clonus, suggesting the presence of an upper motor neuron disease. Although the magnetic resonance imaging (MRI) study conducted 6 hours after TACE did not show any abnormal findings in the spinal cord, it was possible that the size of cord lesion was too small or the timing of imaging study was too early to demonstrate distinct abnormal findings. So, we speculated that TACE caused an ischemic injury to the spinal cord taking into account the neurologic symptoms and signs observed, and immediately started a steroid pulse therapy of 1,000 mg of methyl prednisolone per day for 5 days. In addition, a rehabilitation treatment was instituted while maintaining urination and defecation through Foley catheter and enema. Muscle relaxant and pregabalin were used for muscular stiffness and severe paresthesia, respectively.
After the treatments, muscle strength of both legs increased gradually, and somatic senses were fairly recovered. On the 14th day after the injury, muscle strength of both legs reached MRC scale 3-5. From the 19th day after the injury, the patient became able to try natural urination and defecation with a gradual withdrawal of urinary catheterization and enema. From the 45th day after the injury, he was able to walk by himself at last, and muscle strength and proprioception were almost normalized. However, he was still on medication because of unremitting paresthesia such as numbness in sole, even though 6 months passed after the injury. The follow-up bone scan showed that the uptake in the lesion of seventh right rib decreased compared to the previous image (Fig. 3C).
It requires basic knowledge of the anatomic structure of spinal cord blood supply to understand the paraplegia occurring due to TACE. One anterior and two posterior spinal arteries are the main blood vessels supplying the spinal cord, which arise from vertebral artery and run down along the length of the spinal cord, providing blood to anterior 3/4 and posterior 1/4 territory of it, respectively.9 The anterior spinal artery is anastomosed with 6-10 anterior segmental medullary arteries derived from spinal branches of cervical, intercostal and lumbar arteries, and the biggest one is called artery of Adamkiewicz.10,11 The right and left posterior spinal arteries are connected with 10-23 posterior radicular arteries also originating from spinal branches of cervical, posterior intercostal, and lumbar arteries (Fig. 5).9,12 Due to this anatomical structure, when an embolic event occurs in the anterior spinal artery, main ischemic damage occurs in the anterior 3/4 part of spinal cord including anterior and lateral funiculus, and consequent functional disorders chiefly occur on a motor system rather than a sensory system, and their manifestations are rather serious. In contrast, ischemic damage in the territory of posterior spinal artery, i.e. the posterior 1/4 part of spinal cord, causes sensory disorders more often than motor dysfunction, and the symptoms are much milder than those of anterior spinal artery syndromes.
The patient of this case has been nearly recovered from motor dysfunction but severe paresthesia lasted till the time of discharge. Therefore, this patient has a high possibility of posterior spinal infarction due to embolization in the posterior spinal artery. During TACE, it was impossible to make a superselection of the anterior branch of seventh right posterior intercostal artery, which supplied the metastatic costal lesion, because the branch was so tortuous and narrow. Therefore, small fraction of embolic materials are supposed to have flowed through the spinal branch of seventh posterior intercostal artery to the posterior radicular artery of the corresponding segment of the spinal cord. This ischemic damage of spinal cord coincides with the dermatomes below the chest where sensory and motion disorders occurred.
There exist a few reports regarding the paraplegia associated with TACE. Tufail et al5 reported a case that occurred after performing TACE via the right lower phrenic artery. In this case, they suggested that the cause of paraparesis was anastomosis between the lateral branches of the right inferior phrenic artery and intercostal arteries. Chung et al6 documented a case related with TACE done by way of 11th and 12th intercostal arteries. However, any detailed prognosis was not described in their cases. Kim et al8 reported 2 cases of paraplegia that developed after TACE through intercostal arteries. In the first case, sensory disorders were improved quickly but motor disturbances were not recovered completely even after 2 months from the injury. This suggests a spinal cord injury was caused by the infarction of the anterior spinal artery. In contrast, the other case showed visualization of right posterior spinal artery during right ninth intercostal artery angiogram, and the patient showed complete recovery of sensory and motor functions after 2 months. The incidence of TACE-related paraplegia appears to be very rare; prevalence rate of 0.3%.6,7 And the two cases reported by Kim et al8 occurred during a decade in the institution where over 400 cases of TACE were being performed every year. However, it is understood from these case reports that TACE using intercostal artery always has a risk of paraplegia. In addition, the risk is especially high when it is difficult to select the tumor supplying vessel through intercostals artery, or dorsal branches of posterior intercostal artery might be seen during angiography.7 Therefore, alternative modalities other than TACE should be first considered for the lesion requiring TACE via intercostals arteries.
REFERENCES
2. Bruix J, Llovet JM. Prognostic prediction and treatment strategy in hepatocellular carcinoma. Hepatology 2002;35:519-524. 11870363.


3. Llovet JM, Real MI, Monta├▒a X, Planas R, Coll S, Aponte J, et al. Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet 2002;359:1734-1739. 12049862.


4. Pietrosi G, Miraglia R, Luca A, Vizzini GB, Fili' D, Riccardo V, et al. Arterial chemoembolization/embolization and early complications after hepatocellular carcinoma treatment: a safe standardized protocol in selected patients with Child class A and B cirrhosis. J Vasc Interv Radiol 2009;20:896-902. 19497762.


5. Tufail K, Araya V, Azhar A, Hertzog D, Khanmoradi K, Ortiz J. Paraparesis caused by transarterial chemoembolization: A case report. World J Hepatol 2010;2:289-291. 21161010.



6. Chung JW, Park JH, Han JK, Choi BI, Han MC, Lee HS, et al. Hepatic tumors: predisposing factors for complications of transcatheter oily chemoembolization. Radiology 1996;198:33-40. 8539401.


7. Dai H, Ding H, Liu F, Yao Z, Li L, Li C, et al. Complications of chemoembolization for hepatic neoplasms. Saudi Med J 2007;28:1208-1212. 17676203.

8. Kim JH, Yeon JE, Jong YK, Seo WK, Cha IH, Seo TS, et al. Spinal cord injury subsequent to transcatheter arterial chemoembolization in patients with hepatocellular carcinoma. Dig Liver Dis 2010;42:67-70. 19546038.


9. Sliwa JA, Maclean IC. Ischemic myelopathy: a review of spinal vasculature and related clinical syndromes. Arch Phys Med Rehabil 1992;73:365-372. 1554311.


10. Bowen BC, DePrima S, Pattany PM, Marcillo A, Madsen P, Quencer RM. MR angiography of normal intradural vessels of the thoracolumbar spine. AJNR Am J Neuroradiol 1996;17:483-494. 8881243.


Figure┬Ā1
(A) Abdominal CT scan showing a soft-tissue mass with contrast enhancement in the right chest wall, measuring 2.2 cm, which was suspected to have metastasized from intrahepatic HCC in the adjacent segment 5 following radiofrequency ablation (RFA; arrow). (B) Follow-up CT performed 5 months after treating the soft-tissue lesion of the chest wall with RFA. An osteolytic lesion is observed in the 7th rib (arrow) neighboring the RFA-treated chest-wall lesion (arrow). (C) Follow-up CT after radiotherapy with a total of 39 Gy to the costal lesion. The osteolytic lesion has persisted without any significant change (arrow).

Figure┬Ā2
Positron-emission tomography (PET) scan performed 5 months later after RFA of the soft-tissue mass in the right chest wall. A metastatic malignant lesion is suspected because of the hypermetabolism noted in the right 7th rib. (A) Anterior view. (B) Right lateral view.

Figure┬Ā3
(A) Whole-body bone scan conducted immediately after the radiotherapy. An uneven increased uptake was evident at the metastatic lesion of the hepatocellular carcinoma in the right 7th rib. (B) Four months later, the intensity and extent of uptake had increased at the same lesion relative to the previous image. (C) A follow-up bone scan was performed 4 months after a TACE procedure for the costal metastasis; the intensity of uptake was decreased at the metastatic lesion.
Figure┬Ā4
Intercostal arteriograph obtained during TACE on the metastatic lesion of hepatocellular carcinoma in the right 7th rib (black arrow). Embolization was conducted by injecting a mixture of doxorubicin, Lipiodol, and Gelfoam through the right posterior intercostal artery, which courses through the lower part of the 7th rib. Microcatheter insertion was attempted for embolization, but it was difficult to progress due to the narrowness and severe tortuosity of the vessel (white arrow). Blood vessels that are considered to be the dorsal branches of the posterior intercostal artery can be seen.

Figure┬Ā5
The anterior spinal artery is anastomosed with 6-10 anterior segmental medullary arteries derived from spinal branches of the cervical, intercostal, and lumbar arteries; the largest is called the artery of Adamkiewicz. The right and left posterior spinal arteries are connected to 10-23 posterior radicular arteries that also originate from spinal branches of the cervical, posterior intercostal, and lumbar arteries.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




