| Clin Mol Hepatol > Volume 28(2); 2022 > Article |
|
ABSTRACT
Background/Aims
Methods
Results
ACKNOWLEDGMENTS
FOOTNOTES
SUPPLEMENTAL MATERIAL
Supplementary┬ĀFigure┬Ā1.
Supplementary┬ĀFigure┬Ā2.
Supplementary┬ĀTable┬Ā1.
Figure┬Ā1.
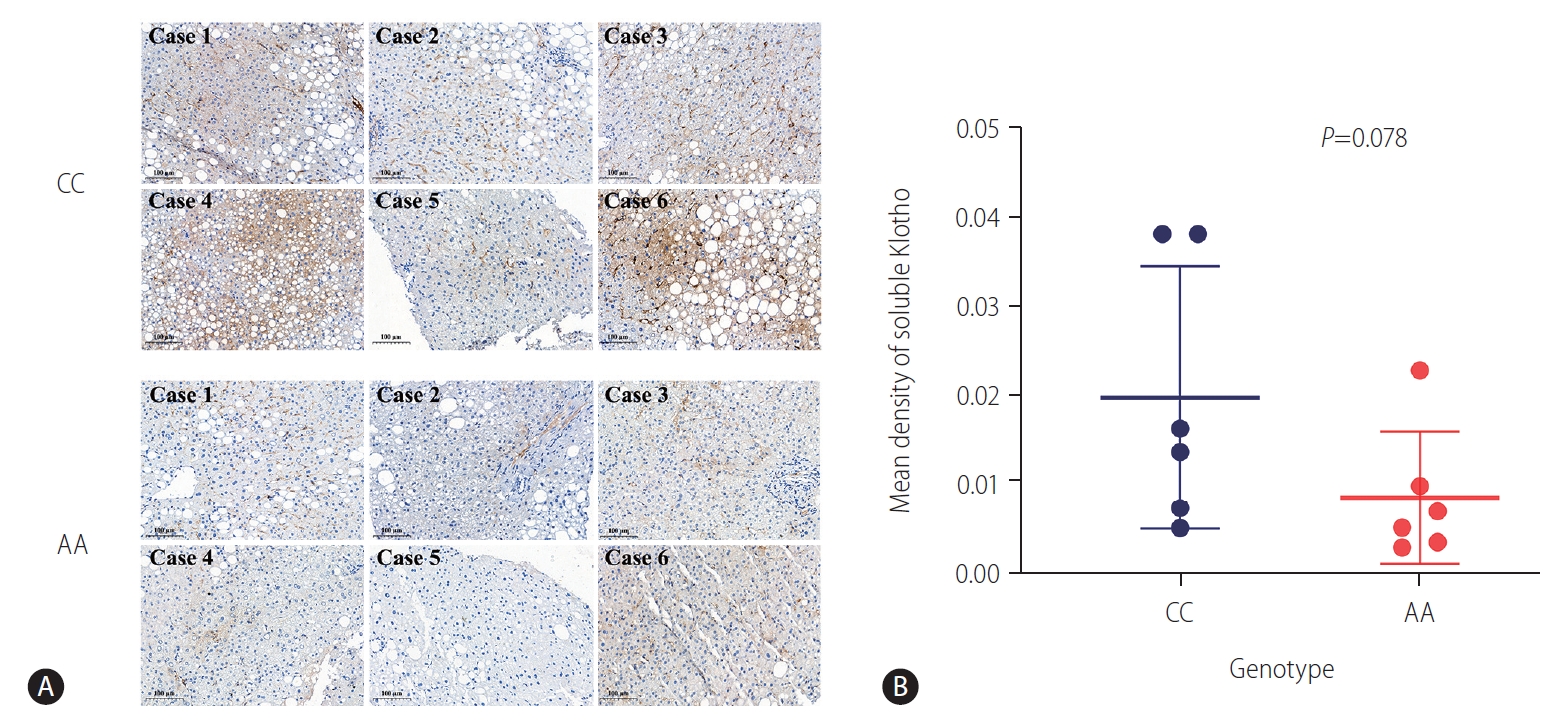
Figure┬Ā2.
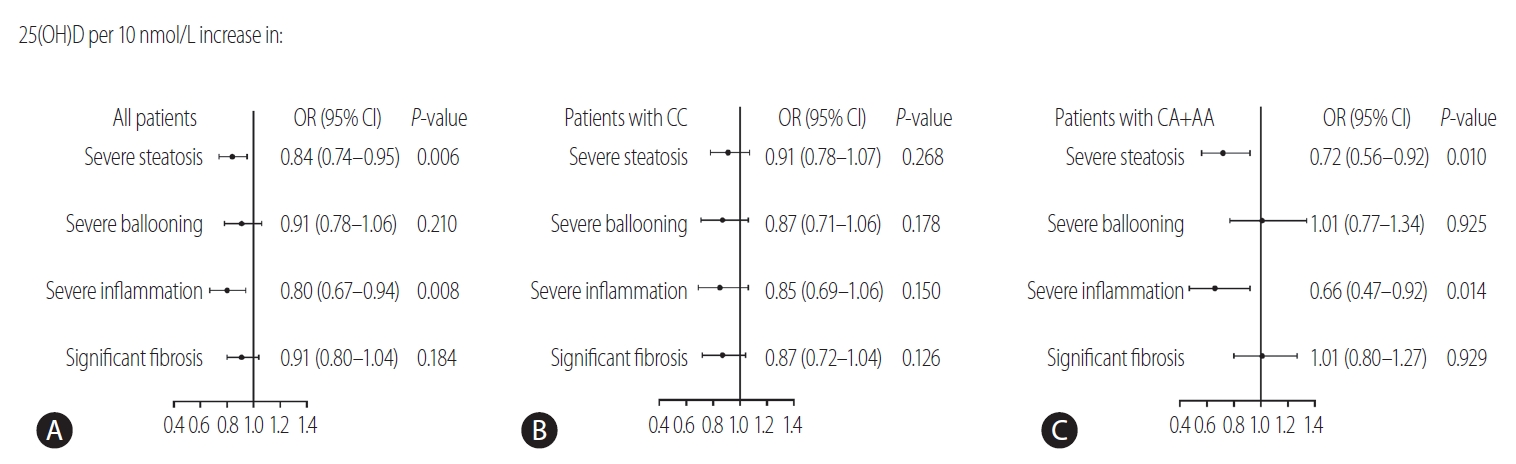
Figure┬Ā3.
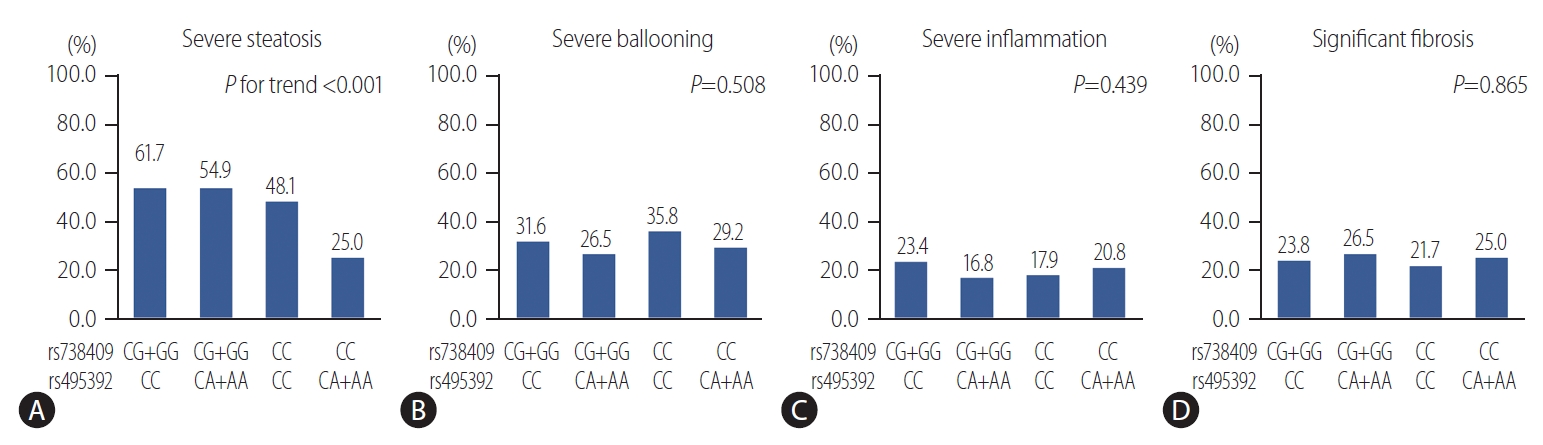
Table┬Ā1.
Values are presented as mean┬▒standard deviation for those with a normal distribution, medians (p25ŌĆōp75) for those with a non-normal distribution, or number (%).
NAFLD, non-alcoholic fatty liver disease; BMI, body mass index; HOMA-IR, homeostasis model assessment-insulin resistance; AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase; GGT, ╬│-glutamyl transpeptidase; TC, total cholesterol; TG, triglycerides; HDL-C, high-density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; 25(OH)D, 25-hydroxyvitamin D; NAS, NAFLD Activity Score.
Table┬Ā2.
|
Severe steatosis |
Severe ballooning |
Severe inflammation |
Significant fibrosis |
|||||
|---|---|---|---|---|---|---|---|---|
| OR (95% CI) | P-value | OR (95% CI) | P-value | OR (95% CI) | P-value | OR (95% CI) | P-value | |
| Crude model | ||||||||
| ŌĆāCC | Ref. | - | Ref. | - | Ref. | - | Ref. | - |
| ŌĆāCA+AA | 0.61 (0.42ŌĆō0.89) | 0.010 | 0.73 (0.48ŌĆō1.09) | 0.126 | 0.81 (0.51ŌĆō1.29) | 0.383 | 1.18 (0.77ŌĆō1.81) | 0.437 |
| Adjusted model* | ||||||||
| ŌĆāCC | Ref. | - | Ref. | - | Ref. | - | Ref. | - |
| ŌĆāCA+AA | 0.66 (0.45ŌĆō0.98) | 0.040 | 0.75 (0.50ŌĆō1.13) | 0.171 | 0.85 (0.53ŌĆō1.36) | 0.493 | 1.17 (0.75ŌĆō1.81) | 0.493 |
Table┬Ā3.
|
60Ōēż FLI <78* |
FLI Ōēź7ŌĆĀ |
|||
|---|---|---|---|---|
| OR (95% CI) | P-value | OR (95% CI) | P-value | |
| All (n=3,830) | ||||
| ŌĆāCC | Ref. | - | Ref. | - |
| ŌĆāCA | 0.95 (0.75ŌĆō1.21) | 0.700 | 0.81 (0.57ŌĆō1.15) | 0.240 |
| ŌĆāAA | 0.82 (0.53ŌĆō1.25) | 0.364 | 0.36 (0.17ŌĆō0.73) | 0.006 |
| LSM Ōēż6.9 kPa (n=2,428) | ||||
| ŌĆāCC | Ref. | - | Ref. | - |
| ŌĆāCA | 0.90 (0.66ŌĆō1.21) | 0.479 | 0.71 (0.43ŌĆō1.17) | 0.182 |
| ŌĆāAA | 0.83 (0.50ŌĆō1.36) | 0.467 | 0.17 (0.05ŌĆō0.49) | 0.002 |
| LSM >6.9 kPa (n=370) | ||||
| ŌĆāCC | Ref. | - | Ref. | - |
| ŌĆāCA | 1.26 (0.57ŌĆō2.82) | 0.565 | 1.84 (0.71ŌĆō4.93) | 0.210 |
| ŌĆāAA | 0.77 (0.11ŌĆō4.29) | 0.781 | 1.13 (0.11ŌĆō8.76) | 0.910 |
Table┬Ā4.
Table┬Ā5.
Abbreviations
REFERENCES
- TOOLS
-
METRICS

- ORCID iDs
-
Mohsen Ghanbari

https://orcid.org/0000-0002-9476-7143Ming-Hua Zheng

https://orcid.org/0000-0003-4984-2631 - Related articles
-
Recent updates on pharmacologic therapy in non-alcoholic fatty liver disease2024 January;30(1)
Implications of comorbidities in nonalcoholic fatty liver disease2023 April;29(2)
Non-invasive biomarkers of liver fibrosis in non-alcoholic fatty liver disease2023 April;29(2)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Supplement1
Supplement1 Print
Print



