| Clin Mol Hepatol > Volume 26(4); 2020 > Article |
|
ABSTRACT
ACKNOWLEDGMENTS
FOOTNOTES
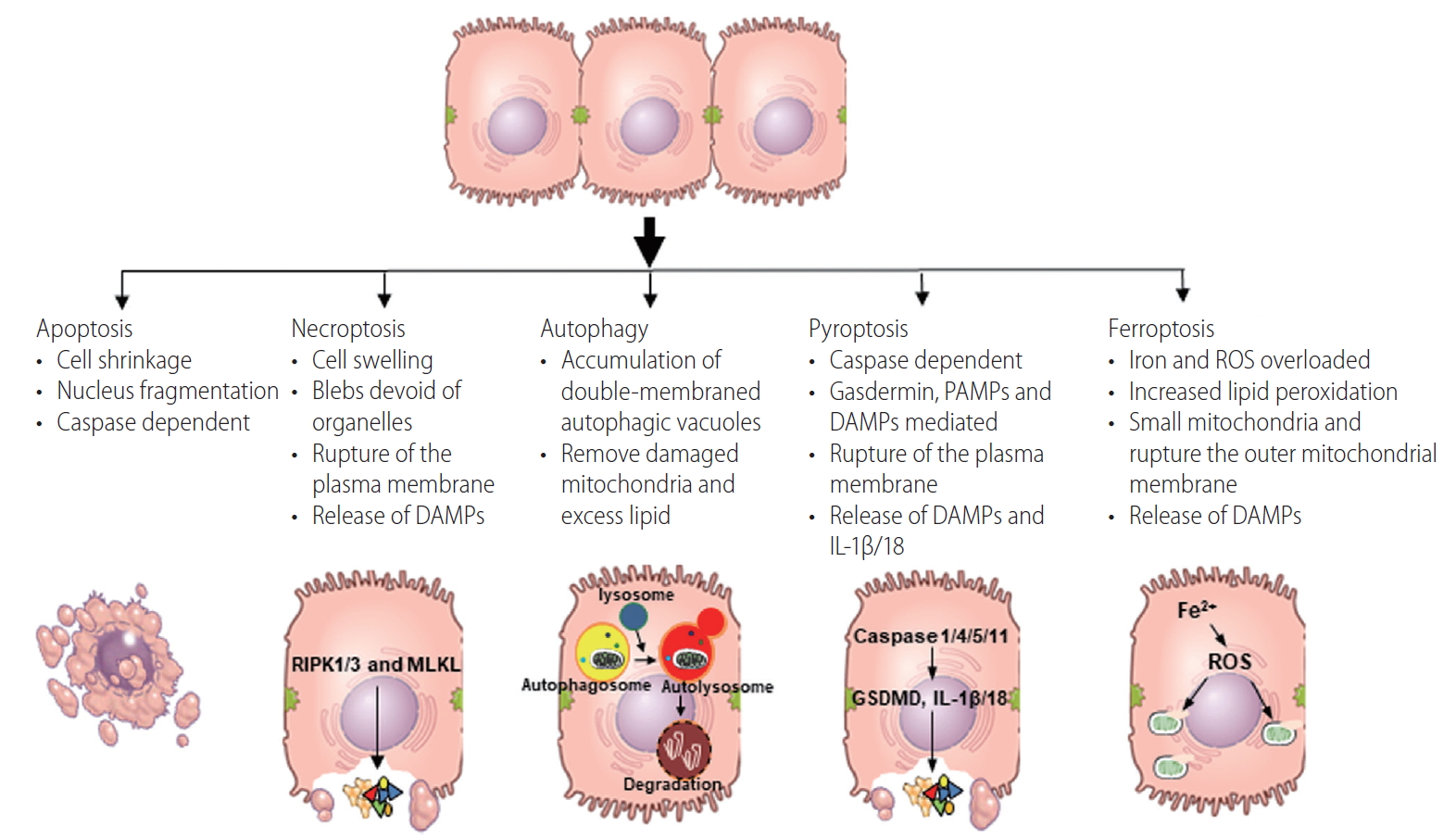
Figure┬Ā1.
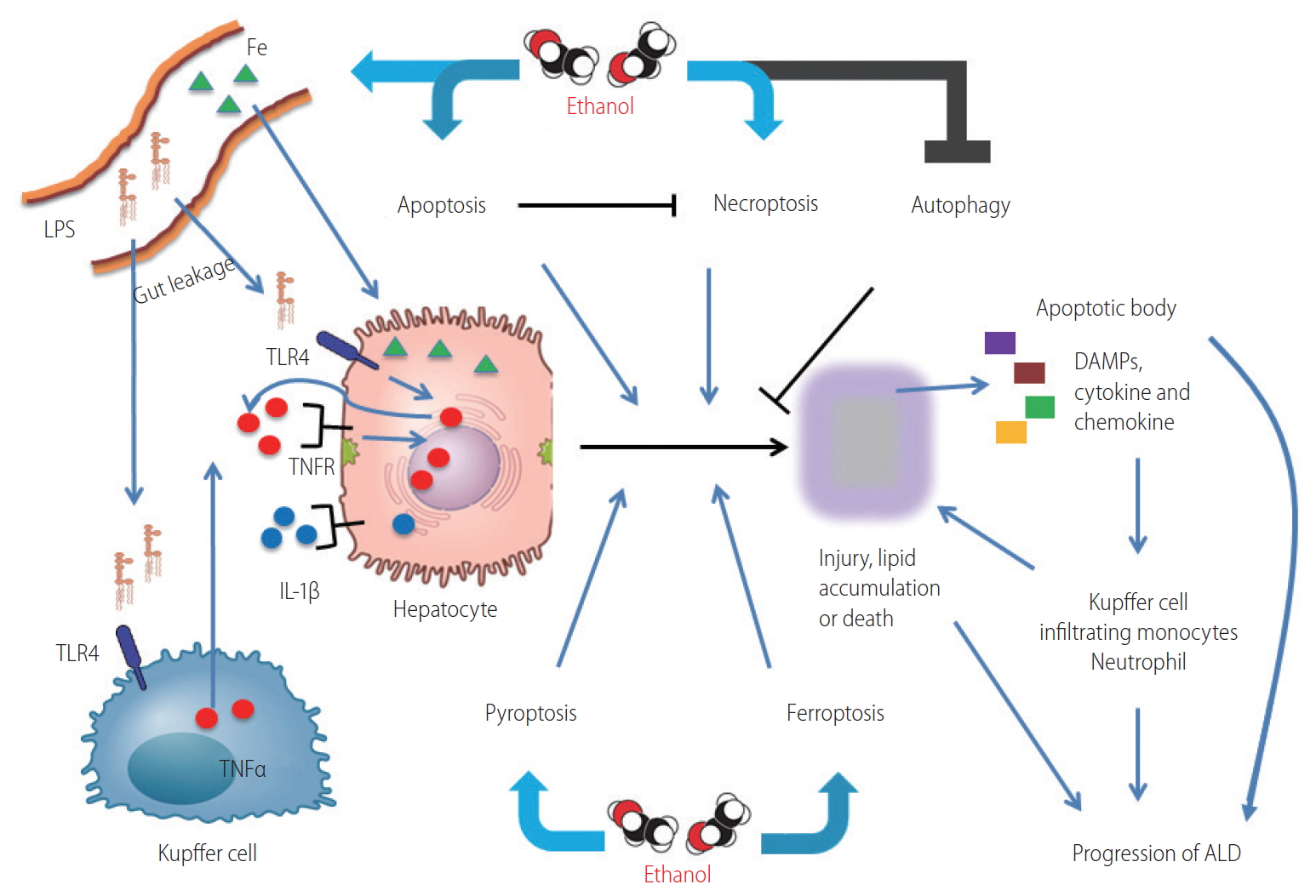
Figure┬Ā2.
Table┬Ā1.
| Apoptosis | Necroptosis | Autophagy | Pyroptosis | Ferroptosis | |
|---|---|---|---|---|---|
| Morphological features | Cell shrinkage, nucleus fragmentation, membrane blebbing, apoptotic body [7] | Cell swelling, blebs devoid of organelles, release of DAMPs [4,11,12] | Accumulation of double-membraned autophagic vacuoles [21-23] | Cell swelling, osmotic lysis, release of DAMPs, IL-1╬▓ and IL-18 [30-32] | Electron-dense mitochondria, rupture the outer mitochondrial membrane, the presence of lipid peroxidation. |
| Release of DAMPs [36] | |||||
| Trigger | ER stress, ROS, hypoxia [5,9,10] | ER stress, ROS, hypoxia [5,19,20] | ER stress, ROS, hypoxia [21-23] | PAMPs, DAMPs, ER stress [30-33] | Iron accumulation, ROS, ER stress [36,37,42,43] |
| Key proteins | Initiation (caspase-2, -8, -9, and -10), execution (caspase -3, -6, and -7) Bcl-2, FADD [7-9] | RIP1, RIP3, and MLKL [5,12,19,20] | ULK 1 kinase complex, class III PI3K complex, ATG5, ATG7, LC3, mTORC1, [21-23] TFEB [24,27] | GSDMD, inflammasomes, caspase 1/4/5/11, IL-1╬▓ and IL-18 [30-32] | GSH, GPX4, [37,45] hepcidin [42-44] |
| Hepatocyte | ROS, ER stress, CYP2E1 cause apoptosis in mitochondria. | The activation of RIP1-RIP3-MLKL axis cause necroptosis under caspase-8 inhibition. [5,19-20] | Macrophagy deletes unfolded protein. [21] Lipophagy reduces the accumulation of lipids. [21] | Activated inflammasomes actlvate caspase 1/11, which mature GSDMD and pro-, IL-1╬▓ and pro-IL-18, resulting in release of DAMPs, IL-1╬▓ and IL-18 [30-32] | Reactive hydroxyl radical by iron accumulation injures lipid membranes to induce lipid peroxidation and membrane instability, which can ultimately result in leakage of cellular material and cell death. [38-43] |
| TNF╬▒ signal induced caspase-8 results in apoptosis. [4,5,10] | |||||
| The role in or to immune cells | Kupffer cells produce TNF╬▒ by LPS-TLR4 signal activation from leakage-gut. [5] | Immune cells are activated by DAMPs released from necroptotic hepatocytes [5,14-16] | Autophagy plays a role of anti-inflammatory response and anti-steatosis via cannabinoid receptor 2 in Kupffer cells. [28] | DAMPs, IL-1╬▓ and IL-18 by pyroptosis recruit and activate macrophages, neutrophils and lymphocytes to produce pro-inflammatory cytokines. [5,30-32,34] | Loss of iron in macrophage stores via lowhepcidin by ethanol consumption results in hyperabsorption of iron [44] |
| Role in ALD | Promote | Promote | Suppress | Promote | Promote |
ALD, alcohol-associated liver disease; DAMPs, damage-associated molecular patterns; IL, interleukin; ER, endoplasmic reticulum; ROS, reactive oxygen species; PAMPs, pathogen-associated molecular patterns; FADD, FAS associated death domain protein; RIP, receptor interacting protein; MLKL, mixed lineage kinase domain like pseudokinase; PI3K, phosphatidylinositol 3-kinase; ATG, autophagy-related genes; LC3, light chain 3; mTORC1, mTOR complex 1; TFEB, transcription factor EB; GSDMD, gasdermin D; GSH, glutathione; GPX4, glutathione peroxidase-4; TNF╬▒, tumor necrosis factor ╬▒; LPS, lipopolysaccharides; TLR4, toll-like receptor 4.
Abbreviations
REFERENCES
- TOOLS
-
METRICS

- ORCID iDs
-
Laura E. Nagy

https://orcid.org/0000-0002-0580-2809 - Related articles
-
JCAD, a new potential therapeutic target in cholestatic liver disease2024 April;30(2)
Implications of comorbidities in nonalcoholic fatty liver disease2023 April;29(2)
The growing burden of non-alcoholic fatty liver disease on mortality2023 April;29(2)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



