| Clin Mol Hepatol > Volume 26(2); 2020 > Article |
|
ABSTRACT
FOOTNOTES
SUPPLEMENTAL MATERIAL
Supplementary Fig. 1
Supplementary Fig. 2
Supplementary Fig. 3
Supplementary Fig. 4
Supplementary Table 1
Figure 1.
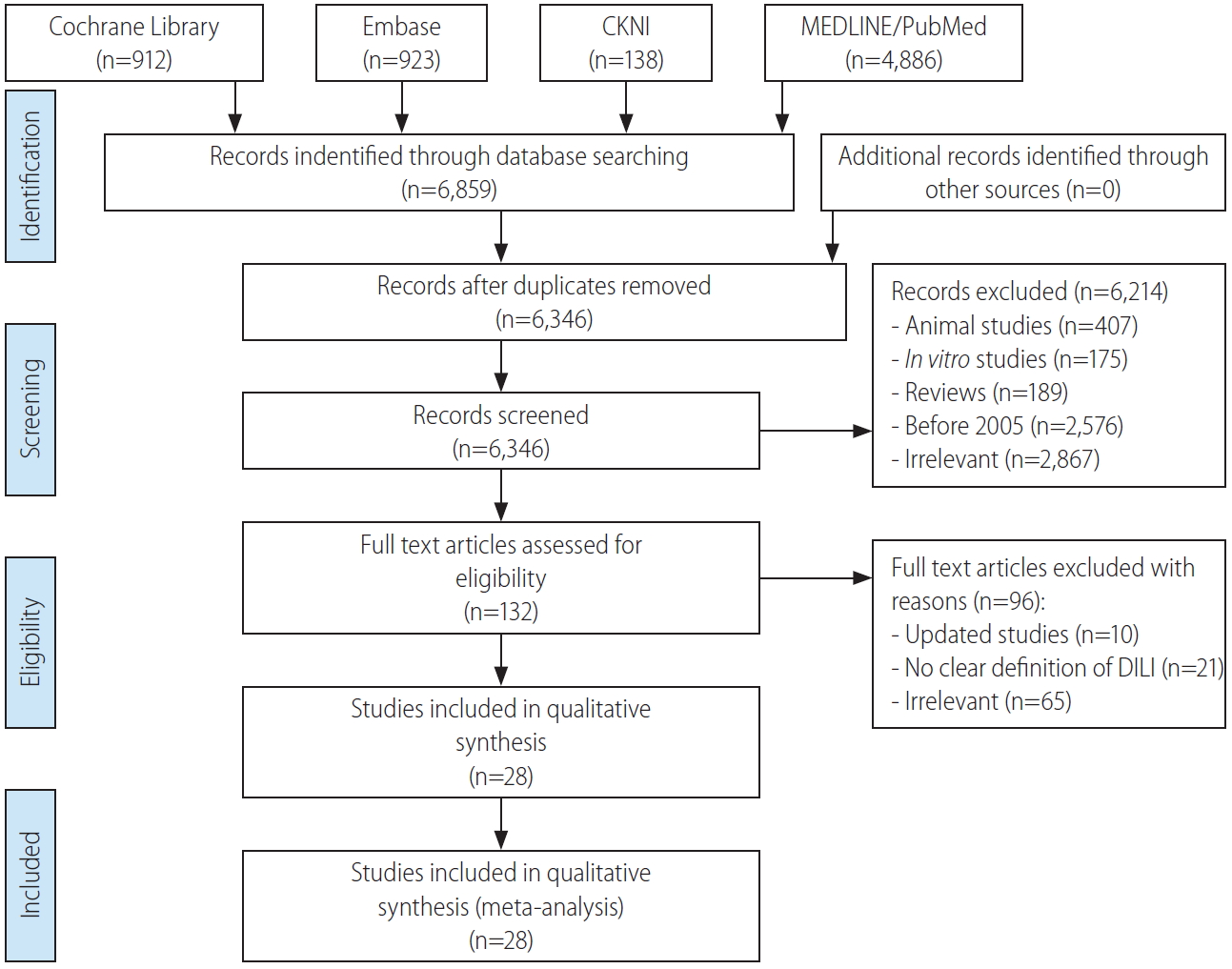
Figure 2.
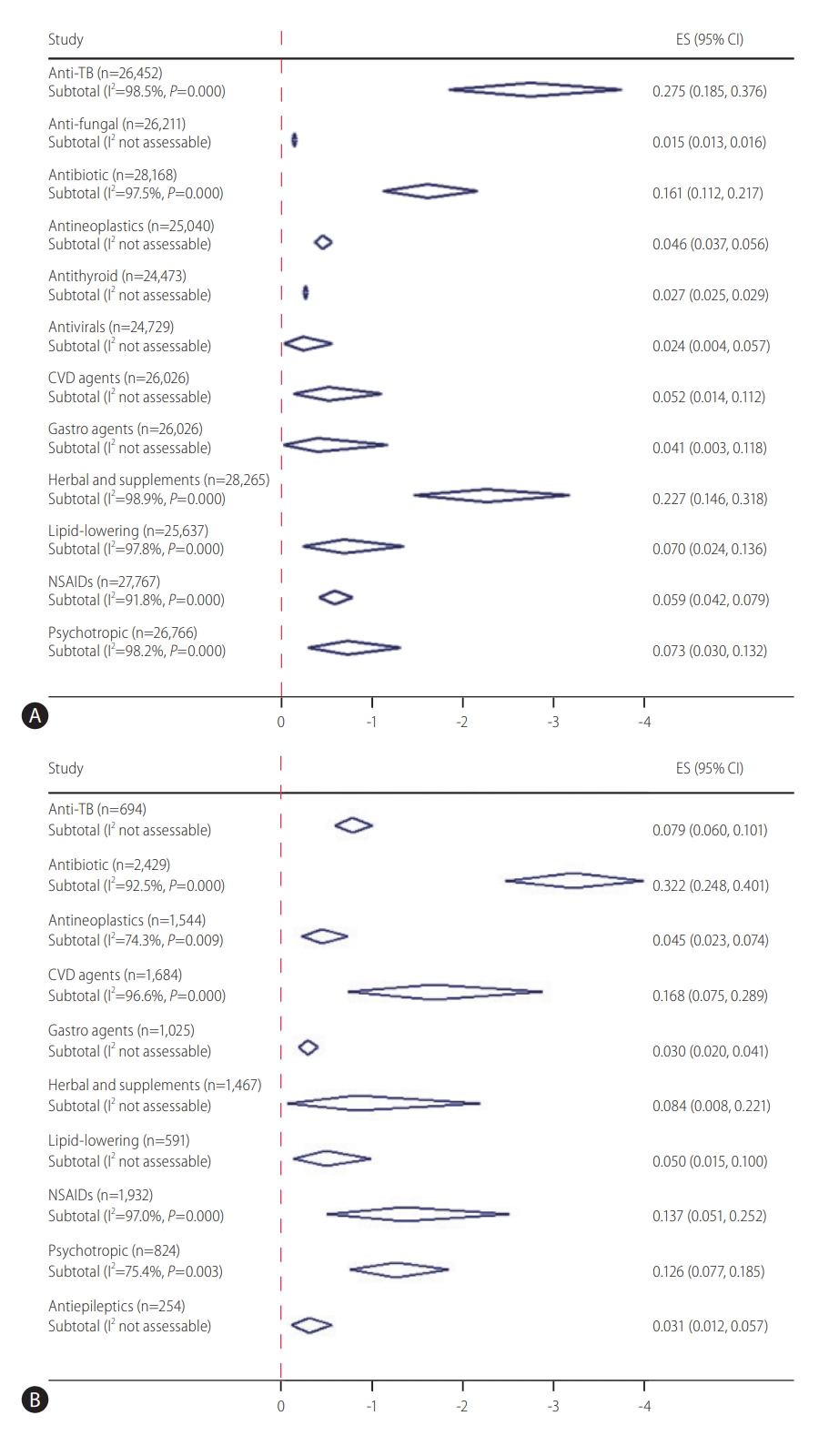
Figure 3.
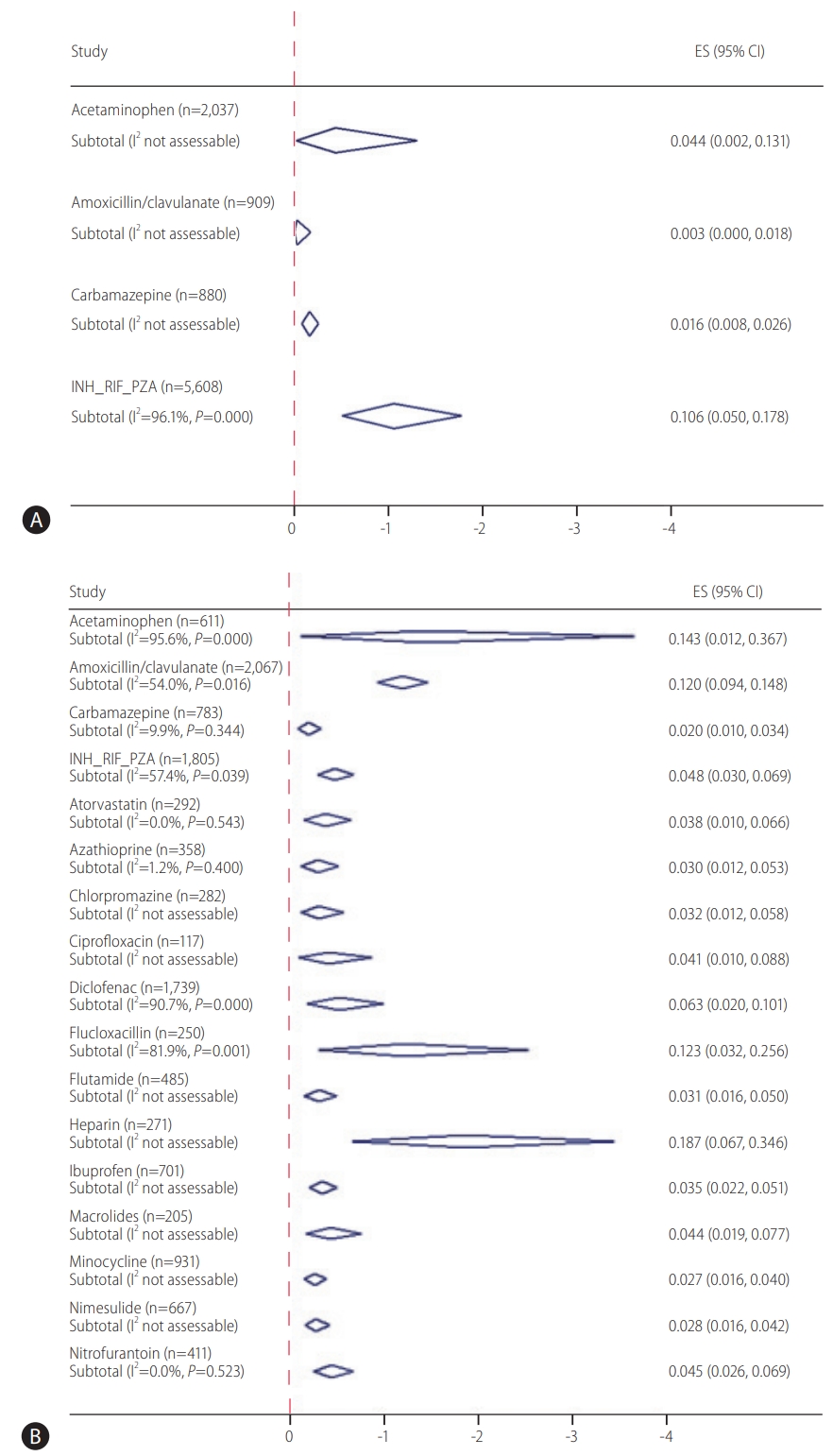
Table 1.
| No | Study | Country/region | Causality assessment for DILI | Time periods | Publication type (report, journal article, abstract) | Hospital-/registrybased | Drug classification type (single agent or class) | Total number of DILI records | Included acetaminophen |
|---|---|---|---|---|---|---|---|---|---|
| Studies from the West | |||||||||
| 1 | Andrade et al. [5] (2005) | Spain | CIOMS/RUCAM | Apr 1994 to Aug-04 | Prospective | Prospective Regional registry | Both | 461 | Yes 13 cases |
| Clinical judgement, CIOMS by 3 independent experts | |||||||||
| 2 | Bessone et al. [14] (2016) | Latin America | CIOMS/RUCAM | 2011 to 2014 | Prospective | Latin American registry | Single | 206 | Yes but 0 cases |
| 3 | Björnsson et al. [4] (2013) | Iceland | CIOMS/RUCAM | Mar 2010 to Feb 2012 | Prospective | National | Both | 96 | No (specifically excluded) |
| 4 | Chalasani et al. [15] (2015) | USA | DILIN methods | Sep 2004 to May 2013 | Observational longitudinal | National | Both | 899 | No (specifically excluded) |
| 5 | Sgro et al. [3] (2002) | France | Global imputability score I | Nov 1997 to Nov 2000 | Population based | Regional | Both | 34 | Yes (1 died: prolonged acetaminophen) |
| 6 | De Valle et al. [16] (2006) | Swedish outpatient clinic | CIOMS | 1995 to 2005 | Retrospective review of case records | 1 university hospital outpatients urban area | Both | 77 | Not mentioned |
| 7 | de Abajo et al. [17] (2004) | UK | Description like RUCAM but not specifically stated | 1994 to 1999 | Population based case control (5,000 controls) | GP research database in the UK | Both | 128 | Yes: 12 from acetaminophen |
| 8 | Ibáñez et al. [18] (2002) | Spain | Description like RUCAM but not specifically stated | 1992 to 1998 | Population based prospective | 12 hospitals collaborating network | Both | 107 | Yes: 12 from acetaminophen |
| 9 | Carey et al. [19] (2008) | US Mayo (inpatients) | CIOMS | 1998 to 2006 | Retrospective search with codes | Inpatient visits at Mayo Hospital | Both | 40 | Yes: 40 had DILI, 27 from acetaminophen |
| 11 | Hussaini et al. [20] (2007) | Cornwall England | CIOMS | 1998 to 2004 | Retrospective analysis | Rural population | Both | 28 | Not mentioned |
| 12 | Meier et al. [21] (2005) | Switzerland | CIOMS | Jan 1996 to Dec 2000 | Medical records review | Pharmacoepidemiological databases | Both | 88 | Yes |
| 13 | Sabaté et al. [22] (2007) | Barcelona Spain 12 hospitals | Jaundice, ALT AST | Jan 1993 to Dec 1999 | Multi centre prospective case control | 12 hospitals | Drugs | 126 | Yes |
| 2,700,000 | |||||||||
| 14 | Sistanizad Peterson. [23] (2013) | Tasmania Australia | CIOMS | Jun 2008 to July 2009 | Retrospective | 1 major hospital serving 250,000 people | Drugs | 17 | Yes |
| 15 | Galan et al. [24] (2005) | US | Description like RUCAM but not specifically stated | 1993 to 2002 | Retrospective review of records | 1 tertiary care hospital | Both | 32 | Yes but 0 cases |
| 16 | Vega et al. [25] (2017) | Delaware, US | CIOMS | 2014 | Prospective | DILN | Both | 23 | Yes |
| Studies from the East | |||||||||
| 17 | Suk et al. [6] (2012) | Korea | RUCAM | May 2005 to May 2007 | Prospective | 17 referral hospitals (nationwide) | Class | 371 | Yes |
| 18 | Kwon et al. [26] (2012) | Korea | WHO-UMC | Jan 2007 to Dec 2008 | Retrospective-registry of spontaneous reports of adverse drug reactions | 9 regional pharmacovigilence centres in Korea (nationwide) | Both | 567 | Yes |
| 19 | Zhou et al. [27] (2013) | China | Various | 1994 to 2011 | Retrospective review of electronic and manual searches | Multiple centres | Class | 24,112 | Yes |
| 20 | Ou et al. [28] (2015) | China | CIOMS | Jan 2011 to Dec 2014 | Retrospective review of inpatient records | Inpatients - 1 hospital | Both | 361 | Yes |
| 21 | Lee et al. [29] (2012) | Taiwan | ICD–9 code (case cross over comparison of diagnosis) | 1997 to 2004 | Retrospective | Population based database (insurance) | Both | 4,857 | Unclear |
| 22 | Takikawa et al. [30] (2009) | Japan | DDW-J 2004 | Jan 1997 to Dec 2006 | Retrospective | 29 facilities (nationwide) | Class | 1,676 | Yes |
| 23 | Huang et al. [31] (2013) (abstract) | Taiwan | RUCAM | Unclear | Retrospective | 6 medical centres across Taiwan | Class | 1,099 | Yes |
| 24 | Sobhonslidsuk et al. [32] (2016) | Thailand | ICD-10 (toxic liver disease) | 2009 to 2013 | Retrospective | Population based database (nationwide) (DILN Taiwan) | Single | 589 | Yes |
| 25 | Rathi et al. [33] (2016) | India | RUCAM | 2014 to 2015 | Prospective | 1 tertiary care hospital in metropolitan India | Class | 82 | Unclear |
| 26 | Bektas et al. [34] (2008) (abstract) | Turkey | Various | Unclear | Retrospective | Single centre | Class | 170 | Unclear |
| 27 | Jaiprakash et al. [35] (2012) | India | AST/ALT | Jul 2006 to Jul 2007 | Retrospective | 1 tertiary care hospital in rural South India | Class | 65 | Unclear |
| 28 | Devarbhavi et al. [36] (2010) | India | RUCAM | 1997 to 2008 | Retrospective | Inpatients - 1 hospital | Both | 244 | Yes |
DILI, drug induced liver injury; CIOMS, Council for International Organizations of Medical Sciences; RUCAM, Roussel Uclaf Causality Assessment Method; GP, general practice; ALT, alanine aminotransferase; AST, aspartate transferase; WHO-UMC, World Health Organisation-Uppsala Monitoring Center; ICD, International Classification of Diseases; DDW-J, The Digestive Disease Week Japan.
Table 2.
Table 3.
Table 4.
REFERENCES
- TOOLS
-
METRICS

- ORCID iDs
-
Seng Gee Lim

https://orcid.org/0000-0003-0994-4932 - Related articles
-
Global prevalence of MAFLD-related hepatocellular carcinoma: A systematic review and meta-analysis
Drug-induced liver injury caused by iodine-1312016 June;22(2)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Supplement1
Supplement1 Print
Print



