INTRODUCTION
About 350 million people world-wide are chronically infected with hepatitis B (HBV),
1,
2 which can result in severe liver disease that eventually progresses to cirrhosis and hepatocellular carcinoma.
3,
4 To date, interferon alpha and five nucleos(t)ide analogs (NAs), namely, lamivudine (LAM), adefovir (ADV), entecavir (ETV), clevudine and telbivudine have been approved for the treatment of chronic hepatitis B (CHB) in Korea. In South-East Asia, oral nucleosides are prescribed more frequently than interferon because genotype C is common.
5 Drug resistance often results from prolonged oral nucleoside treatment. LAM was first released for general use in Korea in 1999. Thereafter, the remaining four antiviral agents were introduced in a sequential manner. ETV resistance occurs frequently in patients with LAM resistance.
6 Moreover, LAM-resistant patients with CHB sometimes also exhibit ADV resistance, although ADV is thought to be a good rescue therapy for LAM resistance.
7 In addition, clevudine (CLV) and telbivudine (LdT) are very similar to LAM in terms of their chemical structure and resistance profile. Thus, an alternative treatment for patients with ADV or ETV resistance is needed. One possible alternative is ADV-ETV combination therapy. This approach has the advantage that the drugs may synergize in terms of efficacy while preventing the emergence of mutants. Its disadvantage is its high cost.
The present study investigated the efficacy of ADV-ETV combination therapy in patients with CHB who did not respond to ADV add-on or high dose (1 mg) ETV therapy. In addition, factors that promoted an initial virological response (IVR) to the combination therapy were identified.
MATERIALS AND METHODS
Subjects
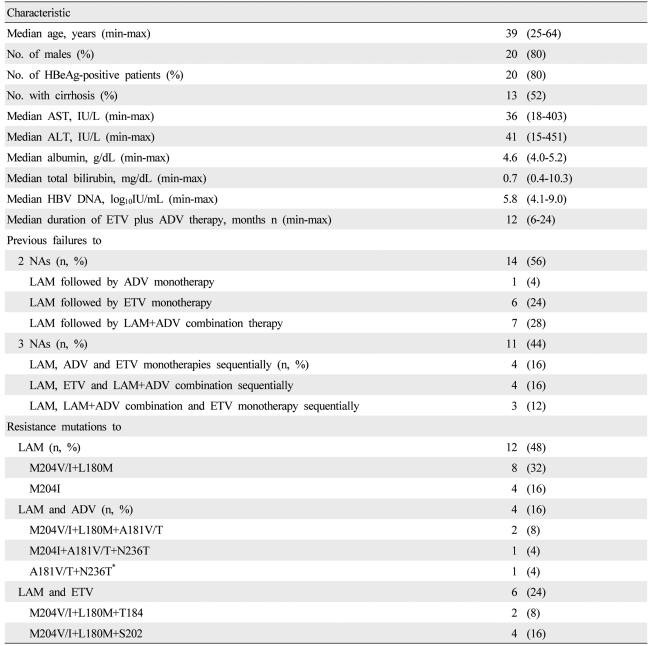
In total, 25 patients with nucleoside-refractory CHB were enrolled in the study. All patients exhibited virological breakthrough (VBT) before starting combination therapy, as defined by an increase in HBV DNA levels of at least 1 log10copies/mL (log) from the nadir during rescue therapy such as ADV add-on and high-dose ETV therapy. Patients were considered to be refractory if they had an inadequate virological response with or without documented genotypic mutations while they were receiving antiviral therapy. An inadequate virological response was defined as an HBV DNA level >4 log at 6 months of rescue therapy. The patients were followed up regularly at 3-month intervals.
The inclusion criteria were as follows: patients were positive for hepatitis B surface antigen (HBsAg) at least 1 year before commencing combination therapy, had HBV DNA level >4 log, and received both ADV and ETV once daily for at least 6 months. Several biochemical parameters and HBV DNA levels were tested at baseline and every 3 months during combination therapy.
None of the patients were co-infected with hepatitis C virus or human immunodeficiency virus or concomitant liver diseases such as alcoholic liver disease or autoimmune liver disease. The IVR was defined as HBV DNA levels below 4 log at 6 months of combination therapy. The continued virological response (CVR) was defined as HBV DNA levels below 4 log after the 6 month time point.
Analysis of virological markers
Routine biochemical tests were performed during therapy by using standard procedures. HBsAg, hepatitis B e-antigen (HBeAg), and anti-hepatitis B e-antigen (anti-HBe) were tested by using a commercial radioimmunoassay kit (Abbott Laboratories, Chicago, IL, USA). HBV DNA levels were determined quantitatively by real time PCR (Roche Molecular Systems, Branchburg, NJ, USA) that had a detection limit of 70 copies/mL. The HBV genotype was not examined because all patients were known to have genotype C.
8,
9
Genotypic analysis
All baseline patient samples were analyzed for genotypic resistance before starting the combination therapy and at the end of treatment. On-treatment was defined as the time point just before starting the ADV-ETV combination therapy. Off-treatment was defined as the end of combination therapy. Moreover, when VBT was observed, patient samples were analyzed again to identify the mutations that were associated with the genotypic resistance. Genotypic resistance was investigated by restriction fragment mass polymorphism analysis, as described previously by Lee et al (Green-Cross Medical Laboratories, Giheung, Korea).
10
Statistical analysis
Statistical testing was performed by using SPSS version 12 (SPSS Inc., Chicago, IL, USA). The data are reported as median (range). The HBV DNA data were logarithmically transformed prior to analysis. Continuous variables were compared by using the Mann-Whitney U test and Student's t test. Categorical data were compared by using the Pearson Žć2 test or Fisher's exact test. The cumulative probability of achieving CVR was estimated by using the Kaplan-Meier method. Factors associated with an IVR were analyzed by univariate and multivariate analysis. A P-value less than 0.05 was considered to be statistically significant.
DISCUSSION
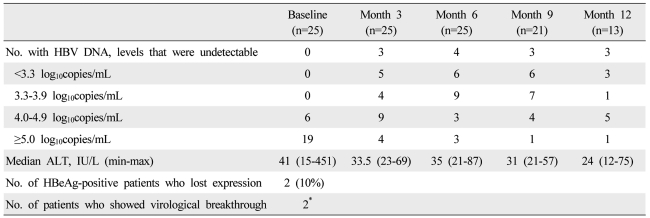
Combination therapy was moderately efficacious for those refractory patients with CHB who participated in the study. The IVR rate was 76%. Moreover, the proportion of patients who had high viral loads (Ōēź5.0 log) dropped from 76% at baseline to 5% after 6 months of treatment. Two patients (10%) lost HBeAg, and the biochemical response rate after the first 6 months of treatment was 76%.
With regard to the IVR in this study, most of the patients who had high viral loads at baseline developed intermediate or low viral loads after combination therapy. While an IVR of 76% may not appear to be particularly high, it can actually be considered to be a good response when one realizes that these patients are multi-drug resistant patients with CHB who previously responded poorly to monotherapies with all other oral NAs.
CVR was defined as the HBV DNA levels below 4 log after the 6 month time point. The theoretical background of this definition was originated from the previous studies. Lim et al defined the virological response as the same level.
11 The patients exposed to less NAs showed better treatment response. Lai et al
12 suggested that HBV DNA suppression should continue until the level decreases to less than 10
4 copies/mL to prevent the development of complication.
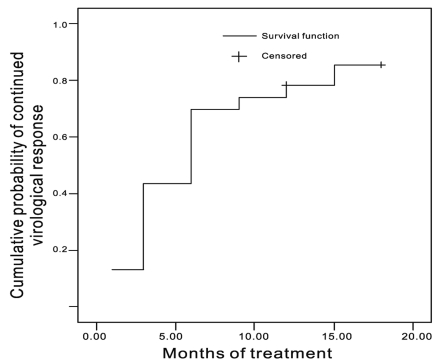
The CVR dropped from 76% at 6 months to 54% at 9 months (
Fig. 1). This is likely to be due to a blip phenomenon: blips can be defined as small fluctuations in HBV DNA levels that do not represent. Another reason was that the four patients with a good response dropped out of the study after 9 months because of the financial burden of the drugs. Actually, their HBV DNA levels at 6 months were 3.2, 3.0, 2.0 and 1.0 log, respectively.
The cumulative probability of achieving CVR was shown by
Figure 2. Kaplan-Meier analysis could be applied only if rebound HBV DNA levels after CVR were not observed in any of the patients during the observation period. Therefore, two patients with breakthroughs at 6 and 15 months, respectively, were excluded for Kaplan Meier analysis. We strongly suspect that these two patients showed poor compliance at these time points.
The sequential use of NAs monotherapies increases the risk of selection of multi-drug resistant strains.
13 As a result, the development of multi-drug resistance to LAM, ADV and ETV is becoming a significant problem in Korea.
14 Combination therapy with ETV and tenofovir has been recommended for the treatment of patients who are resistant to LAM and ADV.
15 However, since tenofovir is currently not available in Korea, we speculated that ADV-ETV combination therapy may be a good alternative for patients with multi-drug resistance. However, Korean government insurance only permits the use of one antiviral agent in principle, which means that combination therapy places a considerable financial burden on patients in Korea. Drug adherence should be approached as a social problem rather than as a patient's problem.
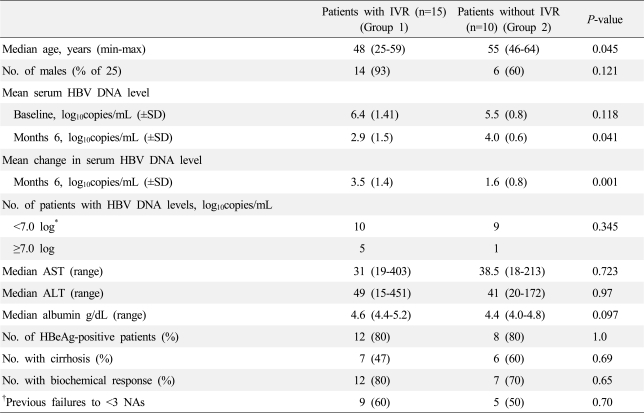
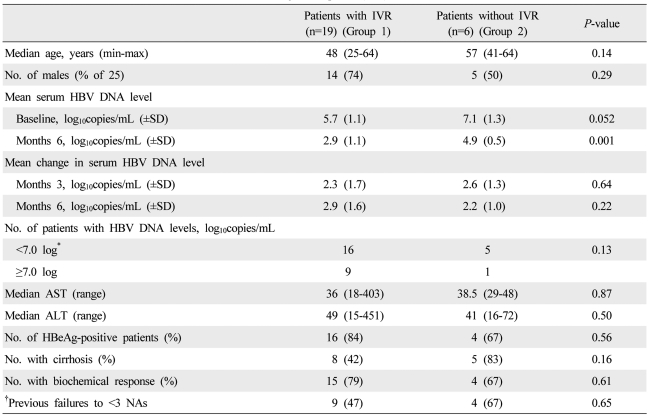
Baseline clinical prognostic factors that could predict IVR or CVR could not be identified. However, if the patients were divided into two groups according to whether they exhibited a decrease in HBV DNA by more than 2 log after 6 months of treatment, only age differed significantly between the two groups upon univariate analysis (data not shown). Multi-variate analysis was performed for four significant factors (
P-value <0.1 in univariate analysis). However, none of these factors were proven to be prognostic factors for IVR upon multi-variate analysis (data not shown). Another study showed that patients who were refractory to ADV and who achieved IVR with ETV monotherapy had higher baseline ALT and AST levels than those who did not achieve IVR.
14 A recent study also showed that low pre-treatment HBV DNA levels and previous exposure to fewer NAs were associated with a virological response.
11
The loss of HBeAg or a biochemical response during the early treatment period could not be analyzed because too few of the patients had HBeAg (n=20) or high ALT levels (n=16) at baseline. One patient exhibited a biochemical VBT after 15 months of treatment. This patient confessed that he had sometimes failed to take his medication around 15 months after treatment. A new report about medication compliance that was published recently found that nine of 10 patients who experienced an initial VBT, but did not have confirmed genotypic resistance, had undetectable HBV DNA levels at the most recent follow-up, which was on average 7 months after the initial VBT. These data suggest that medication noncompliance can be a common cause of VBT.
16
With regard to the genotypic resistance data of the present study, the genotypic resistance profile at the on-treatment time point was compared to that at the off-treatment time point. Mutations associated with LAM and ETV resistance were maintained in four of five patients, but ADV resistance had disappeared in three patients by the off-treatment time point. We speculate that ETV can somehow control ADV-resistant viruses, but ADV cannot control the virus with ETV-and LAM-resistant viruses. Indeed, Reijnders et al
17 reported that viruses with ADV-resistant mutations are expected to remain sensitive to ETV. Moreover, in vitro study reported that viruses with the ADV-resistance rtN236T mutation remains sensitive to ETV, albeit with a <3-fold change in IC
50, while viruses with the ADV-resistance rt181V/T mutations remain completely sensitive to ETV.
18,
19
The limitations of the present study were, first, that the follow-up period was short. Second, drug adherence in the patients was not checked. The three patients who had taken ETV showed an inadequate response to the ADV-ETV combination therapy and we suspect poor drug adherence in these patients. Third, four patients dropped out because of the economic burden of the drug regimen. Two patients completed 6 months of treatment, and the other two patients completed 9 months of treatment. Finally, the present study was retrospective.
In conclusion, patients who experienced ADV or ETV resistance after becoming refractory to LAM showed a good IVR in response to ADV-ETV combination therapy. This combination therapy appeared to be more effective for ADV-resistance mutations than ETV resistance mutations. This combination regimen is thus a good alternative or bridge therapy for patients with multi-drug resistance until tenofovir become available. Further studies with larger number of patients are needed to determine the exact efficacy of this regimen and the factors that favor a good IVR.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



