| Korean J Hepatol > Volume 17(3); 2011 > Article |
ABSTRACT
A leukemoid reaction is defined as reactive leukocytosis exceeding 50,000/mm3, with a significant increase in early neutrophil precursors, and can be a paraneoplastic manifestation of various malignant tumors. A 71-year-old male patient complained of decreased appetite, fatigue, and abdominal fullness. He had a palpable, firm liver, and laboratory investigations suggested leukemoid reaction. Liver dynamic computed tomography revealed a hypervascular mass, and an ultrasound-guided fine-needle aspiration of the mass confirmed hepatocellular carcinoma (HCC) with a sarcomatoid component. The leukocyte count of the patient had increased to 147,800/mm3, and he died 10 days after admission. This is a rare case of leukemoid reaction in a patient with sarcomatous HCC.
A leukemoid reaction refers to a reactive leukocytosis exceeding 50,000/mm3. Leukemoid reactions are generally benign and not fatal of themselves, but leukemoid reaction can imitate more serious condition such as chronic myelogenous leukemia. Leukemoid reaction can also be a paraneoplastic manifestation of several cancers such as lung, gastrointestinal, genitourinary, ovarian, head and neck cancers,1 and hepatocellular carcinoma (HCC).2 Sarcomatoid hepatocllular carcinoma likely forms a larger tumor than ordinary HCC but prognosis of this disease is as yet unclear. Leukemoid reaction in patients with sarcomatoid hepatocelluar carcinoma was fatal in our case.
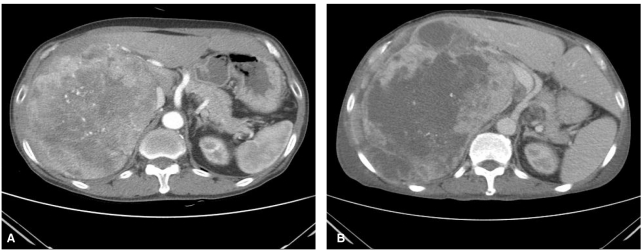
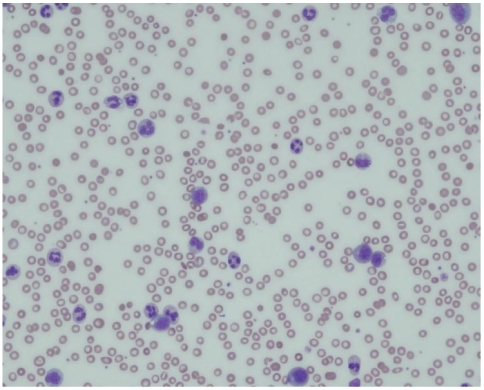
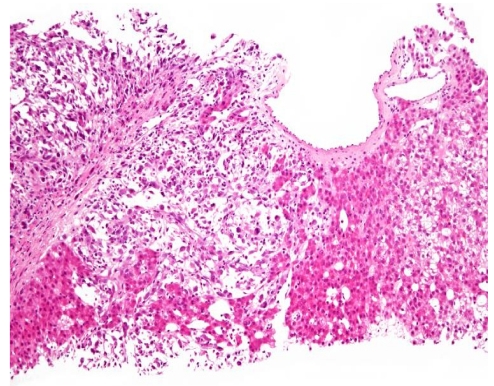
A 71-year-old male patient was admitted to hospital due to right upper quadrant discomfort. Seven months ago, he had been referred to the Department of Surgery because of huge liver mass. Liver dynamic computed tomography (CT) revealed 20 cm sized huge liver mass looked like hepatocelluar carcinoma (Fig. 1A), but had not undergone further diagnostic evaluation and treatment due to unresectability and refusal of the patient. For recent three weeks, he had experienced diminished appetite, fatigue and abdominal fullness. Abdominal examination showed palpable firm liver, 4 cm below the right costal margin without tenderness. His body temperature was normal. Laboratoy investigation revealed a hemoglobin of 8.0 g/dL, total leukocyte count of 68,800/mm3 with a differential count of neutrophils 95%, lymphocytes 3% a monocytes 2%. Peripheral blood smear showed leukemoid reaction (Fig. 2). Other laboratory tests showed, bilirubin 0.5mg/dL, serum aspartate aminotransferase 60 U/L, alanine aminotransferase 10 U/L, alkaline phosphatase 2,820 U/L, total protein 5.8 g/dL with albumin 2.4 g/dL, prothrombin time 1.49 (INR), C-reactive protein (CRP) 21.63 mg/dL. Platelets, blood glucose, creatinine, electrolytes were within normal limits. Alpha fetoprotein (AFP) level was 1.3 ng/mL (normal range below 8.1), and protein induced by the absence of vitamin K or antagonist II (PIVKA II) was over 2,000 mAu/mL (normal range below 40). Viral markers were negative. He had normal chest X-ray and normal urine analysis results. The blood culture sets, urine and sputum culture sets were all negative. All of our work-up of infective etiology were negative. Liver dynamic CT examination on admission showed increased size of hypervascular mass (from 20 cm to 24 cm) with central necrosis in right hepatic lobe with newly developed multiple small attenuated lesion in both hepatic lobes and enhancing peritoneal nodules (Fig. 1B) compared with previous CT of 8 months ago. An ultrasound guided fine needle aspiration from liver lesion was performed and confirmed HCC with sarcomatoid component (Fig. 3). The patient was treated with systemic antibiotics, nutritional support and non-opioid analgesics. After three days of admission, he complained of aggravated abdominal pain and laboratory investigation showed extreme leukocytosis of 110,000/mm3 with a differential count of neutrophils 97%, lymphocytes 2% a monocytes 1%. Therefore, the pain was managed with morphine. In spite of supportive management, anorexia and lethargy were aggravated. The leukocyte count had increased to 147,800/mm3, and the patient succumbed after 10 days of admission.
A extreme leukocytosis can be caused by severe infection, a malignancy or use of certain drugs and a leukocytosis exceeding 50,000/mm3 with a significant increase in early neutrophil precursors is referred to as a leukemoid reaction.3 Not much is known and studied about the incidence and course of leukemoid reaction.4
Cancer associated leukemoid reaction can be caused by increased granulocyte colony stimulating factor, or other growth factors, which are considered to be produced by the malignant cells.5 A paraneoplastic leukemoid reaction can be seen in patients with various cancers like bronchus carcinoma, urinary tract carcinoma, oropharyngeal carcinoma, esophageal carcinoma, cholangicarcinoma, cervix or ovarian carcinoma, melanoma, multiple myeloma.1 However, HCC rarely presents with leukemoid reaction.2
HCC with sarcomatous feature is a rare neoplasm that is found about 2% of surgically resected HCCs.6-8 Some kind of anticancer therapy, such as transarterial chemotherapy or chemoembolozation, radiofrequency ablation, are presumed to promote sarcomatous change in HCC.9 The patient in the present case had not undergone anticancer therapy, but huge mass can lead to ischemic change similar to the effect of anticancer therapy. Therefore, we suspect that huge mass may effect on the emergence of sarcomatous change.
Sarcomatous HCC has a high incidence of metastasis and likely forms a larger tumor than ordinary HCC, the interior of the tumor often shows hemorrhage and necrosis like present case.10,11 It is likely that tumor progression and necrosis may be associated with an increase in inflammatory cytokine response resulting in leukemoid reaction.
Only a few cases of leukemoid reaction in patient with ordinary HCC has been reported,12-14 and leukemoid reaction in various kinds of sarcoma have been reported.4,15,16
Due to the limited data and case reports, we are not able to conclude the association between sarcomatous HCC with leukemoid reaction, but, this is the first report in the English literature of leukemoid reaction in a patient with sarcomatous HCC.
REFERENCES
2. Ochoa JF. Hepatoma presenting as a single cavitary lung mass and leukemoid reaction. Chest 1981;80:250. 6265157.

3. Hoffman R, Benz EJ, Shattil SJ, Furie B, Silberstein LE, McGlave P, et al. Hematology: basic principles and practice. 2009. 5th ed. Philadelphia: Churchill Livingstone; p. 702.
4. Halkes CJ, Dijstelbloem HM, Eelkman Rooda SJ, Kramer MH. Extreme leucocytosis: not always leukaemia. Neth J Med 2007;65:248-251. 17656811.

5. Hocking W, Goodman J, Golde D. Granulocytosis associated with tumor cell production of colony-stimulating activity. Blood 1983;61:600-603. 6600635.


6. Nasser SM, Choudry UH, Nielsen GP, Ott MJ. A leukemoid reaction in a patient with a dedifferentiated liposarcoma. Surgery 2001;129:765-767. 11391379.


7. Maeda T, Adachi E, Kajiyama K, Takenaka K, Sugimachi K, Tsuneyoshi M. Spindle cell hepatocellular carcinoma. A clinicopathologic and immunohistochemical analysis of 15 cases. Cancer 1996;77:51-57. 8630939.


8. Kakizoe S, Kojiro M, Nakashima T. Hepatocellular carcinoma with sarcomatous change. Clinicopathologic and immunohistochemical studies of 14 autopsy cases. Cancer 1987;59:310-316. 2433017.


9. Kojiro M, Sugihara S, Kakizoe S, Nakashima O, Kiyomatsu K. Hepatocellular carcinoma with sarcomatous change: a special reference to the relationship with anticancer therapy. Cancer Chemother Pharmacol 1989;23(Suppl):S4-S8. 2466583.


10. Ueda K, Matsui O, Kawamori Y, Nakanuma Y, Kadoya M, Yoshikawa J, et al. Hypervascular hepatocellular carcinoma: evaluation of hemodynamics with dynamic CT during hepatic arteriography. Radiology 1998;206:161-166. 9423667.


11. Miyata R, Shimazu M, Kawachi S, Tanabe M, Aiura K, Wakabayashi G, et al. Left trisegmentectomy and combined resection of the inferior vena cava, without reconstruction, for giant cystadenocarcinoma of the liver. J Hepatobiliary Pancreat Surg 2005;12:272-276. 15995820.


12. Jain P, Roop Rai R, Jha A. Hepatocellular carcinoma manifesting as cavitary lung metastases and leukemoid reaction. J Gastrointestin Liver Dis 2008;17:355-356. 18836637.

13. Tilak V, Rai M, Singh VP, Rai AK, Raman R. Hepatocellular carcinoma presenting as neutrophilic leukaemoid reaction-a rare entity. J Indian Med Assoc 2007;105:462. 464-465. 18236911.

14. Rosenfeld CS, Sloan B, Egitto DJ. Multiple hepatoma with leukemoid reaction. N Y State J Med 1982;82:359-361. 6283440.

Figure┬Ā1
Computed tomography (CT) findings. (A) 7 months prior to the patient's admission. (B) CT scan obtained on admission showing a hypervascular mass of increased size with central necrosis in the right hepatic lobe.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




