KASL clinical practice guidelines for liver cirrhosis: Varices, hepatic encephalopathy, and related complications
Article information
PREAMBLE
Background and aims
Patients with decompensated cirrhosis with complications have a very poor prognosis and require careful management. Varices are common complications in patients with cirrhosis. Although the prognosis of variceal bleeding has improved with recent advances in diagnosis and treatment, the mortality rate remains 12–22%. Hepatic encephalopathy (HE) is known to occur in 10–14% of patients with cirrhosis and 16–21% of patients with decompensated cirrhosis. More than 20% of cirrhotic patients who visit emergency rooms in Korea present with HE. Therefore, cirrhosis is a serious disease in Korea and requires specific Korean guidelines for diagnosis, treatment, and prevention. In 2005, the Korean Association for the Study of the Liver (KASL) enacted a clinical practice guideline (CPG) for the treatment of cirrhosis complications including ascites, hepatorenal syndrome, varices, and HE. In 2011, the guidelines for the treatment of cirrhosis were revised to integrate antifibrotic treatment and update the diagnosis and treatment advice for variceal bleeding, cirrhotic ascites, and HE. In 2017, the CPG for liver cirrhosis was revised for ascites and related complications. At this time, KASL is revising the CPG for liver cirrhosis to address varices and HE following ascites and related complications. To date, many studies have addressed the prevention and treatment of gastroesophageal variceal bleeding and HE, and many guidelines have been based on those studies, but most of them contain foreign data that are difficult to apply to Korean clinical practice. Therefore, these revised guidelines for the treatment of varices and HE are offered for Korean practice to reflect the latest research results and extensive discussions within the revision committee. This guideline contains the opinions of experts and is intended to be a practical reference for the care of patients with varices and HE; it is not an absolute standard of care. The best choices for each patient’s care vary from case to case, and the judgment of the doctor in charge is important. As medical evidence and new findings accumulate in the future, these guidelines will require ongoing supplementation and revision. This guideline may not be modified or altered without permission.
Target population
This guideline discusses patients with varices, HE, and related complications (esophageal varices [EVs] and bleeding, gastric varices and bleeding, portal hypertensive gastropathy, covert and overt HE) caused by liver cirrhosis. It is intended for clinicians and other medical personnel who are in charge of diagnosing and treating patients with liver cirrhosis. This guideline is also intended to provide practical clinical and educational information and directions for resident physicians and fellows in training, practitioners, and their trainers and supervisors.
Development, funding, and revision process
Comprising 14 hepatologists, the Clinical Practice Guideline Committee for Liver Cirrhosis: Varices, HE, and related complications (the Committee) was organized by the KASL Board of Executives. Funding for the revisions was provided by KASL. Each committee member collected and analyzed source data in his or her own field, and the members then wrote the manuscript together.
Literature review
The Committee selected keywords and questions using PICO (Patient/Problem, Intervention, Comparison, Outcome) assessments and systematically collected and reviewed international and domestic literature available in PubMed, MEDLINE, KoreaMed, the Korean Medical Database, and other databases. In addition to published articles, abstracts of important meetings published before January 2019 were evaluated.
Levels of evidence and grades of recommendation
The Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) system (Table 1) was applied to grade the evidence and recommendations. The levels of evidence are based on the possibility of change in the estimate of clinical effect by further research and are described as high (A), moderate (B), or low (C). The recommendations are also classified as strong (1) or weak (2) by the GRADE system based on the quality of evidence, the balance between the desirable and undesirable effects of an intervention, generalizability, and socioeconomic aspects (including cost and availability). Each recommendation is labeled with the level of relevant evidence (A–C) and corresponding recommendation grade (1, 2) as follows: A1, A2, B1, B2, C1, C2.
List of key questions
The Committee selected the following key questions about varices, HE, and related complications to cover in this guideline.
Key varix-related questions
1) How should varices be monitored?
2) Who needs monitoring for varices?
3) How can the development and progression of EVs be prevented?
4) Who needs treatment to prevent initial esophageal variceal bleeding?
5) What is the proper management for preventing initial esophageal variceal bleeding?
6) How can acute esophageal variceal bleeding be diagnosed?
7) What is the appropriate pharmacological treatment for acute esophageal variceal bleeding?
8) What is the proper endoscopic treatment for acute esophageal variceal bleeding?
9) What are the options for rescue treatment when endoscopic treatment of acute variceal bleeding fails?
10) What is the primary treatment to prevent EVs from rebleeding?
11) What are the options for rescue treatment when primary treatment to prevent EVs from rebleeding fails?
12) Who needs treatment to prevent gastric variceal bleeding?
13) What is the proper treatment to prevent gastric variceal bleeding?
14) What is the proper treatment of acute gastric variceal bleeding?
15) What is the primary treatment to prevent gastric varices from rebleeding?
16) How should portal hypertensive gastropathy be classified?
17) How should portal hypertensive gastropathy be managed?
Key HE-related questions
1) How should HE be diagnosed and classified?
2) How should overt HE be defined and diagnosed?
3) What are the precipitating factors of overt HE?
4) What differential diagnoses should be considered in diagnosing overt HE?
5) Is the measurement of serum ammonia helpful in diagnosing overt HE?
6) Is radiologic image evaluation of the central nervous system helpful in diagnosing overt HE?
7) What neurophysiological or neuropsychological tests are clinically necessary to diagnose overt HE?
8) How should the acute phase of overt HE be treated, and how should recurrence be prevented?
9) Are branched chain amino acids helpful in treating and preventing overt HE?
10) Is L-ornithine-L-aspartate (LOLA) helpful in treating and preventing overt HE?
11) Is proper education helpful in preventing the recurrence of and readmission for HE?
12) How should covert HE be defined and diagnosed?
13) What is the clinical significance of covert HE?
14) How should covert HE be treated?
15) How should the quality of life of HE patients be assessed? Does treating HE improve patient quality of life?
Review of the manuscript and approval process
Each manuscript written by members was reviewed and approved through meetings of the Committee. An updated manuscript was reviewed at a meeting of the advisory board and opened to a public hearing attended by KASL members, members of related organizations, and representatives from patient associations. The final manuscript was approved by the KASL Board of Executives.
Release of the guidelines and plan for updates
The revised guideline (The KASL Clinical Practice Guidelines for Liver Cirrhosis: Varices, Hepatic Encephalopathy and Related Complications) was released at a KASL meeting on 22 June 2019. The Korean version of the guideline is available on the KASL website (http://www.kasl.org).
VARICES
Varices are a frequent complication of liver cirrhosis and a leading cause of mortality in patients with liver cirrhosis. Varices were present in 52.2% of patients who received endoscopy for variceal screening [1], and the incidence of varices was significantly higher in patients with Child-Pugh class B/C than in those with Child-Pugh class A (35–43% vs. 48–72%) [1,2]. Portal hypertension, which is the most common complication of liver cirrhosis, is the main determinant in the development of varices. Increased intrahepatic vascular resistance to portal flow leads to the development of portal hypertension, which is aggravated by splanchnic vasodilatation and an increase in portal blood flow caused by hyperdynamic circulation [3-5]. When the portal pressure increases above a threshold, collaterals develop at the site of communication between the portal and systemic circulation, of which varices are the most important. With the aggravation of portal hypertension, the collaterals grow and eventually rupture. Bleeding from varices is a major complication of portal hypertension and a leading cause of mortality in patients with liver cirrhosis. Therefore, preventing variceal development and progression, preventing bleeding from varices, appropriately managing acute bleeding from varices, and preventing variceal rebleeding are critical in patients with liver cirrhosis.
The incidence of varices in cirrhotic patients without varices at baseline is 5–9% at 1 year and 14–17% at 2 years [6,7]. The main risk factor for variceal development in these patients is a higher hepatic venous pressure gradient (HVPG) [6]. Small EVs often progress to large varices; the incidence of progression from small to large EVs is 12% at 1 year and 25% at 2 years. The independent risk factors of EV progression are alcoholic cirrhosis, decompensated disease, and splenomegaly [7]. The 1-year incidence of variceal bleeding in patients with cirrhosis and varices without a previous history of bleeding is approximately 12% (5% for small varices and 15% for large varices), and the main risk factors of bleeding are larger varices, the presence of redness over the varices, and decompensated disease [8]. Although the mortality rate has decreased significantly during the past several decades thanks to improvements in diagnostic and therapeutic modalities [9,10], it remains as high as 12–22% [11-14]. In addition, rebleeding is frequent, up to 60% within 1 year, without appropriate treatment to prevent it [15].
Surveillance of varices
Endoscopic surveillance of varices
Given the high prevalence of varices and poor prognosis with variceal bleeding, monitoring varices is important in patients with liver cirrhosis. Therefore, upon first diagnosis with liver cirrhosis, endoscopy should be performed to look for varices and assess the risk of bleeding. Diagnosis of liver cirrhosis is not difficult in patients with decompensated liver cirrhosis accompanied by ascites or variceal bleeding, but a liver biopsy is needed to diagnose patients with compensated cirrhosis who have no clinical symptoms or signs. However, liver biopsy is an invasive procedure with a risk of serious complications [16]. Furthermore, doubt has been cast on the accuracy of liver biopsy because of the risk of sampling errors [17,18] and intra- and interobserver variability [18,19].
Liver cirrhosis can disappear with appropriate treatment of the underlying liver disease [20,21], though portal hypertension can accompany the severe stage of fibrosis (F3) [22,23]. Various practice guidelines recommend surveillance for hepatocellular carcinoma in patients with liver fibrosis, even before the development of cirrhosis [24,25]. Therefore, the alternative term compensated advanced chronic liver disease (cACLD) has been proposed for patients with severe fibrosis (F3) and compensated liver cirrhosis to better reflect that the spectrum of severe fibrosis and cirrhosis is a continuum in asymptomatic patients and that distinguishing between these two conditions is often clinically impossible [26]. A liver stiffness value, measured by transient elastography, of <10 kPa can rule out cACLD, and a value between 10 and 15 kPa is suggestive of cACLD but needs further tests for confirmation. A value >15 kPa is highly suggestive of cACLD [26]. Endoscopic surveillance of all patients with cACLD can cause problems, such as an increase in medical costs due to an increase in unnecessary tests. Therefore, noninvasive screening tests have been proposed for patients with EVs, especially those whose EVs have a high risk of bleeding, to reduce unnecessary endoscopic surveillance. The Baveno VI criteria suggest that endoscopic surveillance can be avoided in cACLD patients with a liver stiffness <20 kPa and a platelet count >150×109/L because they are at very low risk for varices that need to be treated [26]. Augustin et al. [27] expanded the Baveno VI criteria to say that endoscopic surveillance can be avoided in cACLD patients with liver stiffness <25 kPa and a platelet count >110×109/L. However, considering that noninvasive screening for varices that need to be treated is not particularly reliable [28,29] and endoscopy is more easily accessed in Korea than in Western countries, we do not deem screening by noninvasive test to be useful in Korea.
Surveillance of EVs
The incidence of EV development in cirrhotic patients without varices is 5–9% at 1 year and 14–17% at 2 years [6,7]. Small EVs progress to large varices at the rate of 12% after 1 year and 25% after 2 years [7]. Therefore, endoscopic surveillance should be performed more frequently in patients with small EVs than in those without EVs. In addition, because the type of underlying liver disease (e.g., alcoholic cirrhosis) and liver function (e.g., decompensated cirrhosis) are risk factors for the progression of EVs, they should be taken into account when determining the surveillance interval. Endoscopic surveillance should be performed at 2–3-year intervals in patients with compensated liver cirrhosis and at 1–2-year intervals in those with decompensated liver cirrhosis [30,31].
EVs can be classified as large or small according to their size, with a breakpoint at 5 mm in diameter [32], or they can be classified as F1 (linearly dilated, small and straight varices), F2 (beady varices, tortuous and occupying less than one third of the esophageal lumen), or F3(nodular varices, large and occupying more than one third of the esophageal lumen) [33]. However, because the F2 and F3 classifications are fairly subjective and prophylactic treatment is recommended both for F2 and F3, F1 is usually classified as small, and F2 and F3 are classified together as large.
[Recommendations]
1. In patients diagnosed with liver cirrhosis, screening endoscopy is recommended to determine the presence of varices and assess the risk of bleeding. (A1)
2. In endoscopy, EVs are classified as small (F1) and large (F2 or F3), and the presence of redness should be evaluated. (B1)
3. To identif y the development and progression of EVs, endoscopic surveillance should be performed at 2–3-year-intervals in patients with compensated liver cirrhosis and at 1–2-year intervals in those with decompensated liver cirrhosis. The frequency of endoscopic surveillance could be modified according to the type and severity of underlying liver disease. (B1)
Preventing the formation and progression of EVs
Appropriate treatment for the underlying liver disease can improve liver fibrosis, which could improve portal hypertension and prevent the development of complications. In patients with hepatitis B virus-related liver cirrhosis, the cirrhosis disappeared from the liver biopsy reports of 74% after 5 years of treatment with tenofovir disoproxil fumarate [20], and in a meta-analysis, hepatic histologic improvement was observed in chronic hepatitis C patients treated with pegylated interferon±ribavirin [34]. In an earlier study of patients with nonalcoholic fatty liver disease, the degree of weight loss correlated with the degree of histologic improvement [35]. Furthermore, the incidence of EVs was significantly lower in patients with a sustained virologic response (SVR) to pegylated interferon+ribavirin treatment than in those without an SVR [36-38]. In a recent study, portal pressure was significantly lower in patients with an SVR to direct-acting agents than in those without an SVR in patients with hepatitis C virus-related liver cirrhosis [39].
Because the development of GEVs is a direct consequence of portal hypertension, reducing the portal pressure through the use of nonselective beta-blockers (NSBBs) from the early stage of liver cirrhosis could theoretically ameliorate the formation of GEVs. However, a placebo-controlled study to determine whether NSBBs could prevent the formation of varices in 213 patients with cirrhosis and portal hypertension without GEVs, the incidence of varices or bleeding from varices did not differ between timolol group and the placebo group (39% vs. 40%, P=0.89), and serious adverse events developed more frequently in the timolol group than the placebo group (18% vs. 6%, P=0.006) [6]. Therefore, the use of NSBBs to prevent the formation of varices is not recommended.
Several studies have evaluated whether NSBBs can prevent or delay the growth of small varices, and the results conflict. One study found a significant reduction in the rate of progression to large EVs in the nadolol group compared with the placebo group in patients with cirrhosis and small EVs (7% vs. 31% at 2 years, 20% vs. 51% at 5 years; P<0.001) [40], but another study showed that propranolol offered no benefit for the prevention of progression to large varices (23% in the propranolol group vs. 19% in the placebo group, P=0.786), even though the reduction in portal pressure was significantly greater in the propranolol group [41]. A recent meta-analysis suggests that NSBBs are not effective in preventing the progression from small to large varices [42]. Another study found that the incidence of progression to large varices across 24 months was significantly lower in the carvedilol group than the placebo group (20.6% vs. 38.6%, P=0.04), leading those researchers to suggest that carvedilol is a safe and effective way to delay the progression of small to large EVs in patients with cirrhosis [43].
Carvedilol reduces portal pressure by means of an anti-a1-mediated decrease in intrahepatic resistance and splanchnic vasoconstriction. Because intrahepatic vasoconstriction is the main pathologic mechanism in the development of portal hypertension during early-stage liver cirrhosis, it could be more effective than other medications in preventing the progression of varices in patients with early-stage cirrhosis [44]. However, further studies are needed to confirm the effects of carvedilol.
[Recommendations]
1. Appropriate treatment for the underlying liver disease is recommended to prevent the formation of EVs. (A1)
2. NSBBs (propranolol and nadolol) are not recommended to prevent the formation of EVs in cirrhotic patients without EVs. (A1)
3. In patients with small EVs that are not red, NSBBs (propranolol and nadolol) or carvedilol could be considered to prevent the progression of EVs. (B2)
Prevention of first variceal bleeding in patients with EVs
In patients with liver cirrhosis and EVs, variceal bleeding occurs at a yearly rate of 5–15% of cases. Active prevention of the first variceal bleeding is indicated in patients at a high risk of bleeding, such as patients with large varices (F2, F3), decompensated cirrhosis, or varices with red color signs on endoscopy [8,45].
Prevention of first variceal bleeding in patients with small EVs
In cirrhotic patients with small EVs, the risk of bleeding is low (3% at 2 years and 8% at 4 years) and remains low in patients whose varices remain small at the follow-up endoscopy, though it increases significantly when the varices become large. An increase in Child-Pugh score during follow-up appears to be a significant predictor of enlarged varices and thus an increase in bleeding risk [46]. The prevention of first bleeding in patients with small EVs depends on their risk of bleeding. Patients with small varices with red color signs on endoscopy or decompensated cirrhosis have an increased risk of bleeding and should consider using NSBBs [26,47].
Prevention of first variceal bleeding in patients with large EVs
NSBBs and EVL
Meta-analyses of randomized controlled trials (RCTs) have shown that the use of NSBBs can prevent first variceal bleeding in cirrhotic patients with large EVs [48,49]. A study comparing NSBBs and EVL as primary prophylaxis in patients with high-risk EVs found no significant difference between them in bleeding rates (relative risk [RR], 0.86; 95% confidence interval [CI], 0.55–1.35) [50]. A meta-analysis of RCTs evaluating the efficacy of EVL and pharmacological therapy in preventing first EV bleeding in patients with cirrhosis also found no significant difference in the rate of variceal bleeding between the two groups [51]. Another meta-analysis found that EVL significantly reduced the rate of first variceal bleeding and severe adverse events than NSBBs in patients with large EVs [52]. Thus, in most studies, the efficacy of EVL in preventing first variceal bleeding was similar to that of NSBBs, and in some studies, the efficacy of EVL was superior to NSBBs. Therefore, either NSBBs or EVL is recommended for the prevention of first variceal bleeding in patients with large EVs. The choice of treatment should be based on clinician expertise and patient preference, characteristics, contraindications, and adverse events [26,47].
Carvedilol
Carvedilol is known to be more effective in reducing portal pressure than propranolol [53-55]. In a multicenter RCT comparing the efficacy of carvedilol and EVL in preventing first variceal bleeding in cirrhotic patients with large EVs, carvedilol had lower rates of first variceal bleeding (10% vs. 23%, P=0.04), but there was no significant difference in overall mortality or bleeding-related mortality during follow up [56]. In another RCT comparing the efficacy of carvedilol and EVL for primary prophylaxis of EV bleeding, the carvedilol and EVL groups had comparable variceal bleeding rates (8.5% vs. 6.9%, P=0.61) [57]. In a study assessing the efficacy of carvedilol, propranolol, and EVL for the primary prevention of variceal bleeding in patients with large varices, no significant differences among the groups were found in the risk of bleeding (15.4% vs. 10.8% vs. 10.2%, P=0.071), but the incidence of adverse events was the highest in the propranolol group [58]. In studies comparing the efficacy of carvedilol, NSBBs, and EVL for the primary prevention of EV bleeding, carvedilol was similar to NSBBs and EVL or superior to EVL. Therefore, carvedilol can also be used to prevent first variceal bleeding in patients with high-risk EVs.
Combination therapy of EVL and NSBBs
The combination of EVL and NSBBs for the primary prophylaxis of variceal bleeding could have a synergistic effect from the direct eradication of varices by EVL and the reduction of portal pressure by NSBBs. Several studies have compared the efficacy of combination therapy with that of monotherapy based on that hypothesis. In RCTs comparing EVL plus propranolol with EVL alone for preventing first variceal bleeding in patients with high-risk EVs, the combination therapy did not show any difference from EVL alone in first bleed occurrence or mortality during follow up. However, the recurrence of varices was lower in the combination group than in the EVL alone group [59,60]. No difference in the rate of first variceal bleeding was also found between EVL plus nadolol combination therapy and nadolol alone (14% vs. 13%, P=0.90) [61]. However, another study reported that EVL and propranolol combination therapy lowered the rate of first variceal bleeding compared with propranolol alone (6% vs. 31%, P=0.03) [62]. Because most studies have shown that EVL and NSBBs combination therapy for the primary prophylaxis of EV bleeding do not differ in bleeding rate or mortality compared with monotherapy, combination therapy is generally not recommended. However, some studies have reported that EVL and NSBBs combination therapy reduced the rate of first variceal bleeding and variceal recurrence compared with monotherapy. Therefore, combination therapy can be considered in selected patients. A recent meta-analysis of RCTs showed that combination therapy with EVL and NSBBs reduced the rate of first variceal bleeding compared with placebo and isosorbide-5-mononitrate (ISMN) [63].
ISMN
In an RCT of cirrhotic patients with EVs, the ISMN group and propranolol group had no significant difference in bleeding rate, but the mortality rate during follow up was higher in the ISMN group (72.3% vs. 47.8% at 6 years, P=0.006) [64]. In a multicenter RCT comparing EVL, propranolol, and ISMN, the EVL and propranolol groups did not differ significantly, but the EVL group had a significantly lower rate of first variceal bleeding than the ISMN group (7.5% vs. 33% at 2 years, P=0.03) [65]. A multicenter RCT compared propranolol plus placebo with propranolol plus ISMN for the prevention of EV bleeding. The rate of first variceal bleeding did not differ significantly between the groups (10.6% vs. 12.5% at 2 years, P>0.05) [66]. Therefore, ISMN alone or in combination with NSBBs is not recommended for the prevention of first variceal bleeding.
Treatment practices for the prevention of first esophageal variceal bleeding
NSBBs
The advantages of NSBBs include low cost, ease of administration, and not requiring follow-up endoscopies. Propranolol is started at 20–40 mg twice a day and adjusted every 2–3 days until the treatment goal (resting heart rate of 55–60 beats per minute) is achieved. The maximum dose is 320 mg daily in patients without ascites and 160 mg daily in patients with ascites. Nadolol is started at 20–40 mg once a day and adjusted every 2–3 days until the treatment goal is achieved. The maximum dose is 160 mg daily in patients without ascites and 80 mg daily in patients with ascites. Systolic blood pressure should not decrease <90 mmHg [47].
The disadvantages of NSBBs are that about 15% of patients have contraindications to therapy, and another 15% or so require dose reduction or discontinuation because of side effects [47]. Contraindications to NSBBs include sinus bradycardia, insulin-dependent diabetes mellitus, obstructive pulmonary disease, heart failure, aortic valve disease, second- or third-degree atrioventricular heart block, and peripheral arterial insufficiency [67]. Side effects of NSBBs include dizziness, fatigue, general weakness, dyspnea, headache, hypotension, bradycardia, and erectile dysfunction [47,58,66,67]. Discontinuing NSBBs can increase the risk of variceal bleeding and mortality. Thus, treatment with NSBBs should be continued indefinitely [68,69]. In patients with contraindications or discontinuation due to severe side effects or poor compliance with NSBBs, EVL is recommended [68].
In patients with end-stage liver disease, such as refractory ascites or spontaneous bacterial peritonitis, the administration of NSBBs has not yet been established. In cirrhotic patients with refractory ascites, the use of NSBBs can lower arterial pressure, decrease survival time [70], and increase the risk of paracentesis-induced circulatory dysfunction [71]. In addition, among patients with cirrhosis and spontaneous bacterial peritonitis, NSBBs increase the risk of hepatorenal syndrome and acute kidney injury and reduce survival time [72]. However, other studies have reported that the use of NSBBs increased or did not affect survival time in cirrhotic patients with refractory ascites [73,74]. Another study found that treatment with low-dose propranolol (80 mg/day) increased survival time in patients with spontaneous bacterial peritonitis [75]. The role of NSBBs in patients with refractory ascites or spontaneous bacterial peritonitis thus remains uncertain, and clinicians must carefully consider the risks and benefits when deciding whether to administer them. If NSBBs are administered, thorough monitoring of blood pressure and renal function is necessary, and dose reduction or discontinuation should be considered in patients who develop low blood pressure or impaired renal function. Discontinuation of NSBBs can increase the risk of EV bleeding; thus, if NSBBs are stopped, EVL should be considered [26].
Carvedilol
Adjusting the dose of carvedilol is easier than adjusting the dose of NSBBs because it is not guided by heart rate. Carvedilol is started at 6.25 mg once a day (or 3.125 mg twice a day), and after 3 days increased to 6.25 mg twice a day. The maximum dose is 12.5 mg daily. Systolic blood pressure should not be decreased <90 mmHg [47].
EVL
The advantages of EVL are that it can be performed in the same session as screening endoscopy, and it has few contraindications. The disadvantages of EVL are the side effects associated with sedation and the risk of causing dysphagia, esophageal ulcerations, strictures, and bleeding. Although the incidence of side effects is higher with NSBBs, severe side effects, such as ulcer bleeding at the ligation site, are more likely to be associated with EVL [47]. Some studies have reported that proton pump inhibitors (PPIs) significantly reduce the size of post-EVL ulcers or the rate of post-EVL ulcer bleeding [76-78]. In cirrhotic patients, the long-term use of PPIs can increase the risk of spontaneous bacterial peritonitis and HE, so PPIs should be used with caution [79-81]. Meanwhile, because EVL is a local therapy that does not act on the pathophysiology of portal hypertension, not only is it unable to prevent complications other than variceal bleeding, but it also requires follow-up endoscopies to assess variceal recurrence, even after variceal eradication [47], defined as a case in which varices are not seen or become too small to be ligated. Repeat EVL can be performed at intervals of 2–8 weeks until variceal eradication is achieved. Follow-up endoscopies should be performed 1–6 months after variceal eradication and every 6–12 months thereafter [47,59,82].
[Recommendations]
1. In cirrhotic patients with small EVs that have a high risk of bleeding (decompensated cirrhosis or red color signs on endoscopy), the use of a NSBBs (propranolol or nadolol) should be considered to prevent first variceal bleeding. (B1) NSBBs are adjusted every 2–3 days until the resting heart rate reaches 55–60 beats per minute.
2. In cirrhotic patients with large EVs, the use of a NSBBs (propranolol or nadolol), carvedilol, or EVL is recommended to prevent first variceal bleeding. (A1) A combination of NSBBs and EVL can also be considered. (B2)
Diagnosis and management of acute esophageal variceal bleeding
Diagnosis of acute esophageal variceal bleeding
In patients with upper gastrointestinal bleeding, variceal bleeding caused by portal hypertension can be suspected if the patients show jaundice, ascites, HE, splenomegaly, collateral circulation of the abdominal vessels, lower extremity edema, or spider angiomas. A definite diagnosis can be established by endoscopic examination. If blood clots or white nipples appear on the surface of the varices, or if blood is found in the stomach without a potential bleeding focus other than EVs, acute EV bleeding can be diagnosed [45].
General management of acute esophageal variceal bleeding
Acute EV bleeding is a medical emergency requiring intensive care. It is essential to protect the circulatory and respiratory status of the patient regardless of the cause of bleeding. Volume resuscitation via adequate fluid therapy and a packed red blood cell (PRBC) transfusion should be initiated to restore and maintain hemodynamic stability. A recent RCT showed that bleeding-related mortality (5% vs. 9%, P=0.02) and the incidence of serious adverse events (12% vs. 18%, P=0.01) were significantly decreased in the “restrictive” PRBC transfusion group (initiating PRBC transfusion at a hemoglobin threshold of 7 g/dL and maintaining it at 7–9 g/dL) compared with the “liberal” PRBC transfusion group [83]. Improved survival in the restrictive transfusion group might be associated with lower rates of hemostasis failure and serious adverse events. In patients with acute EV bleeding, adequate fluid therapy/PRBC transfusion should be performed while considering age, cardiovascular disease, presence or absence of ongoing bleeding, and hemodynamic status. Excessive fluid therapy/PRBC transfusion may increase the portal pressure and aggravate bleeding from the varices, so that should be taken into account [84]. Regarding correction of coagulopathy, clinical studies of recombinant factor VIIa have not shown a clear benefit, and therefore the routine use of fresh frozen plasma or recombinant factor VIIa is not recommended [85,86]. Although the efficacy of platelet transfusion in patients with acute EV bleeding has not been proven because of a lack of clinical studies, it can be considered in patients with severe thrombocytopenia.
Pharmacological treatment of acute esophageal variceal bleeding
Cirrhotic patients presenting with acute gastrointestinal bleeding have a high risk of developing bacterial infections, therefore initiation of prophylactic antibiotic treatment at the time of admission is necessary. Meta-analyses of RCTs have shown that the use of antibiotic prophylaxis reduces the risk of infections, recurrent bleeding, and bleeding-related death [87,88]. A recent meta-analysis demonstrated that prophylactic antibiotic treatment was associated with a decrease in bleeding-related mortality (RR, 0.79; 95% CI, 0.63–0.98), mortality from bacterial infections (RR, 0.43; 95% CI, 0.19–0.97), development of bacterial infections (RR, 0.35; 95% CI, 0.26–0.47), and rebleeding (RR, 0.53; 95% CI, 0.38–0.74) [88]. However, another recent retrospective study questioned the usefulness of the routine antibiotic prophylaxis in cirrhotic patients experiencing acute variceal bleeding because of a very low incidence of bacterial infections (2%) and mortality (0.4%) in Child-Pugh class A patients with acute variceal bleeding, even in the absence of prophylactic antibiotic treatment [89]. No prospective study has evaluated the usefulness of antibiotic prophylaxis, and therefore the routine use of prophylactic antibiotics is recommended for all cirrhotic patients presenting with variceal bleeding, regardless of their Child-Pugh class. In a previous RCT comparing intravenous ceftriaxone (1 g every 24 hours) and oral norfloxacin (400 mg every 12 hours) for the prophylaxis of bacterial infection in cirrhotic patients with gastrointestinal bleeding, the incidence of proven or possible infections (11% vs. 33%, P=0.003), proven infections (11% vs. 26%, P=0.03), and spontaneous bacterial peritonitis or bacteremia (2% vs. 12%, P=0.03) was significantly lower in the ceftriaxone group [90]. However, controversy remains about whether those results are applicable to general cirrhotic patients because that was study conducted in Spain among patients with advanced cirrhosis, and most of the Gram-negative bacilli detected in the patients receiving oral norfloxacin were norfloxacin-resistant strains. Therefore, it is necessary to select appropriate antibiotics based on local antimicrobial susceptibility patterns. Generally, short-term (maximum 7 days) antibiotic prophylaxis with intravenous ceftriaxone (1 g every 24 hours) is recommended in patients with acute variceal bleeding.
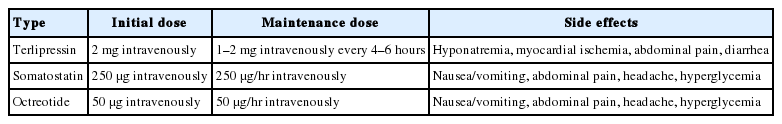
Vasoactive agents, such as vasopressin, terlipressin, somatostatin, and octreotide, are effective in supporting hemostasis in patients with acute variceal bleeding by decreasing portal pressure. In a meta-analysis, the use of vasoactive agents in patients with acute variceal bleeding was significantly associated with a reduction in 7-day mortality (RR, 0.74; 95% CI, 0.57–0.95) and an increase in the hemostasis rate (RR, 1.21; 95% CI, 1.13–1.30) [91]. In patients with suspected variceal bleeding, vasoactive agents should be initiated as soon as possible, together with prophylactic antibiotics, before the diagnostic endoscopy. Vasopressin reduces portal pressure by inducing systemic and splanchnic vasoconstriction, but it is not now recommended for patients with acute variceal bleeding because of the significant side effects, such as an increase in peripheral vascular resistance and reduction in cardiac output and coronary blood flow. Although terlipressin, a synthetic analogue of vasopressin, is the only drug proven to reduce bleeding-related mortality (RR, 0.66; 95% CI, 0.49–0.88) [92], its side effects, such as hyponatremia and myocardial ischemia due to coronary artery vasoconstriction, should be considered [93,94]. A recent meta-analysis [91] and a Korean multicenter RCT [11] comparing three vasoactive agents (terlipressin, somatostatin, and octreotide) found no significant differences among them regarding the hemostasis rate and survival time. In patients with acute variceal bleeding, it is recommended that one of the vasoactive agents should be started as soon as possible (Table 2) and continued for 3–5 days [26,47].
Endoscopic treatment of acute esophageal variceal bleeding
If acute variceal bleeding is suspected, endoscopy should be performed as soon as possible to confirm the hemorrhagic focus and hemostasis. Endoscopic hemostasis should be done when acute EV hemorrhage is confirmed by endoscopy. EVL is the endoscopic treatment of choice for patients with acute bleeding from EVs. Endoscopic injection sclerotherapy (EIS) is no longer recommended as standard treatment for acute EV bleeding because of its higher incidence of treatment failure, bleeding-related mortality, and adverse events compared with EVL [95-99]. In a meta-analysis comparing EVL and EIS in patients with acute EV bleeding, bleeding-related mortality did not differ significantly (RR, 0.95; 95% CI, 0.77–1.17), but the risk of rebleeding was reduced (RR, 0.68; 95% CI, 0.57–0.81) and the rate of variceal eradication was increased (RR, 1.06; 95% CI, 1.01–1.12) in patients undergoing EVL compared with EIS [100]. Most practice guidelines recommend endoscopy within 12 hours after presentation with suspected variceal bleeding, but that recommendation lacks evidence. A previous Taiwanese retrospective study reported that delayed endoscopy (>15 hours after admission) was an independent risk factor of inhospital mortality (odds ratio [OR], 3.67; 95% CI, 1.27–10.39) [101]. In addition, a prospective observational study of 101 patients with acute EV bleeding showed that the 6-week rebleeding rate (18.9% vs. 38.9%, P=0.028) and mortality (27% vs. 52.8%, P=0.031) were significantly lowered in patients undergoing early endoscopy (≤12 hours) compared with those undergoing delayed endoscopy (>12 hours) [102]. However, because those studies were performed without randomization, several confounders that can delay the endoscopy, such as hemodynamic instability, might have influenced the results. Therefore, until the results of large RCTs are reported, endoscopy should be performed as soon as possible in patients with suspected acute EV bleeding. However, the specific timing should be determined by the hemodynamic status of individual patients and the experience and medical resources of the institution.
Once endoscopy and EVL have been performed, early placement of a transjugular intrahepatic portosystemic shunt (TIPS) can be considered in carefully selected patients at high risk for rebleeding. Early TIPS placement reduced the rates of treatment failure and bleeding-related mortality in an RCT [103] of patients with a HVPG >20 mmHg and in an RCT [104] of patients with Child-Pugh class C cirrhosis (score of 10–13) or Child-Pugh class B cirrhosis with active bleeding on endoscopy despite intravenous administration of a vasoactive agent. However, because these two trials excluded patients with Child-Pugh class A cirrhosis, Child-Pugh class B cirrhosis without active bleeding during endoscopy, ChildPugh class C with a score of 14–15, patients >75 years, HCC beyond the Milan criteria, or a creatinine level greater than 3 mg/dL, it should be considered that those study results apply to only a very small portion of patients with acute variceal bleeding. Notably, a recent prospective observational study showed that the 1-year rebleeding risk was significantly decreased (3% vs. 49%, P<0.001), but 1-year survival did not differ between patients with and without a TIPS (66.8±9.4% vs. 74.2±7.8%, P=0.78) [105]. Further studies are needed to evaluate the beneficial effect of early TIPS placement.
Recently, the efficacy of applying hemostatic powder via endoscopy within 2 hours of admission was evaluated in 86 randomized patients with acute variceal bleeding [106]. Cirrhotic patients with acute variceal bleeding received standard medical treatment and were randomized to receive either immediate endoscopy with hemostatic powder application within 2 hours of admission followed by early elective endoscopy the next day (that is, within 12–24 hours of admission) for definitive treatment (EVL for EV bleeding or endoscopic variceal obturation [EVO] for gastric variceal bleeding; study group) or early elective endoscopy only (control group). Improved rates of hemostasis and survival time in the study group suggested the therapeutic potential of endoscopic application of hemostatic powder, an easy procedure requiring minimal expertise.
Rescue treatment for patients with hemostasis failure
Failure to control acute EV bleeding is defined as death or the need to change therapy (defined by one of the following criteria) within 5 days of an acute bleeding episode [107].
- Fresh hematemesis of ≥100 mL of fresh blood ≥2 hours after the start of a specific pharmacological treatment or therapeutic endoscopy
- Development of hypovolemic shock
- 3 g drop in hemoglobin (9% drop in hematocrit) within 24 hours without transfusion
TIPS placement is considered the best rescue treatment for patients with inadequate bleeding control despite combined pharmacological and endoscopic therapy [108]. A prospective observational study to evaluate the efficacy of TIPS in 58 patients who failed to achieve hemostasis after EIS and pharmacological treatment reported that the TIPS achieved control of the bleeding in 52 patients (90%), and 1-year and 3-year survival rates were 51.7% and 40.2%, respectively [108]. Balloon tamponade is still used as a bridge therapy and provides hemostasis in 80–90% of patients, but the rebleeding rate after deflation is as high as approximately 50% [109,110]. Moreover, because it is associated with a high rate of serious complications, such as esophageal ulceration, esophageal rupture, and aspiration pneumonia, balloon tamponade should not exceed 24 hours [111]. In a small RCT, a self-expandable, esophageal covered metal stent was tested as an alternative to balloon tamponade in patients in whom pharmacological and endoscopic treatment failed to control bleeding [112]. Although survival in the esophageal stent group was not improved compared with the balloon tamponade group, bleeding control was higher (85% vs. 47%, P=0.037), and serious adverse events were lower (15% vs. 47%, P=0.077) in the esophageal stent group [112]. This stent can be placed endoscopically without radiological guidance, and it can stay in place for up to 2 weeks. However, because only 28 patients were included in that study, further study is warranted.
[Recommendations]
1. Endoscopy should be performed in patients with suspected esophageal variceal bleeding. (A1)
2. Endoscopic treatment should be performed in patients with acute esophageal variceal bleeding. (A1)
3. In patients with acute esophageal variceal bleeding, restrictive PRBC transfusion is recommended with the goal of maintaining a hemoglobin level of 7–9 g/dL. (A1)
4. Short-term antibiotic prophylaxis should be instituted in patients with acute esophageal variceal bleeding. (A1)
5. If esophageal variceal bleeding is suspected, vasoactive agents should be initiated as soon as possible after admission. (A1)
6. Early TIPS placement can be considered in patients at high risk of rebleeding. (B2)
7. A TIPS is a possible rescue treatment for patients in whom bleeding control fails despite combined pharmacological and endoscopic therapy. (A2)
8. Balloon tamponade can be considered as a bridge therapy for patients who fail to achieve hemostasis after endoscopic treatment. (B2)
Prevention of esophageal variceal rebleeding
Definition of esophageal variceal rebleeding
EV rebleeding is defined as recurrent bleeding after an absence of bleeding for at least 5 days following recovery from acute EV bleeding [107]. An average of 60% of patients with acute EV bleeding experience rebleeding within 1–2 years, and the mortality rate from rebleeding is 33%. Therefore, appropriate treatment to prevent rebleeding is necessary [15,48].
Diagnosis of esophageal variceal rebleeding
The diagnosis of EV rebleeding is the same as the diagnosis of acute EV bleeding. Clinically significant rebleeding can be suspected in a patient who has recurrent melena or hematemesis with 1) hospitalization or the need for a transfusion, 2) a decrease in hemoglobin of more than 3 g /dL, or 3) death within 6 weeks [107].
Prevention of esophageal variceal rebleeding
NSBBs and EVL are the most common methods used to prevent EV rebleeding. NSBBs, which reduce portal pressure, have been reported to be more effective than placebo at preventing rebleeding in several RCTs [113-115]. The combination of an NSBBs plus ISMN could improve portal pressure reduction [116], but it could also increase the incidence of side effects such as headache and dizziness [117]. EVL is the endoscopic treatment of choice for the prevention of EV rebleeding. EVL should be repeated every 2–8 weeks until variceal eradication is achieved. Periodic endoscopic follow-up is needed to detect the recurrence of varices even after achievement of variceal eradication. Several systematic reviews and meta-analyses comparing EVL alone to NSBBs alone demonstrated no difference in the rebleeding rate [51,118,119], but the overall mortality rate during follow-up was significant higher with EVL alone (RR, 1.25; 95% CI, 1.01–1.55) [51] or not different [119]. In a long-term follow-up study, the rebleeding rate was higher (30% vs. 64%, P=0.001) but the survival time was longer (30% vs. 49%, P=0.013) in patients treated with the combination of an NSBBs plus ISMN [120].
Several RCTs and meta-analyses comparing the combination of EVL plus NSBBs to EVL alone or NSBBs alone showed that the combination therapy had lower overall rebleeding and variceal rebleeding [121-124]. Therefore, the combination of EVL plus an NSBBs has been suggested as the primary treatment for preventing EV rebleeding. A recent meta-analysis demonstrated that the rebleeding rate decreased (RR, 0.44; 95% CI, 0.28–0.69) and the mortality rate during follow-up tended to decrease with the combination of EVL plus a NSBBs (RR, 0.58; 95% CI, 0.33–1.03) compared with EVL alone. However, although the overall rebleeding rate tended to decrease (RR, 0.76; 95% CI, 0.58–1.00), the mortality rate during follow-up did not differ between the combination of EVL plus NSBBs and NSBBs alone [125]. These results suggest the importance of NSBBs in preventing EV bleeding.
RCTs comparing carvedilol to EVL (36.4% vs. 35.5%, P=0.857) and carvedilol to the combination of nadolol plus ISMN (51% vs. 43%, P=0.46) did not show any significant difference in rebleeding rate, and the side effects of carvedilol were less than those with the combination of nadolol plus ISMN (1.6% vs. 28.3%, P<0.0001) [126,127]. Therefore, the use of carvedilol to prevent EV rebleeding can be considered, but no studies have compared the combination of EVL plus carvedilol with the combination of EVL plus an NSBBs, which is currently considered to be the primary treatment to prevent rebleeding. Further studies using carvedilol to prevent EV rebleeding are required.
In a meta-analysis of studies about preventing variceal rebleeding by using NSBBs to reduce portal pressure, the risk of variceal rebleeding was significantly reduced (OR, 0.17; 95% CI, 0.09–0.33; P=0.0001) when the HVPG was decreased to the target level (reduction in HVPG of ≥20% or to ≤12 mmHg) compared with the non-responding group [128]. A recent RCT comparing HVPGbased medical therapy with TIPS placement to reduce variceal rebleeding showed lower incidence of rebleeding within 2 years (26% vs. 7%, P=0.002) in the TIPS group, but there was no significant difference in mortality during follow-up between the two groups, and the incidence of HE was lower (8% vs. 18%, P=0.05) in the HVPG-based medical therapy group [129]. Considering that a TIPS is a limited treatment method, HVPG-based medical therapy is a useful way to prevent rebleeding if HVPG measurement is possible. However, because HVPG measurement is invasive, it is not widely practiced in many hospitals.
An RCT comparing TIPS placement with a combination of EVL plus an NSBBs to prevent variceal rebleeding found a lower variceal rebleeding rate in the TIPS group (0% vs. 29%, P=0.001), but the incidence of HE within 1 year in that group was higher (35% vs. 14%, P=0.035). There was no difference in the follow-up mortality rate (32% vs. 26%, P=0.418) between the two groups [130]. Therefore, the use of TIPS is not recommended as a primary treatment for the prevention of variceal rebleeding, and it should instead be considered a rescue therapy for patients with primary treatment failure [131]. In addition, liver transplantation is considered a rescue therapy for patients with recurrent variceal rebleeding because it exhibits good long-term results [132,133].
[Recommendations]
1. In patients with acute esophageal variceal bleeding, treatment to prevent variceal rebleeding is recommended. (A1)
2. The combination of endoscopic variceal ligation (EVL) plus NSBBs is recommended as the primary treatment for esophageal variceal bleeding. (A1) If the combination treatment is difficult to perform, use of a NSBBs or EVL alone is recommended. (A1)
3. If primary treatment for esophageal variceal rebleeding fails, TIPS placement should be considered as a rescue therapy. (B1)
4. Liver transplantation might be considered in patients with recurrent variceal rebleeding. (B1)
Definition of gastric varices and prevention of primary bleeding
Definition and classification of gastric varices
Gastric varices are enlarged submucosal veins of the stomach that cause critical upper gastrointestinal bleeding. GVs occur in approximately 20% of patients with portal hypertension, and the bleeding rate in 2 years is known to be 25% [134]. The incidence of gastric varices is lower than that of EVs, but their rebleeding rate and mortality rate are higher because they cause severe bleeding [134-136].
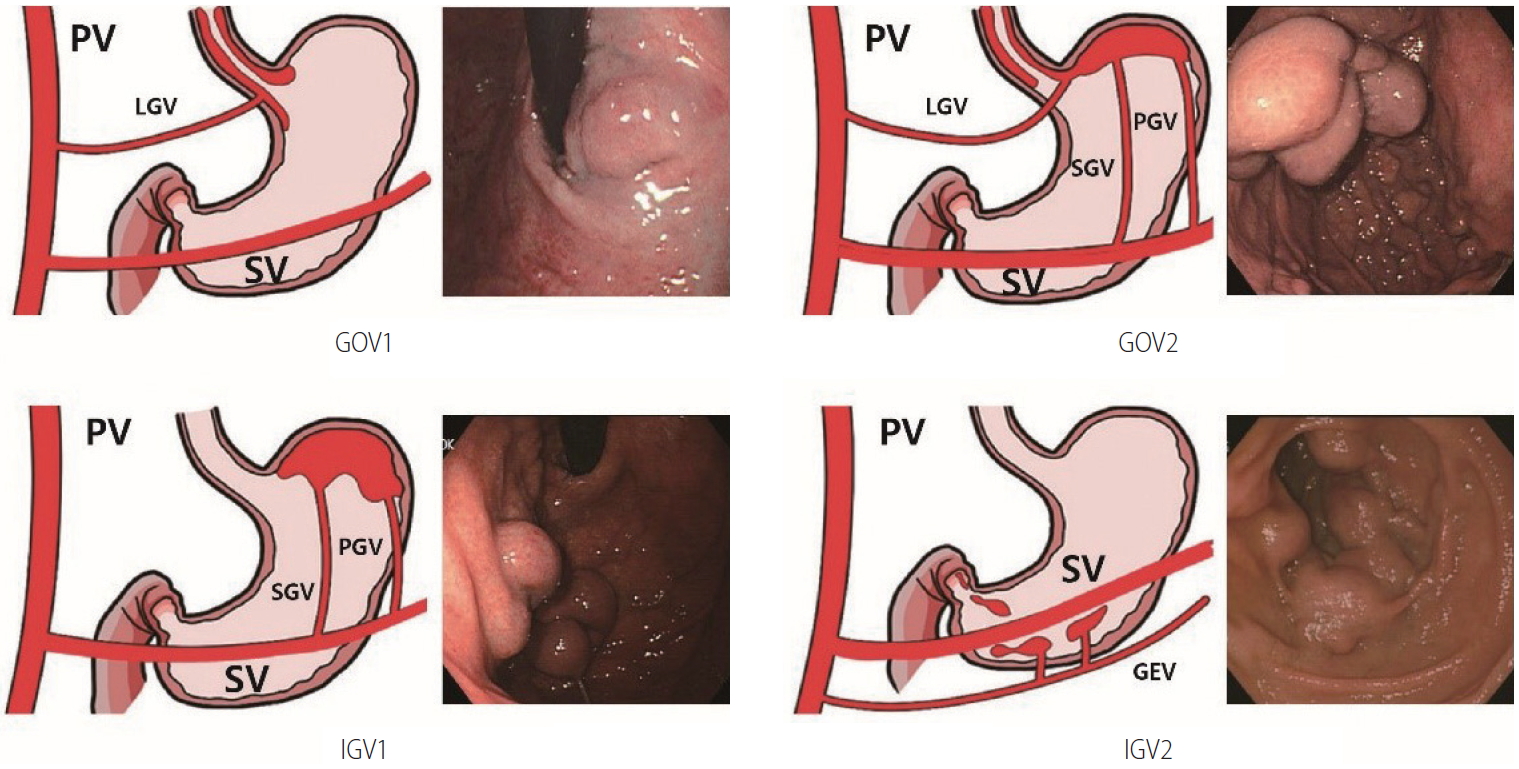
Gastric varices are classified as gastroesophageal varices (GOV) or isolated gastric varices (IGV) depending on their location and relation to any EVs (Fig. 1). GOVs are classified by whether they extend along the lesser curvature (GOV1) or the gastric fundus (GOV2). IGV are classified as varices located in the fundus (IGV1) and those in any other region, i.e., stomach or duodenum (IGV2) [134]. The incidence of GOV1s is about 74%.
Prevention of primary bleeding of gastric varices
The risk factors for gastric variceal bleeding are location (IGV1>GOV2>GOV1), variceal size, redness, and severe liver dysfunction [26,47,93,137-139].
To prevent bleeding from GOV1s, follow the guidelines for the prevention of EV bleeding. In a Korean study of 85 patients with GOV1s, the GOV1s also disappeared when EVs were eliminated by EVL (64.7%) [140]. For GOV2s and IGV1s, EVO, balloon-occluded retrograde transvenous obliteration (BRTO), and vascular plug-assisted retrograde transvenous obliteration (PARTO) can be considered to prevent bleeding [141,142]. NSBBs are non-invasive and can be used because they can reduce other side effects in patients with cirrhosis.
One randomized study reported the prevention of first gastric variceal bleeding. It enrolled 89 patients with GOV2s or IGV1s larger than 10 mm [141]. The effects of EVO (cyanoacrylate), an NSBB, and simple observation were compared. For the prevention of gastric variceal bleeding, EVO (10%) was superior to an NSBB (38%) and simple observation (53%) [141]. The survival rate of the EVO group (93%) was higher than that of the simple observation group (73%), but it did not differ from that of the NSBB group (83%). In a meta-analysis of patients with a high risk of gastric variceal bleeding, BRTO was effective in preventing gastric variceal bleeding (clinical success rate, 97.3%) [142]. In a recent study of 73 patients, PARTO was found to be a safe procedure without serious side effects that effectively prevented gastric variceal bleeding (Fig. 2) [143,144].
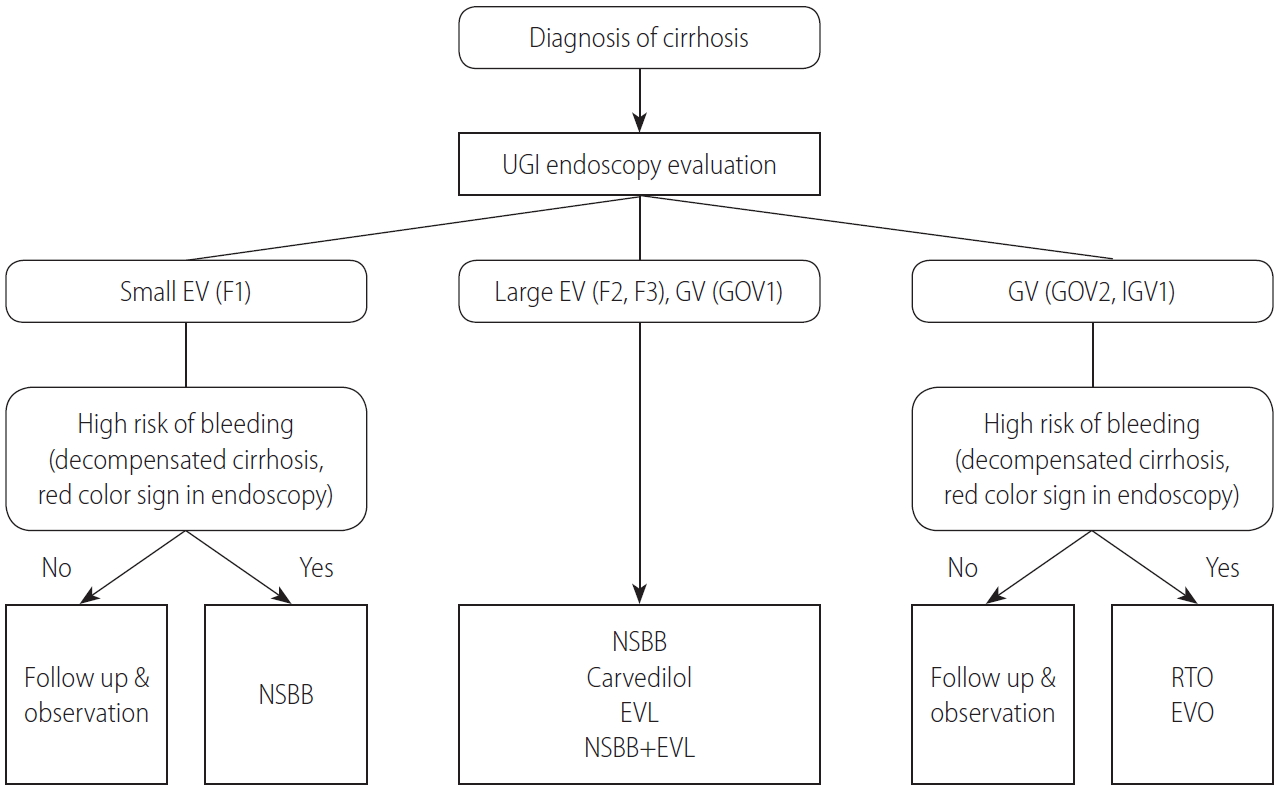
The prevention of initial variceal bleeding. UGI, upper gastrointestinal; EV, esophageal varix; GV, gastric varix; GOV, gastroesophageal varix; IGV, isolated gastric varix; NSBB, non-selective beta blocker; EVL, endoscopic variceal ligation; RTO, retrograde transvenous obliteration; EVO, endoscopic variceal obturation.
[Recommendations]
1. Primary prevention of bleeding for GOV1s follows the recommendations for EVs. (B1)
2. The group at high risk for bleeding (redness or severe liver dysfunction) from GOV2s or IGV1s can be treated with BRTO, PARTO, or EVO. (B2)
Management of bleeding from gastric varices
Bleeding from gastric varices is less common than from EVs; however, the risks of rebleeding or varix-related death are much higher in patients bleeding from gastric varices. The gastric varices that bleed are generally large and have high blood flow in the channel, which makes massive bleeding common in patients with large gastric varices [134,145,146]. Gastric varices exhibit unique characteristics and have a greater variety of sizes, forms, locations, and collateral vessels than EVs. An individualized approach might be needed because few well-controlled clinical trials have tested treatments for gastric variceal bleeding. Until sufficient evidence accumulates, clinicians should seek the best option for each patient based on the patient’s general condition and bleeding patterns and the clinician’s medical resources and expertise [145].
Management of bleeding from gastric varices
Endoscopic therapy
Urgent endoscopic examination, within 12 to 24 hours, is necessary when a patient is suspected to have active bleeding from gastric varices. Endoscopic examination can visualize the bleeding sites and directly enable proper hemostatic treatments [47,147].
EVO
EVO achieves hemostasis and induces variceal eradication by an intravariceal injection of tissue adhesive agents (cyanoacrylates). Active or recent bleeding from fundic varices (GOV2s, IGV1s) or GOV1s can be managed with EVO. Special care is needed to prevent complications from the adhesive agents, such as ocular injury, damage to endoscopic devices, or the impaction of an injection needle into a varix [148]. Medical personnel are advised to wear goggles during the procedure. The working channel of a scope can be occluded by adhesive agent that spills during the procedure, so it can be helpful to flush the channel with olive oil in advance. To inject the sticky mixture quickly, a large needle is generally used (21 G or 22 G). The injection site is determined based on the direction of blood flow inside the varix. Because the intravariceal pressure is usually concentrated in the most protruding part of the varix, avoid that site if possible. The injection needle should be long enough to pass through the thick gastric wall (5 mm or longer). 2-N-butyl cyanoacrylate, which is the most commonly used agent in Korea, is used as a 1:1 mixture with lipiodol to delay the polymerization reaction. About 1 mL of mixture is used in each session, and the injection can be repeated until hemostasis is achieved. The initial volume and ratio of the mixture can be adjusted to accommodate the variceal size, intravariceal blood flow, and bleeding pattern (active or stabilized). If the bleeding is severe or the variceal size is large, the volume of the mixture can be increased to 2 mL at a time. As soon as the injection is finished, 1 mL of distilled water or saline should be pushed into the catheter to ensure that the mixture remaining in the catheter is injected into the varix. Then, the needle should be retracted quickly to prevent intravariceal impaction of the needle. The success rate of EVO for hemostasis was 91–97%, and the rebleeding rate was 17–49% in patients with active gastric variceal bleeding [149-153]. The common complications following EVO are systemic embolism, infection, fever, gastric perforation, gastric ulcer, and peritonitis [154].
EVL
As with EVs, EVL is frequently performed for GOV1 bleeding. EVL for gastric varices showed an initial hemostasis rate of 80–90% and a rebleeding rate of 14–56% in patients with GOV1s [140,155-158]. However, it should be noted that the depth and size of gastric varices differ from those of EVs. Ligation might not be adequate due to the thick gastric mucosa. Gastric ulcers, where the bands fall off, will expose submucosal varices directly to gastric acid and food materials. This situation could increase the risk of massive bleeding from the ulcers [154,155,158,159]. In patients with fundal variceal bleeding, the effect or safety of EVL has not been fully explored. In a small randomized trial, EVL showed a significantly higher rebleeding rate than EVO in patients with IGV1 bleeding (83.3% vs. 7.7%, P=0.003) [155].
Radiologic intervention
Radiologic intervention is one useful hemostatic therapy for the management of bleeding from gastric varices. Sufficient consultation with interventional radiologists is needed in advance. Imaging tests, such as computed tomography (CT), should be performed before the procedure to confirm that the collateral veins are accessible and that no contraindications to the procedure are present.
TIPS
TIPS placement is a procedure that robustly decompresses portal hypertension by making a bypass between the hepatic vein and the portal vein. In small non-randomized trials, both TIPS and EVO achieved a hemostasis rate of more than 90%. Complications, such as HE and stent occlusion, and medical costs were higher with the TIPS than with EVO [160,161]. However, TIPS placement is a useful rescue therapy when initial hemostasis fails [162-164]. The success rate of TIPS in controlling bleeding as a rescue therapy is 90–100%, with a rebleeding rate of 16–40% [162-166]. Moreover, since non-covered stents have been replaced by covered stents, the occlusion and stenosis rates have decreased to 8% [167,168]. HE can be prevented by decreasing the stent diameter. In a randomized study, the incidence rates of HE within 2 years were 43% and 27% in patients with a conventional stent (10 mm) and those with a smaller one (8 mm), respectively (P=0.03) [168]. TIPS is contraindicated in patients with heart failure or severe pulmonary hypertension because it can abruptly increase preload to the heart. It is difficult to perform the procedure in patients with main portal vein thrombosis. When a cyst, abscess, or mass is blocking the accessible tract in the liver or the intrahepatic bile ducts are markedly dilated, it is difficult to perform TIPS [169].
RTO
RTO obliterates gastric varices by infusing a sclerosant or embolic agent in a retrograde manner through a gastrorenal shunt. An accessible shunt should be confirmed by CT prior to the procedure. After occluding the shunt with a balloon catheter, a sclerosant, such as ethanolamine oleate or sodium tetradecyl sulfate, is infused into the gastric varices [170,171]. In a recent, large, retrospective study, the technical success rate of BRTO was 95% [172]. Another multicenter study, in which 23% of patients had GOV1s, had a technical success rate of 97% [173]. However, the EVs recurred or became aggravated in 20–41% of patients after the procedure [172,173]. A recent meta-analysis also showed favorable results. The technical success and major complication rates of BRTO were 96.4% and 2.6%, respectively. The clinical success rate, defined as no recurrence of gastric varices or complete obliteration of varices on subsequent imaging, was 97.3% [142].
If a shunt is too large for balloon catheter occlusion, BRTO is not possible. Moreover, BRTO requires that patients retain the balloon catheter for several hours, until the sclerosing agent has hardened in the varices. In rare cases, the balloon can rupture during the procedure, and a systemic embolism of the sclerosing agent can occur. Therefore, a novel intervention, PARTO, was recently developed. PARTO uses a vascular plug with or without coils instead of a balloon and uses a gelatin sponge as the embolic agent [143]. A multicenter prospective study showed that complete thrombosis of gastric varices and shunts was achieved in 98.6% of patients. No recurrent variceal bleeding or development of HE occurred during follow-up. Moreover, 40% of patients showed improvement in their Child-Pugh scores [144]. Thus, PARTO is a noteworthy treatment that can replace BRTO in patients with gastric varices and a gastrorenal shunt. However, more data on the long-term efficacy and safety of PARTO are needed.
Treatment of gastric variceal bleeding
General management of gastric variceal bleeding
In patients with cirrhosis and acute upper gastrointestinal bleeding, a restrictive blood transfusion strategy (with a target range for the post-transfusion hemoglobin level of 7 to 9 g/dL) and antibiotic prophylaxis improved survival [83,174]. Although patients enrolled in the studies were small, the same transfusion strategy can be recommended for those with gastric variceal bleeding. The beneficial effects of vasoactive agents (terlipressin, octreotide, somatostatin) have not been fully proved in patients with gastric variceal bleeding, either. However, considering their ability to decrease portal hypertension, their use in patients with bleeding from gastric varices can be recommended [91,158,175,176].
Treatment of GOV1 bleeding
GOV1s, which are an extended type of EV, develop along the lesser curvature and receive blood from the left gastric vein. When EVs are eradicated by endoscopic treatments, the gastric varices also concomitantly disappear in 60–65% of patients [134,140]. Because of their close relationship in pathophysiology, the management of bleeding from cardiac varices (GOV1s) is similar to that for EV bleeding [177]. However, it should be noted that sufficient ligation can be difficult for gastric varices because of their large size and deeper location. Furthermore, subsequent post-ligation ulcers might be exposed to gastric acid or food material [154,155,158,159]. According to small clinical trials and observational studies, EVO produces more favorable outcomes than EVL. The initial hemostasis rates with EVO and EVL in patients with GOV1 bleeding were 85–100% and 80–90%, respectively. The rebleeding rates following EVO and EVL were 3–26% and 14–56%, respectively [140,155-158]. However, most of those trials were small; the evidence needed to recommend one of these treatments over the other remains insufficient [140,155,157,158,178]. Therefore, clinicians may choose either EVO or EVL based on their expertise, available medical resources, and the variceal condition (size or extent).
Treatment of GOV2 or IGV1 bleeding
GOV2s are a type of gastric varix that extends from EVs toward the fundus. IGV1s are varices localized in the fundus in the absence of EVs [134]. Both GOV2s and IGV1s are usually called gastric fundic varices. Unlike EVs, fundic varices are supplied with blood from the posterior gastric vein or short gastric vein [179,180]. Bleeding from the fundus usually occurs in a stage of large varix. Management of fundic variceal bleeding can be difficult because massive or recurrent bleeding is frequently accompanied. Moreover, collateral shunts or blood circulation around the fundic varices are very diverse. Therefore, it is difficult to apply simple or uniform treatments for fundic variceal bleeding [181]. Urgent endoscopic examination is always needed in patients with suspicious fundic variceal bleeding in order to direct visualization of bleeding sites and to apply immediate treatments. EVO is one of the most commonly performed in patients with bleeding from fundic varices [182] EVO achieved initial hemostasis more often than EVL (OR, 4.44; 95% CI, 1.14–17.3). In particular, the rebleeding rate following EVO was significantly lower than that following EVL in patients with IGV1s (OR, 0.06; 95% CI, 0.01–0.58) [183]. TIPS placement and EVO are both effective treatments to control bleeding, with a hemostasis rate of more than 90%. Because of complications such as HE, stent occlusion, and higher cost, TIPS placement over EVO is not recommended as a first-line treatment [160,161]. However, TIPS placement is an effective rescue therapy when endoscopic therapy fails. The hemostasis rate of TIPS in a rescue setting is 90–100% [162-166]. BRTO also achieved a high hemostasis rate (more than 90%) [184-186]. However, BRTO showed a significantly lower rebleeding risk (OR, 0.27; 95% CI, 0.09–0.81) and a lower risk of HE (OR, 0.05; 95% CI, 0.02–0.13) than TIPS [186]. Improvement in liver function was also demonstrated following BRTO [187]. However, all those results are based on mostly small retrospective studies.
In a small prospective study, BRTO and EVO had similar hemostasis and technical success rates. However, the rebleeding rate was significantly lower in the BRTO group than the EVO group (15.4% vs. 71.4%, P<0.01) [188]. These results should be interpreted carefully, however, because BRTO was performed only in patients without active bleeding; all the patients with active bleeding were treated with EVO.
In summary, current data suggest that EVO, TIPS, BRTO, or (theoretically) PARTO can be used as the initial treatment for patients bleeding from fundic varices. Because of a lack of evidence, treatments should be chosen based on individual situations in consideration of patients’ safety and the applicability of each therapy in the relevant medical facility.
Use of PPIs
Currently, PPIs are used in many patients to prevent ulcer bleeding following endoscopic treatments. However, their effectiveness and duration of treatment have not been fully explored. Long-term use of PPI can increase risk of infection and subsequently cause spontaneous bacterial peritonitis and HE [79]. However, a recent retrospective study showed that PPI use decreased the rebleeding risk following EVO (OR, 0.554; 95% CI, 0.352–0.873) [189].
Rescue therapy in case of endoscopic failure
A TIPS can be urgently placed when endoscopic treatments fail. The hemostasis rate with rescue TIPS was 90–96% in patients with gastric varices, which is comparable to that with EV bleeding [162,163]. In a few small studies, BRTO also showed comparable outcomes in patients who failed to achieve initial hemostasis. BRTO can be considered as a rescue therapy when a patient was hemodynamically stabilized and has an accompanying gastrorenal shunt [184,186]. As a bridging therapy, a balloon tamponade can be applied to control massive bleeding until rescue therapy is ready [109].
[Recommendations]
1. In p atient s with gas tric variceal b le e ding, gener al management, such as prophylactic antibiotics, restrictive transfusion, and vasoactive agents, can be provided as they are for esophageal variceal bleeding. (B1)
2. Gastric varices extending from EVs along the lesser curvature (GOV1s) can be treated with either EVO or EVL, depending on the size and location of the bleeding varix. (B1)
3. In patients with bleeding from fundic varices (GOV2s, IGV1s), EVO should be considered first. (A1) Retrograde transvenous obliteration (BRTO or PARTO) or TIPS can be used depending on the bleeding status (active or stabilized) and the presence of an accessible shunt. (B1)
4. A PPI can be used following endoscopic treatments to prevent post-procedure ulcer bleeding. (B2)
5. Retrograde transvenous obliteration (BRTO or PARTO) or TIPS should be considered as a rescue therapy when endoscopic treatments fail. (B1)
6. Until a rescue therapy is ready, a balloon tamponade can be applied as a bridging therapy. (B2)
Prevention of rebleeding
GOV1s can be managed in the same way as EVs to prevent rebleeding. The eradication of concurrent EVs with EVL and an NSBB can be used if the EVs are medium to large in diameter. Gastric varices subsequently disappeared in 65% of patients when EVs were controlled [140]. The rebleeding rate from GOV1s following eradication of EVs was 16–42% [155,156]. Esophageal EVL can be performed simultaneously with or after treatments for gastric varices. In terms of gastric varices, EVO showed a significantly lower rebleeding rate than EVL in patients bleeding from GOV1s (OR, 0.39; 95% CI, 0.16–0.94) [155,158,183]. However, those studies included only a small number of patients. In a retrospective Korean study, EVO showed beneficial outcomes, with lower 1-year rebleeding rate (3.6% vs. 30.8%, P=0.004) and bleeding-related mortality rate (5% vs. 22%, P=0.05) than EVL [140]. In a different small study, TIPS placement showed a significantly lower rebleeding rate than EVO (21% vs. 65%, P<0.02) [190]. However, it is difficult to draw conclusions from that study alone because its rebleeding rate following EVO was relatively higher than previous reports. If an accessible gastrorenal shunt is identified, BRTO or PARTO might be considered. Unfortunately, evidence to support those interventions in patients with GOV1 bleeding is very limited [173,191].
In patients bleeding from fundic varices (GOV2s or IGV1s), the only predictor for rebleeding following EVO was variceal size (F3). The use of NSBBs failed to decrease the rebleeding rate [192]. In an RCT, eradication of gastric varices with repeated EVO lowered the rebleeding rate significantly compared with NSBBs (10% vs. 44%, P=0.004) [193]. There were no differences in rebleeding (54% vs. 47%, P=0.609) or bleeding-related mortality (42% vs. 47%, P=0.766) between EVO alone and EVO plus an NSBB, respectively [194]. Therefore, use of an NSBB is not recommended to prevent recurrent bleeding from fundic varices. However, NSBBs should be considered if patients have significant portal hypertension or other proven indications, such as large EVs [47]. Clinical trials comparing the rebleeding rates after repeated EVO and TIPS or BRTO are scarce. In a small randomized study of patients with GOV2 bleeding, there was no significant difference in the rebleeding rate between EVO repeated every 4 weeks and TIPS placement (16% vs. 0%, P>0.05) [190]. However, TIPS placement was associated with a higher incidence of complications than EVO [161]. In a meta-analysis, BRTO (7.4%) showed a much lower rebleeding rate than TIPS (22.8%) (OR, 0.27; 95% CI, 0.09–0.81) [186]. For GOV2s, treatment of the accompanying EVs can be performed with or after the treatment of fundic varices, according to the guidelines for treating EVs (Fig. 3).
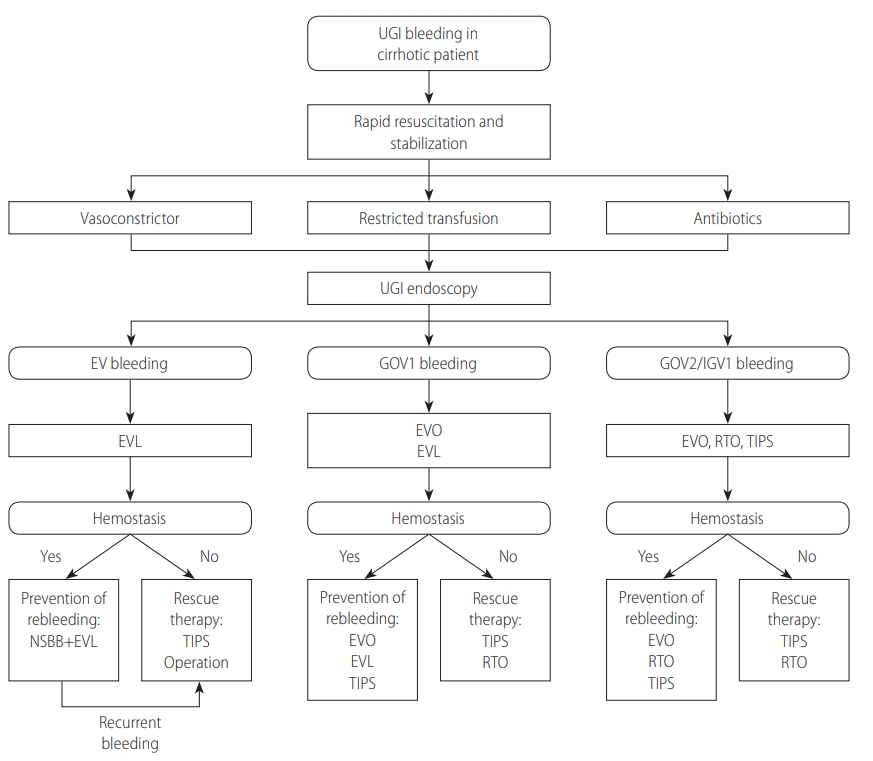
The treatment of acute variceal bleeding and prevention of variceal rebleeding. UGI, upper gastrointestinal; EV, esophageal varix; GOV, gastroesophageal varix; IGV, isolated gastric varix; EVL, endoscopic variceal ligation; EVO, endoscopic variceal obturation; RTO, retrograde transvenous obliteration; TIPS, transjugular intrahepatic portosystemic shunt; NSBB, non-selective beta blocker.
[Recommendations]
1. In patients with remnant or recurrent GOV1s following initial treatments, repeated EVO or EVL can be performed to prevent rebleeding. (B2)
2. In patients with remnant or recurrent fundic varices (GOV2s, IGV1s), EVO or RTO (BRTO or PARTO) can be performed. (B2) If there is no accessible shunt or if complications related to severe portal hypertension (recurrent bleeding from EVs, refractory ascites, or hydrothorax) are not controlled, a TIPS can be placed. (B2)
Other variceal bleeding
In cirrhosis, variceal bleeding at sites other than the stomach and esophagus is very rare, and there are no established treatment guidelines. The most common locations are the rectum, duodenum, and postoperative stomach. A multi-disciplinary approach involving an endoscopist, interventional radiologists, and surgeons should be used to account for the vascular supply. EVO, BRTO, PARTO, TIPS, the coil inserting method, and the like can all be used [195].
Portal hypertensive gastropathy
Definition and diagnosis
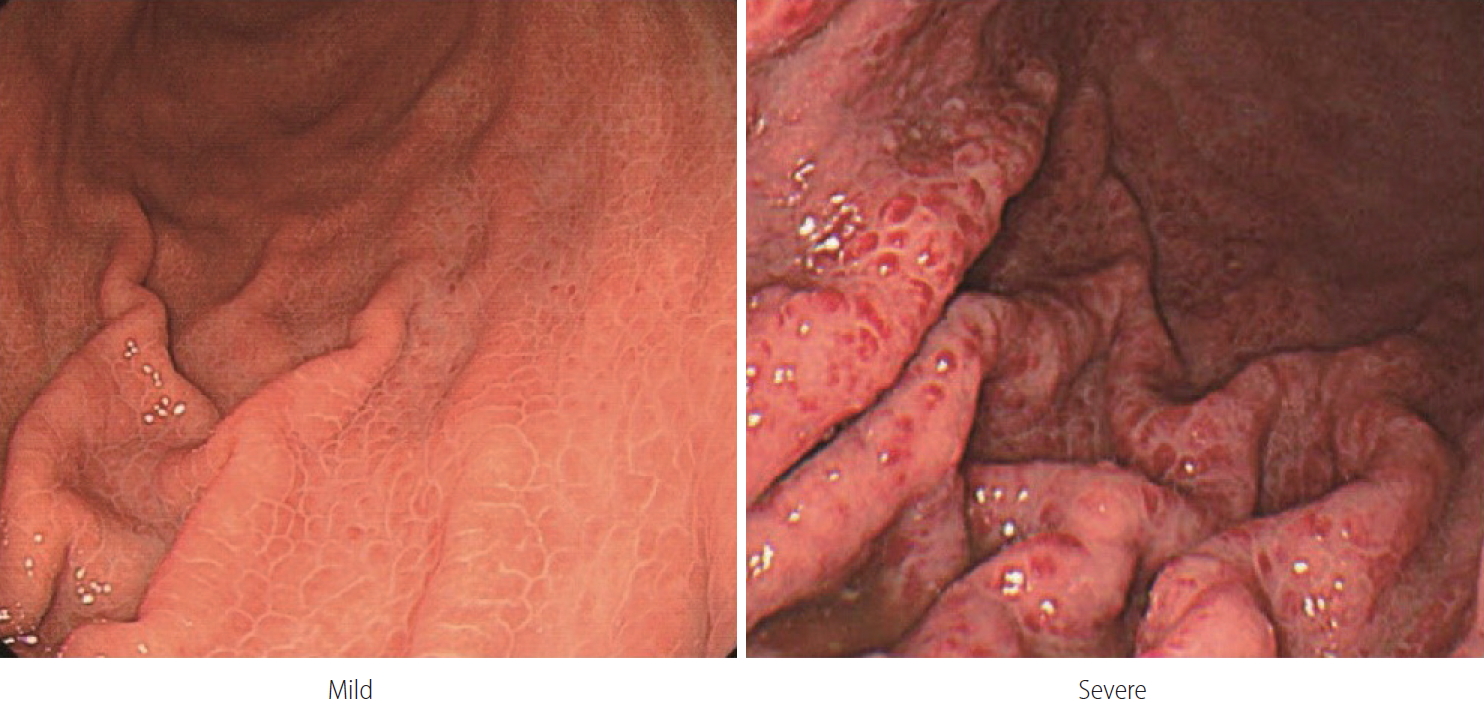
Although the incidence of portal hypertensive gastropathy bleeding in cirrhosis is not high, some patients experience poor quality of life due to chronic bleeding and the associated iron-deficiency anemia and repeated transfusions [196,197]. Portal hypertensive gastropathy is diagnosed when gastric mucosal changes cause a snake-skin appearance or mosaic pattern on endoscopy in patients with portal hypertension [198-200]. When gastric mucosal changes alone are found, it is diagnosed as a mild form. When red or dark brown viscous changes are found along with changes in the gastric mucosa, it is considered to be severe (Fig. 4) [30]. Severe portal hypertensive gastropathy causes more chronic bleeding than the mild form [201].
Portal hypertensive gastropathy is associated with portal hypertension and causes gastric mucosal changes in the stomach and body, and 30% of patients with gastric antral vascular ectasia (watermelon stomach) also have portal hypertension. It is unclear whether portal hypertension is involved in the development of gastric antral vascular ectasia. Gastric antral vascular ectasia causes dilated vessels with fibrin thrombi and fibromuscular hyperplasia of the lamina propria [202].
Treatment of portal hypertensive gastropathy
In chronic bleeding caused by portal hypertensive gastropathy, the goal of treatment is lowering the portal pressure with NSBBs, vasoconstrictors, or a TIPS [203,204]. In cases with active bleeding, endoscopic treatment with argon plasma coagulation can be used. In addition, iron supplementation is recommended [205].
[Recommendations]
1. If chronic bleeding is caused by portal hypertensive gastropathy, nonselective beta-blockers can be used. (B1)
HEPATIC ENCEPHALOPATHY
HE occurs in more than 10% of all cases of cirrhosis and is a critical complication that seriously reduces the quality of life [206]. Because HE can cause serious losses not just for individuals, but also socioeconomically, preventive therapy is of paramount importance. However, because the pathophysiological factors in the development of HE and biomarkers to predict the occurrence of HE have not been sufficiently identified, there are no standardized criteria for diagnosing, classifying, or evaluating the treatment response to HE. It is imperative that those criteria be established in Korea. In particular, quality-of-life assessments and diet and exercise education for patients with HE are clinically important and need to be actively developed.
Definition of HE
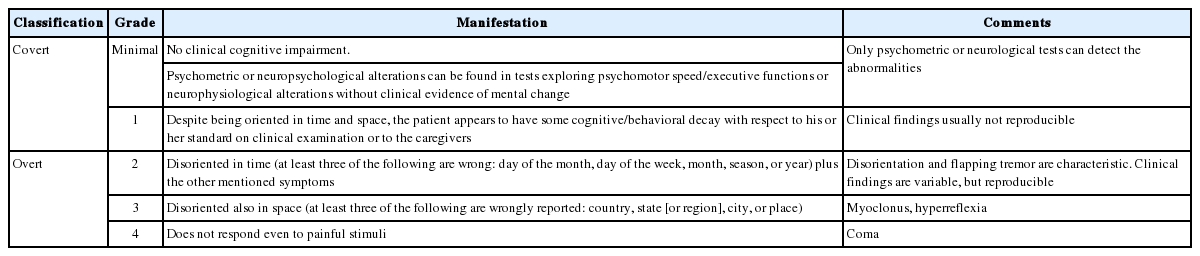
HE is a neuropsychiatric syndrome caused by hepatic dysfunction that manifests as various neurologic and psychiatric abnormalities [207-209]. Clinically, it is classified into overt and covert encephalopathy. Overt HE (OHE) is defined as the occurrence of disorientation, flapping tremor, or asterixis (Table 3). Covert HE (CHE) includes minimal encephalopathy in which cognitive impairment cannot be identified without a cognitive function test and West-Haven criteria grade 1 HE, which means mild cognitive or behavioral change without disorientation [210]. The prevalence of HE is reported to be 10–14% of cirrhotic patients and 16–21% of patients with decompensated liver cirrhosis [206,211]. In Korea, HE was found in 16–21% of hepatitis B virus–related decompensated liver cirrhosis patients [212]. Moreover, 20% of cirrhotic patients admitted to the emergency department were reported to have HE [213].
HE is classified according to the underlying liver disease, clinical course, precipitating factors, and severity of neurologic symptoms [209]. By underlying liver disease, HE is subdivided into three groups: from acute liver failure, from portosystemic bypass or shunting, and from portal hypertension caused by chronic liver disease. HE caused by portal hypertension is classified as episodic, recurrent (more than two times per year), and persistent HE (no fully recovery from behavioral change). When classified by the precipitating factors, HE is divided into precipitated and spontaneous types. Precipitating factors include gastrointestinal bleeding, uremia, sedatives, diuretics, protein overload, infection, constipation, dehydration, and electrolyte imbalance. The severity of HE is classified using the West-Haven criteria (Table 3).
Diagnosis of HE
Clinical symptoms
HE presents with a wide range of clinical patterns, from minimal HE (MHE), in which cognitive impairment cannot be identified without a cognitive function test, to OHE, which is easily detected based solely on symptoms and does not require a cognitive function test. As HE progresses, symptoms such as personality changes, indifference, anxiety, and irritability appear and can reduce sleep quality and quality of life [214]. In some patients, increased muscle tension, hyperreactivity, and the Babinski reflex are present, and they are rarely accompanied by seizures [215,216]. The flapping tremor, a phenomenon in which hand tremors are caused by incongruity in the tension of various muscles resulting from hyperextension of the wrist as the fingers are spread apart, is a common symptom in the early and middle phases of OHE.
Severity classification
The severity of HE is classified using the West-Haven criteria and the Glasgow Coma Scale [207], with the former used as the basic diagnostic criteria. However, due to their large number of subjective factors, the West-Haven criteria suffer from significant interobserver deviation, which makes it difficult to diagnose the first stage (grade 1) of HE in a clinical setting. Therefore, MHE and stage 1 HE are classified as CHE (Table 3) [217]. The International Society for Hepatic Encephalopathy and Nitrogen Metabolism (ISHEN) defines the onset of disorientation or flapping tremor as the start of OHE [218].
Differential diagnosis
HE requires differentiation from underlying brain diseases, such as cerebral hemorrhage and edema, that can accompany cognitive dysfunction. It should also be differentiated from substance abuse, alcoholism, hyponatremia, and psychiatric illnesses. In chronic alcoholics in particular, it can be difficult to differentiate HE from other alcohol-related neurological diseases. For example, Korsakoff syndrome, which is caused by a thiamine deficiency induced by long-term drinking, is characterized by symptoms such as anterograde amnesia and decreased word memory [219], and Wernicke’s encephalopathy is marked by eye movement paralysis, gaze-induced nystagmus, and gait disturbances, in addition to memory lapses [220]. Delirium caused by withdrawal from alcohol also needs to be differentiated from HE. Delirium that results from alcohol withdrawal is characterized by an increased heart rate, cold sweats, loud shouting, and a harsh and repetitive tremor [221]. A differential diagnosis is required for acute hyponatremia, hypoglycemia, and metabolic alkalosis because each can present with symptoms similar to those of HE [222]. The differential diagnosis for hyponatremia requires particular caution because its symptoms are very similar to those of HE, and hyponatremia itself can lead to HE [223]. Subdural hematoma can also present with symptoms similar to those of HE and should be carefully differentiated. Cases of subdural hematoma are commonly accompanied by other neurological symptoms, such as hemiplegia. Encephalitis often presents with symptoms such as headache, fever, vomiting, and stiff neck, but a differential diagnosis is required because those symptoms are not always clear and can be accompanied by sleepiness, drowsiness, and unconsciousness. In cases of dementia, the symptoms appear relatively gradually in most cases, whereas alcohol-related dementia often includes violent tendencies caused by frontal lobe damage, as well as the inability to remember recent events [224].
Diagnostic tests
OHE can be diagnosed based solely on clinical symptoms, but other diseases that can cause cognitive dysfunction should still be ruled out. Brain CT and brain magnetic resonance imaging (MRI) are helpful for differentiating neuropsychological abnormalities caused by underlying brain diseases, such as intracranial hemorrhage [225]. Because the risk of cerebral hemorrhage is about five times higher in patients with liver cirrhosis than in healthy people, brain CT or MRI should be performed if a brain lesion is suspected [226]. Brain MRI, in particular, is helpful for diagnosing HE, in which brain edema is associated with nonspecific symptoms such as headache and vomiting, when acute liver failure is suspected [225]. On T1-weighted MRI, an increased signal in the basal ganglia is commonly observed, but those changes lack the sensitivity and specificity required to diagnose HE [227].
If the diagnosis of HE is difficult, neurophysiological or neuropsychological tests can also be performed. In HE, a characteristic, slow triphasic wave is observed during electroencephalography (EEG) [228]. This slow triphasic wave is an overall periodic waveform in the bilateral frontal lobes that demonstrates bilateral synchronization and is often accompanied by slow background activity; it is usually seen in phase 2 or 3 HE and disappears in comatose patients [225,229]. Once a slow triphasic wave has developed, the clinical outcome is reportedly very poor [230]. In recent studies, the decrease in EEG amplitude in patients with OHE was associated with the severity of HE [231].
The brainstem auditory-evoked-potential test is sensitive for the diagnosis of CHE [226,227,232]. Patients with liver cirrhosis accompanied by CHE exhibit conduction time delays (I–V latency) from the auditory nerve to the midbrain and conduction time delays (III–V latency) from the pontine to the midbrain on the brain auditory-evoked-potential test. It is also known that the risk of developing OHE is increased when abnormal findings are observed on the brain auditory-evoked-potentials test [227]. However, in a study using the cortical auditory-evoked-potential test, the N200 latency was increased in patients with HE [233]. Therefore, the diagnosis of HE cannot be made using EEG alone; further research is required to determine the usefulness of evoked-potential EEG in the diagnosis and prognosis of HE.
Serum ammonia
The venous blood ammonia level is not proportional to the degree of HE and has no association with its prognosis [234]. The metabolism of ammonia is greatly influenced by various organs, such as the kidneys, muscles, brain, and bowel, as well as the liver [235]. However, repeated measurements of ammonia concentrations can help to determine a treatment’s effects [235,236]. If patients with suspected OHE have normal ammonia concentrations, attention should be paid to the differential diagnosis to look for other diseases [234]. There are various methods of measuring ammonia concentrations, such as those involving the venous or arterial blood or plasma. Because the normal range varies depending on the specific measurement method, a suitable reference value should be used. Although the partial pressure of ammonia gas in arterial blood is thought to be closely related to both the neurophysiological test results and the ammonia concentration in the blood–brain barrier in patients with HE, additional studies are needed to determine the clinical usefulness of that value [237]. Regarding other serum markers, some studies have reported increases in the serum S100β concentration that were proportional to the cognitive function test results in HE patients [238].
[Recommendations]
1. To confirm the diagnosis of OHE, other diseases that can cause cognitive impairment must first be ruled out, and the diagnosis must be made based on clinical symptoms. (A1)
2. HE is classified as either OHE, which can be diagnosed using only symptoms, or CHE, which requires a cognitive function test. (B1)
3. In patients with suspected HE, imaging tests, including a brain MRI or a neurophysiological test, can be performed to rule out other diseases that can cause cognitive impairment .(B2)
4. Venous blood ammonia levels are not proportional to the degree of HE and are not associated with its prognosis (A1). However, if patients with suspected HE show normal ammonia concentrations, differentiation from other diseases is required. (B1)
Management of overt HE
The goals of treatment
The goals of treatment are as follows: 1) prevention of secondary damage caused by decreased consciousness and normalization of the patient’s state of consciousness, 2) elimination of social and economic restrictions by preventing recurrence, and 3) improvement of patient prognosis and quality of life. Therefore, appropriate supportive care should be provided to prevent secondary damage (e.g., fall-related injuries or aspiration pneumonia) from an altered consciousness. Furthermore, the precipitating factors should be identified and managed appropriately as soon as possible, and treatments should be initiated using medications that can decrease or eliminate the production of ammonia, the major pathogenic material.
Identification of precipitating factors and management
The precipitating factor can be identified in 80–90% of patients with HE [239]. In many cases, HE can be improved simply by eliminating the precipitating factor; therefore, identifying and promptly managing the precipitating factors is required [240]. The currently known precipitating factors of HE and the corresponding diagnostic tests and treatments are shown in Table 4. According to reports from patients in the Republic of Korea [241,242], gastrointestinal bleeding, infection, dehydration by paracentesis, and constipation were the major precipitating factors.
Management of overt HE
Non-absorbable disaccharides
The primary treatment for HE is nonabsorbable disaccharides such as lactulose (β-galactosido-fructose) or lactitol (β-galactoside sorbitol), which lead to recovery in 70–90% of HE patients [217]. Therapeutic mechanisms involve the reduction of intestinal pH by the production of acetic and lactic acids (via bacterial degradation of lactulose). Another potential mechanism is the ability of the nonabsorbable disaccharides to increase the count of lactobacillus, which do not produce ammonia. Furthermore, nonabsorbable disaccharides convert ammonia to ammonium, rendering it less absorbable, and they also produce an osmotic laxative effect that flushes the ammonia out [93,217]. Based on many clinical studies and their low cost, nonabsorbable disaccharides are recommended as an initial therapeutic opition [209,240]. Uribe et al. [243] found that a 20% lactitol enema had higher efficacy in improving symptoms than a tap water enema (100% vs. 20%, P=0.0037) and that the overall response rate to nonabsorbable disaccharides–based therapy was 82.5%. According to a systematic review and meta-analysis [244], lactulose or lactitol was more effective in improving symptoms than placebo, with a RR of 0.62 (95% CI, 0.46–0.84), This finding was reproduced in another recent study [245], which found an RR of 0.63 (95% CI, 0.53–0.74). When overt HE occurs, 30–45 mL of lactulose (20–30 g) every 1–2 hours should be administered orally until the patient is having at least 2 bowel movements a day. An equivalent daily dose of lactitol is 67–100 g [246]. Thereafter, the dose should be titrated to achieve two to three soft stools per day. If patients are unable to take medications orally, administration via nasogastric tube might be tried. If patients have severe HE (West-Haven criteria of grade 3 or more) or are unable to take medications orally or via nasogastric tube, an enema of 300 mL lactulose and 700 mL water can be performed 3–4 times per day until clinical improvement is noted [240,243,247,248]. In this situation, the enema solution should be retained in the intestine for at least 30 minutes [93].
Non-absorbable antibiotics
Rifaximin, a rifamycin derivative, maintains high concentration levels in the intestine because it is not absorbed, and it remains in an active form until it is excreted.
It inhibits bacterial RNA synthesis by binding to bacterial DNA-dependent RNA polymerase, and it has broad antimicrobial activity against aerobic and anaerobic gram-positive and gram-negative bacteria [93]. So far, several studies have shown that rifaximin has a positive effect in managing HE [242,249-251]. Several RCTs with small sample sizes have assessed the effect of rifaximin as a first-line regimen for OHE. A meta-analysis of those RCTs found that rifaximin had a therapeutic effect similar to that of lactulose or lactitol [249,251-254]. Furthermore, in a recent RCT, patients treated with a combination of rifaximin and lactulose showed a better recovery from HE within 10 days (76% vs. 44%, P=0.004) and shorter hospital stays (5.8 vs. 8.2 days, P=0.001) than those treated with lactulose alone [255]. The maximum dose is 1,200 mg/day, which might limit its use in cases of severe HE (West-Haven criteria of grade 3 or more) because of the need for oral administration [93].
Neomycin and metronidazole are also poorly absorbed by the intestine, affect urea-producing bacteria, and reduce the generation of ammonia, which improves HE [93]. However, they are not recommended for the management of HE because of their side effects, such as intestinal malabsorption, nephrotoxicity, and ototoxicity for neomycin and peripheral neuropathy for metronidazole [209,240,256].
LOLA
Because ornithine and aspartate are important substrates used to metabolize ammonia to urea and glutamine, the administration of LOLA can lower plasma ammonia concentrations, with produces improvements in HE [93,257]. For patients with West-Haven criteria grade 1–2 HE, intravenous LOLA can lower the number connection test (NCT)-A time and plasma ammonia concentrations more effectively than placebo [258]. According to a recent RCT, patients treated with the combination of lactulose and intravenous LOLA (30 g/day) had a lower grade of HE within 1–4 days of treatment, with an OR of 2.06–3.04 and a shorter duration until symptom recovery (1.92 vs. 2.50 days, P=0.002), compared with those who received lactulose alone [259]. Oral LOLA can lower the NCT-A time and plasma ammonia concentrations; [260,261] however, further studies are required to assess its efficacy in managing OHE [257].
Branched-chain amino acids (BCAAs)
Among cirrhotic patients, the capacity for glycogen storage in the liver decreases along with the reduced liver parenchyme. Therefore, catabolism becomes predominant because protein is required for gluconeogenesis. Because BCAAs, such as valine, leucine, and isoleucine, are absorbed in the peripheral tissue, patients with cirrhosis have a lower concentration of the BCAAs and a higher concentration of the aromatic amino acids in the blood compared with healthy people. Thus, BCAA supplementation inhibits proteolysis and decreases the influx of toxic materials via the blood-brain barrier. Furthermore, it plays an important role in muscle metabolism, leading to glutamine production that is useful for detoxifying ammonia [262,263]. According to recent meta-analyses [264-266], oral BCAAs might be beneficial in managing OHE and should be used as an ancillary pharmacological option. However, intravenous BCAAs have no effect on episodic HE [209,240,267].
Others
Because albumin has great anti-inflammatory and immunomodulatory properties, it might be helpful in improving the overall survival time of patients with decompensated liver cirrhosis [268-270]. According to recent research in patients with West-Haven criteria grade ≥2 HE [271], those treated with a combination of lactulose and intravenous albumin (1.5 g/kg/day) showed a better recovery rate within 10 days than those treated with lactulose alone (75% vs. 53.3%, P=0.03) (Table 5).
In addition, polyethylene glycol (PEG), an osmotic laxative, might be tried. Its postulated mechanism of action is flushing ammonia out of the gut, like the nonabsorbable disaccharides [240]. A single RCT comparing PEG (4 liters over 4 hours via oral administration or nasogastric tube) to lactulose only showed it to be superior in terms of clinical improvement over a 24-hour period, documented by a greater decrement in the HE scoring algorithm (Δ 1.5 vs. Δ 0.7, P=0.002) and a shorter median time to resolution (1 day vs. 2 days, P=0.01) [272]. However, further studies are required to assess its efficacy and safety.
Flumazenil, an antagonist of the benzodiazepine receptor, might improve consciousness among patients with severe HE; however, its effect is temporary, and survival time is not improved [273]. Therefore, it is not recommended as a first-line regimen. Nonetheless, it can be used in patients with HE caused by benzodiazepine. Levocarnitine or sodium benzoate might be effective in managing HE because they can lower plasma ammonia concentrations [274,275].
Liver transplantation
Patient with acute liver failure and HE can be considered for liver transplantation because of their poor prognosis [93]. In cases of recurrent OHE, the severity is associated with its overall prognosis [276], and the overall survival rate after an episode of OHE was 42% and 23% at 1 and 3 years, respectively [277]. Therefore, liver transplantation should be considered for such patients. Furthermore, liver transplantation is also indicated in patients with severe HE who do not respond to the above medical treatments.
[Recommendations]
1. Precipitating factors of HE include gastrointestinal bleeding, infection, constipation, infection, excessive intake of protein, dehydration, renal function disorder, electrolyte imbalance, psychoactive medication, and acute hepatic injury. So first, those factors should be recognized and managed. (A1)
2. To manage acute episodic over t HE, non-absorbable disaccharides (e.g., lactulose, lactitol) are recommended. Enema is recommended in severe HE (West Haven criteria grade ≥3) or a clinical situation in which oral intake is inappropriate. (A1)
3. Rifaximin might be combined with non -absorbable disaccharides to treat patients with HE. (B1)
4. Oral BCAA and intravenous LOLA or albumin can be used additionally. (B2)
5. Liver transplantation is indicated in patients with severe HE who do not respond to the medical treatments. (A1)
Prevention of overt HE
Medical therapy
Among patients with OHE, 50–70% will experience a recurrence within 1-year, so secondary prevention for OHE should be started after the first event. As the first-line therapy, nonabsorbable disaccharides (lactulose [278,279], lactutol [280]) should be used. A dose of 30–60 mL of lactulose, allowing 2–3 stools per day, in patients who recovered from acute episodes of OHE significantly reduced the recurrence of OHE (19.6%) compared with the control group (46.8%) [278]. In cases of lactulose/lactitol intolerance, rifaximin can be used as single therapy (400 mg tid or 550 mg bid) [281]. According to a case-control study that included decompensated liver cirrhosis patients, a median 2 years of rifaximin therapy significantly lowered the recurrence of OHE compared with the control group (31.5% vs. 47%, P=0.034) [281].
A prospective RCT by Bass et al. [282] found that 6-months of rifaximin therapy significantly lowered the recurrence of OHE compared with the placebo group (hazard ratio [HR], 0.42; 95% CI, 0.28–0.64); about 91% of that study population used lactulose concomitantly. Non-absorbable disaccharide and rifaximin combination therapy can reduce the recurrence of OHE more than each single therapy [240,282], and it is therefore recommended for recurrent OHE. These medical treatments can effectively prevent OHE recurrence and improve the survival times of patients with OHE [281,283]. Long-term treatment with rifaximin raised concerns about the risk of Clostridium difficile (C. difficile) infection, but recent studies found that C. difficile infection was not increased by rifaximin treatment compared with the control group [283-285].
Long-term oral BCAA treatment is recommended for patients whose oral diet is insufficient because it can improve symptoms and reduce the recurrence of OHE [266,286]. In a meta-analysis of 16 RCTs, oral BCAA reduced the recurrence of OHE (HR, 0.73; 95% CI, 0.61–0.88), but the overall survival time did not differ between the two groups [266].
LOLA can reduce the recurrence of HE. In an RCT including 150 patients, oral LOLA (6 g three times per day) for 6 months significantly reduced the recurrence of OHE (HR, 0.39; 95% CI, 0.17–0.87) [287]. Nonetheless, recent meta-analyses have shown that oral LOLA was not more effective than lactulose or rifaximin for OHE prevention [287,288].
In patients with intractable ascites, intravenous albumin infusion can prevent OHE. A recent prospective RCT showed that long-term intravenous albumin (40 g per week) infusion significantly lowered the risk of grade 3 or 4 OHE (HR, 0.48; 95% CI, 0.37–0.63) and improved overall survival times (HR, 0.62; 95% CI, 0.40–0.95) [269].
Education
A structured educational intervention has been reported to improve patient adherence to prophylactic therapy and reduce readmission with OHE [289]. According to an RCT of 39 patients with a history of OHE, a 15-minute educational session reduced the risk of OHE-related hospitalization (HR, 0.14; 95% CI, 0.02–0.77) [269]. The education of patients and caregivers should include 1) the effects and potential side effects (e.g., diarrhea) of the prescribed medication (lactulose, rifaximin, and so on), 2) the importance of adherence, 3) early symptoms and signs of recurring OHE, and 4) actions to be taken if a recurrence begins [209].
Nutritional management and exercise
Nutritional deficits and subsequent sarcopenia are known to increase complications, including HE [290,291], and lower the overall survival times of cirrhotic patients [292-294]. Therefore, adequate assessment and intervention for nutritional status are recommended. Because most decompensated cirrhotic patients are malnourished, daily energy intake should be 35–40 kcal/kg, and protein intake should be 1.2–1.5 g/kg. Long-term protein restriction should be avoided because it can induce protein catabolism, hepatic dysfunction, and sarcopenia [295].
To take in enough energy, small frequent meals (4–6 times per day including a night snack) improve the long-term prognosis for liver cirrhosis patients while preventing sarcopenia [296], but the direct effect that small meals and a night snack has on OHE prevention has not been fully established.
Exercise can improve the long-term outcomes of cirrhotic patients [297,298]. In particular, cirrhotic patients usually have decreased skeletal muscle volume [291] because hyperammonemia hinders the synthesis of skeletal muscles [299,300]. An adequate exercise program can prevent muscle loss [301], enhance effective ammonia metabolism, and prevent OHE recurrence. However, exercise can temporarily increase the portal pressure in OHE patients [297,298], and it could increase the risk of a fall or fracture in malnourished patients. Therefore, adequate nutritional support should precede exercise therapy (Fig. 5).
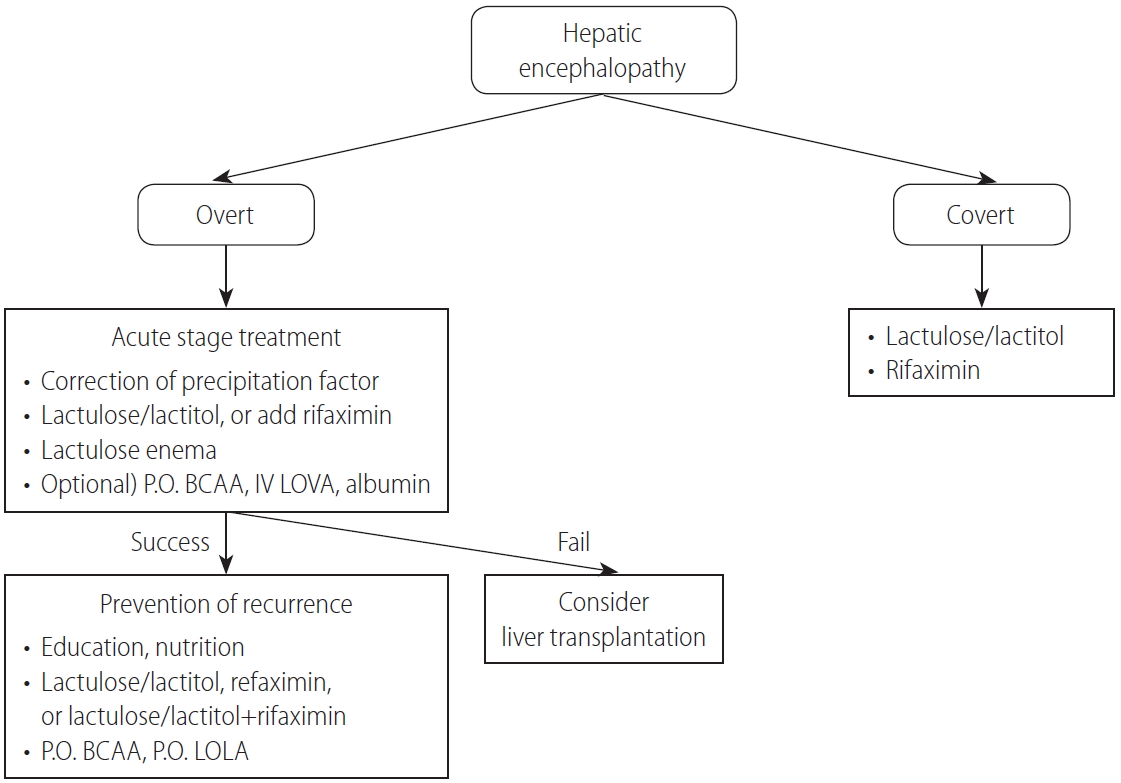
The treatment and prevention of recurrence of hepatic encephalopathy. P.O., per oral; BCAA, branched-chain amino acid; IV LOLA, intravenous L-ornithine-L-aspartate.
[Recommendations]
1. A nonabsorbable disaccharide (lactulose, lactitol) or rifaximin, as single or combined therapy, is recommended for the prevention of overt HE recurrence. (A1)
2. Oral branched-chain amino acid or oral LOLA supplementation can prevent the recurrence of overt HE. (B1)
3. Adequate education of patients and caregivers at the time of discharge is needed to reduce the recurrence of overt HE. (B1)
4. Nutritional assessment and management are needed for decompensated liver cirrhosis patients who experienced overt HE. (B1) Long-term protein restriction should be avoided, and adequate energy and protein intakes are necessary. (B1)
Covert HE
Definition
CHE is regarded as the preclinical stage of OHE, and it includes West-Haven criteria grade 1 and MHE, which is the mildest form of HE [210]. It is difficult to diagnose CHE it because it can be diagnosed only by psychometric or neurophysiologic examination and is without definite clinical manifestations, such as disorientation or asterixis. Furthermore, it is difficult to clinically distinguish MHE and grade 1 HE. Therefore, MHE and grade 1 HE from the West-Haven criteria are often defined as a single syndrome called CHE. Because the concept of CHE was initiated by ISHEN in 2011, most previous studies have been done on MHE; little research has been done on CHE, including West-Haven criteria grade 1 HE [210].
The prevalence of MHE is 22–78% of patients with liver cirrhosis, although the rate can differ depending on the diagnostic method [226,302-308]. The prevalence of MHE is related to prior episodes of OHE, age, severity of liver disease, and the presence of EVs [309]. In a study using the psychometric HE score (PHES) in a single institution in Korea, MHE was seen in 25.6% of patients with cirrhosis, including 20.2% of those in Child-Pugh A, 42.9% in Child-Pugh B, and 60% in Child-Pugh C [310].
Clinical significance
Patients with CHE have impaired cognitive functions such as attention, executive functions, visuospatial perception, psychomotor speed, and reaction times [311]. Those impaired cognitive functions interfere with daily functioning, such as social interactions, alertness, emotional behavior, sleep, home management, and recreation, and lower the quality of life [214,304,312].
Patients with CHE are at risk of falls and fractures [313,314], and their poor cognitive performance increases the risk that they will lose their jobs [315]. Therefore, CHE increases the burden on both individual patients and society. CHE is regarded as the preclinical stage of OHE because of the increased risk of progression to OHE [226,302], and CHE is associated with worsened survival times [316,317]. However, it is difficult to distinguish whether the shortened survival is caused by CHE or hepatic dysfunction.
Diagnosis
To diagnose CHE, the patient must have 1) a disease that can lead to CHE, such as liver cirrhosis or a portosystemic shunt, 2) no other neurological disease, 3) no neurological manifestation such as disorientation or asterixis, and 4) abnormal cognitive or neurophysiologic functioning.
Paper and pencil testing
One of the paper and pencil tests, PHES, consists of five tests (digit symbol test, NCT-A, NCT-B, serial dotting, and line tracing) that measure attention, psychomotor speed, visual perception, and visuo-spatial orientation [318]. The PHES has been widely used to diagnose CHE and has a sensitivity of 96% and a specificity of 100% [319]. It was developed in Germany and has been validated in several countries, including Korea [310,319-324]. It is recommended that at least two of the NCT-A, NCT-B, block design test, and digit symbol test be performed if the full PHES cannot be used due to copyright issues or in places where the PHES has not been validated [208].
The Korean paper and pencil test (KPPT) to evaluate MHE in Korean patients with liver cirrhosis was developed with the support of the Korean Association for the Study of the Liver [325,326]. The KPPT consists of six tests: NCT-A, NCT-B, digit span test (DST), symbol digit modality test (SDMT), word list memory test, and Medical College of Georgia Complex figures. The KPPT short version is configured to be relatively simple to use and contains the NCT-A, NCT-B, DST, and SDMT. A recent prospective multicenter study validated the KPPT short version in Korean patients with liver cirrhosis [325]. The KPPT is available at http://encephalopathy.or.kr/inspection [326].
Computerized testing
Inhibitory control test (ICT)
The ICT is a computerized test that evaluates attention, response inhibition, and working memory [306,327]. In the ICT, the subject is instructed to respond to alternating patterns of the letters X and Y, called the target. Non-alternating presentations of the letters X and Y, called lures, are randomly planted within the sequence of letters. This test evaluates the response times of the subjects and the response rate to the target and lures. The sensitivity and specificity of the ICT are 87% and 77%, respectively, and it is highly reproducible [327]. However, it has not been validated for Korean patients.
Stroop test
The Stroop test evaluates psychomotor speed and cognitive flexibility [328] using two components (the “off” and “on” states). In the “off” state, subjects match the color of the symbol. In the “on” state, subjects match the color of the word when the color of the word and the meaning of the word are incongruent, which evaluates response inhibition.
The computer-based Stroop test shows a sensitivity of 89.1% and a specificity of 82.1% when using the paper and pencil test as a standard test [329]. A recent prospective multicenter study in the US showed that Stroop test had high sensitivity and acceptable inter-center agreement [330]. In addition, the Stroop test has good test–retest reliability, and it has the advantage that it can be easily administered using a smartphone. A Korean Stroop test was developed, and a recent study showed that the Korean Stroop test is valid for diagnosing MHE (area under the curve, 0.74; 95% CI, 0.66–0.83, P<0.001) [331]. The Korean Stroop test is available at http://encephalopathy.or.kr/inspection [326].
Neurophysiological testing
EEG
EEG is a test that reflects cerebral cortical neuronal activity. In patients with CHE, a quantitative EEG analysis shows an increase in the relative power of the θ band and a decrease in the mean dominant frequency [332]. However, EEG can be affected by various conditions that can affect cortical function. In addition, it requires a technician and a neurologist and is associated with both interobserver and intraobserver variability.
Critical flicker frequency (CFF)
CFF measures the frequency at which light begins to flicker noticeably. CFF is highly correlated with paper and pencil testing [333]. In a meta-analysis of nine studies using CFF, the sensitivity and specificity for diagnosing CHE were 61% and 79%, respectively [334]. However, it is not applicable to patients with red-green blindness or Korean patients with cirrhosis because it has not been validated in Korea.
Other tests
The animal naming test (ANT) is a semantic fluency test that consists of listing the names of as many animals as possible in 1 minute. In a prospective study conducted in Italy, the sensitivity and specificity of the ANT for diagnosing CHE were 78% and 63%, respectively, when the cut-off was less than 15, and the ANT was a significant predictor for the development of OHE [307]. In a recent prospective study in Germany, the sensitivity and specificity were 31% and 98%, respectively, when the cut-off was less than 15, and they suggested 23 as a cut-off to increase sensitivity [335]. Nabi et al. [336] reported that a combination of age, sex, and the responses to 4 Sickness Impact Profile (SIP) questions that are highly related to CHE identified patients with CHE with more than 80% sensitivity. However, that test needs to be validated further. Some studies reported that serum cytokines, such as interleukin (IL)-6, IL-17a, interferon-γ [337-339] and 3-nitrotyrosin [340,341], are associated with CHE. However, further studies are needed on the pathophysiology of CHE and the role of the markers in CHE.
Diagnosis and screening
CHE has no clinical signs of HE. It can show abnormalities in cognitive functions in various fields, but each field is not reduced to the same extent. In addition, because one test cannot judge the abnormality in all fields and agreement is poor between tests [342,343], a combination of least two tests is recommended for a diagnosis of CHE [210]. For multicenter studies, a paper and pencil test and one computerized or neurophysiologic test are recommended for a CHE diagnosis. A single institution can use one test that has been validated locally [209].
Because CHE decreases patient quality of life, increases socioeconomic burden, and hastens mortality, it might be necessary to test and diagnose all patients at risk. However, that would increase costs. Therefore, it is advisable to perform a diagnostic test in patients with a history recent of falls or traffic accidents and patients who report a low quality of life or complaints about daily living, such as those who complain of sleep disturbance or a loss of concentration or memory [209,344].
Because most patients with CHE are diagnosed at an outpatient clinic, the screening tests should be performable without any special tools and with high sensitivity, such as the four questions from SIP, the ANT, and the Stroop test using a smartphone.
Treatment
Most studies about treating CHE were performed in a small number of patients with a short duration of treatment. In addition, most studies have focused on improving cognitive function and quality of life; studies are still needed on extending survival and reducing readmissions or the development of OHE.
As with OHE, it is known that nitrogenous substances, especially ammonia, play a major role in CHE. Therefore, treatments can be given to reduce ammonia. The most studied treatment is lactulose, which showed a marked improvement in cognitive function and quality of life [304,345] and decreased the development of OHE [345], compared with the placebo.
Probiotics alter the gut microbiome and inhibit ammonia production in the intestine, thereby improving cognitive function and decreasing the development of OHE [346-348]. However, studies on the effects of probiotics in patients with CHE have low evidence levels [349]. Additional studies are needed to determine the beneficial probiotic species and optimal doses. Rifaximin and nonabsorbable antibiotics also improved cognitive function and quality of life [305] and improved driving ability [350]. However, rifaximin failed to establish non-inferiority over lactulose in non-inferiority studies [351], and lactulose treatment is more cost-effective than rifaximin therapy [350]. Therefore, further studies on the role of rifaximin in the treatment of CHE are warranted. Although LOLA [352], BCAA [353], acetyl L-carnitine [354,355], and nutrition therapy [308] have been reported to improve cognitive function, there is still a lack of evidence for those treatments.
[Recommendations]
1. In patients with liver cirrhosis, the KPPT or the Korean Stroop test can be used to diagnose CHE. (B2)
2. Treatment with lactulose (B1) or rifaximin (B2) can be used to improve cognitive function and quality of life in patients with CHE.
HE and health-related quality of life (HRQoL)
HRQoL in patients with cirrhosis is lower than that of patients with chronic liver disease without cirrhosis. The HRQoL of cirrhotic patients with HE is particularly low [356]. Patients with HE suffer from various degrees of altered consciousness, personality changes, impaired intellectual functioning, and neuromuscular dysfunction. Although HE is not immediately life-threatening, it can greatly interfere with a patient’s functioning, social interactions, and sense of well-being [357]. The occurrence of HE is associated with various complications that can also adversely affect HRQoL. Therefore, the independent effect of HE on HRQoL is not easily measured. Because patients with OHE are unaware of their disease (anosognosia) [358], alterations in their behavior and abilities are more easily recognized by the people living with them than by the patients themselves. The presence of OHE negatively affects both mental and physical functioning, whereas MHE mainly has negative effects on mental health. Several studies have shown that the HRQoL of patients with MHE is lower than that of patients without HE [312,359-362]. Therefore, we suggest that patients with cirrhosis should be screened for the early detection and treatment of HE to improve their HRQoL.
Measuring of health-related quality of life in patients with HE
HRQoL is measured using self-administered, standardized questionnaires in which patients report their health status. The questionnaires are classified as generic and disease-specific [363]. Because generic questionnaires provide an overview of HRQoL, usually taking into account the physical, mental, and social aspects of a patient’s health status, generic questionnaires have the advantage of depicting the relative impacts of different diseases. However, generic questionnaires have the disadvantage of insensitivity to clinically important changes. Thus, generic questionnaires are often combined with disease-specific questionnaires. The most widely used generic questionnaires for measuring HRQoL are the SIP, Nottingham Health Profile (NHP), and Medical Outcomes Study Short Form-36 (SF-36) [364]. The SIP consists of 136 items that measure 12 domains. It requires several minutes to complete, and patients with cognitive dysfunctions sometimes fail to complete it [365]. The NHP measures distress and is useful in patients with moderate or severe disability, but it is not very sensitive to mild disability [366]. The SF-36 is applicable to a wide range of patients, from those with a severe disability to the general population. The SF-36 is easy to complete and has high sensitivity, which makes it the best and most widely used scale in clinical practice. It contains 36 questions that are split into eight domains and provides a physical component summary (PCS) and a mental component summary (MCS) (Supplementary Table 1) [367].
Disease-specific questionnaires have been developed for a variety of chronic diseases, such as renal failure, heart failure, liver cirrhosis, diabetes, and osteoarticular diseases, that greatly affect the HRQoL of patients. Liver-disease-specific questionnaires include the Chronic Liver Disease Questionnaire (CLDQ), Liver Disease Quality of Life (LDQOL), Short Form Liver Disease Quality of Life (SF-LDQOL), and Liver Disease Symptom Index 2.0 (LDSI). The CLDQ comprises 29 questions split into six domains, with domain scores and an overall score presented as 1–7 scales. Higher scores on the CLDQ represent better HRQoL. The CLDQ is short, easily applicable, and correlates with the severity of liver disease [368]. The LDQOL uses the SF-36 and adds 12 liver-specific scales comprising 75 questions. All scales are scored from 0–100, with higher scores representing better HRQoL [369]. The SF-LDQOL uses the SF-36 and adds 36 Likert questionnaires; it is also scored from 1–100 [370]. The LDSI uses 18 items to measure the impact and severity of a patient’s liver disease on daily activities in nine areas (Supplementary Table 2) [371].
Influence of HE on health-related quality of life
Although there is a large consensus about the direct and profound effect that HE has on HRQoL, most studies have focused on MHE [208,372,373]. A small study about the HRQoL of patients with and without HE compared 18 patients experiencing OHE with 57 patients without a previous episode. Patients with a previous episode of OHE had significantly low SF-36 PCS and MCS scores. However, patients with MHE were affected in only one domain, physical functioning, of the SF-36 [374]. One study of 160 cirrhotic patients undergoing liver transplantation found that patients with MHE or OHE had a lower MCS than patients without HE [357].
Cognitive impairment of patients with HE mainly affects areas that require multiple and complicated functions, such as attention, visuospatial abilities, psychomotor speed, balance, and coordination, rather than language or general intellect. In other words, patients can perform daily activities such as wearing clothes or using the toilet, but their overall planning or cognitive function and exercise performance might suffer [318]. Because driving a vehicle requires comprehensive performance and a strategic way of thinking, patients with MHE require attention while driving [375-378]. Cirrhotic patients engaged in professions that required sustained attention and motor coordination are more severely affected by MHE than those with jobs that require mainly verbal abilities. In an outpatient cohort with cirrhosis, up to 60% of blue collar workers lost their jobs, versus only 20% of white collar workers [236].
A disruption of normal sleep-wake patterns is another early sign of HE [379], and regular sleep is a key indicator of perceived health status. Sleep disturbances are included as relevant items in the assessment of HRQoL in the NHP questionnaire, and sleep disturbances in patients with HE negatively affect HRQoL [380]. Patients with MHE report a decrease in the quality of their sleep and in their physical and mental HRQoL [214,381].
Psychological status and a patient’s mood can affect the course of a disease and treatment response, and depression affects social functioning, physical abilities, and health status [382]. Cirrhotic patients suffer not only from liver disease itself, but also from decreased quality of life in the form of poor work performance and an increased risk of accidents. Therefore, there is a need for extensive social attention and research on public social support systems and economic support for cirrhotic patients.
Effects of treatment on HE and health-related quality of life
Although many studies have aimed at improving HE, relatively few studies have aimed at a significant improvement in the HRQoL of patients with HE [383]. Prasad et al. [304] first investigated the effect of treatment-related improvements in cognitive function on HRQoL. Patients with MHE treated with lactulose for 3 months showed a significant improvement in their HRQoL on several SIP subscores, particularly in emotional behavior, mobility, sleep/rest, and recreation and pastimes. In an 8-week study of rifaximin therapy in patients with MHE, the patients showed significantly improved scores in both neuro-psychometric performance and the SIP [305]. Another study reported that rifaximin therapy to prevent a recurrence in patients with HE favorably affected HRQoL as measured by CLDQ scores [384]. Treating OHE patients with oral LOLA [385] and MHE patients with acetyl-L-carnitine [354] also improved HRQoL. However, a 60-day course of probiotic yogurt supplementation had no significant effects on HRQoL in 25 patients with cirrhosis [346].
To date, insufficient studies have been done to establish an association between improved HRQoL and treatments for OHE and MHE. Considering that 10% or more of cirrhotic patients have HE, and 50% of cirrhotic patients with MHE who have not been treated can progress to OHE within 4–24 months [386], it is necessary to change the paradigm of treatment to improve the HRQoL of patients.
[Recommendations]
1. Active diagnosis and treatment of HE improves patient’s health-related quality of life. (A1)
2. Health-related quality of life in patients with HE is assessed by self-administered, standardized questionnaires and can be measured using either generic or disease-specific questionnaires. (B2)
Acknowledgements
KASL Committee for Revision of the Clinical Practice Guidelines for Liver Cirrhosis: Varices, hepatic encephalopathy, and related complications: Jae Young Jang (Committee Chair, Soonchunhyang University College of Medicine), Sang Gyune Kim (Soonchunhyang University College of Medicine), Yeon Seok Seo (Korea University College of Medicine), Moon Young Kim (Yonsei University Wonju College of Medicine), Beom Kyung Kim (Yonsei University College of Medicine), Byung Seok Kim (Catholic University of Daegu School of Medicine), Sung Eun Kim (Hallym University College of Medicine), Ki Tae Suk (Hallym University College of Medicine), Do Seon Song (The Catholic University College of Medicine), Jae Jun Shim (Kyung Hee University College of Medicine), Seung Kak Shin (Gachon University College of Medicine), Yun Bin Lee (Seoul National University College of Medicine), Eun Sun Jang (Seoul National University College of Medicine), Dae Won Jun (Hanyang University College of Medicine).
Notes
Authors’ contributions
Jae Young Jang (Soonchunhyang University College of Medicine): Introduction Writing and Guidelines General Management
Sang Gyune Kim (Soonchunhyang University College of Medicine): Manuscript writing for introduction of HE
Yeon Seok Seo (Korea University College of Medicine): Manuscript writing for introduction of varices part, surveillance of varices, preventing the formation and progression of EVs
Moon Young Kim (Yonsei University Wonju College of Medicine): Guidelines editing and manuscript organization
Beom Kyung Kim (Yonsei University College of Medicine): Manuscript writing for management of overt HE
Byung Seok Kim (Catholic University of Daegu School of Medicine): Manuscript writing for prevention of first EVs bleeding
Sung Eun Kim (Hallym University College of Medicine): Manuscript writing for HE and health-related quality of life
Ki Tae Suk (Hallym University College of Medicine): Manuscript writing for definition of gastric varices and prevention of primary bleeding, other variceal bleeding and portal hypertensive gastropathy
Do Seon Song (The Catholic University College of Medicine): Manuscript writing for covert HE
Jae Jun Shim (Kyung Hee University College of Medicine): Manuscript writing for management of bleeding from gastric varices and prevention of rebleeding
Seung Kak Shin (Gachon University College of Medicine): Manuscript writing for prevention of EVs rebleeding
Yun Bin Lee (Seoul National University College of Medicine): Manuscript writing for diagnosis and management of acute EVs bleeding
Eun Sun Jang (Seoul National University College of Medicine): Manuscript writing for definition of HE, prevention of overt HE
Dae Won Jun (Hanyang University College of Medicine): Manuscript writing for diagnosis of HE
Conflicts of Interest
Jae Young Jang: Received grants from Yuhan; received honoraria from BMS, Gilead, Dong-A ST, Yuhan, and Bukwang
Sang Gyune Kim: Received grants from GE healthcare, Samsung Medison, BMS, Samil, Ildong; received honoraria from BMS, MSD, Gilead, Dong-A ST, Daewoog; consulted Samsung Medison
Yeon Seok Seo: Received honoraria from Yuhan, Bukwang, Samjin, Gilead, Dong-A ST, Samil, Deawoong, Chongkundang, Abbvie, MSD
Moon Young Kim: Received grants from Samjin, Yuhan, Bukwang; received honoraria from BMS, Gilead, Dong-A ST, Yuhan, Daewoong
Beom Kyung Kim: Received honoraria from Samil, Dong-A ST, Yuhan, Celltrion, Deawoong, BMS, Chongkundang
Byung Seok Kim: Received grants from Gilead, Abbvie, MSD, Dong-A ST, Daewoong, Ildong, Chongkundang; received honoraria from Gilead, BMS, Abbvie, Yuhan SK chemical
Sung Eun Kim: Received grants from Daewoong
Ki Tae Suk: Nothing to disclose.
Do Seon Song: Received grants from Alfa Wassermann Jae Jun Shim: Received grants from Samil; received honoraria from MSD, Yuhan; consulted Gilead
Seung Kak Shin: Nothing to disclose.
Yun Bin Lee: Nothing to disclose.
Eun Sun Jang: Received honoraria from Chongkundang
Dae Won Jun: Received grants from Yuhan, BMS, Gilead, Dong-A ST, Hanwha, Samil, Diagen; received honoraria from Yuhan, BMS, Gilead, Dong-A ST, Hawha, Daewoong
Abbreviations
ANT
animal naming test
BCAAs
branched-chain amino acids
BRTO
balloon-occluded retrograde transvenous obliteration
cACLD
compensated advanced chronic liver disease
CFF
critical flicker frequency
CHE
covert hepatic encephalopathy
CI
confidence interval
CLDQ
Chronic Liver Disease Questionnaire
CPG
clinical practice guideline
CT
computed tomography
DST
digit span test
EEG
electroencephalography
EIS
endoscopic injection sclerotherapy
EVL
endoscopic variceal ligation
EVO
endoscopic variceal obturation
EVs
esophageal varices
GOV
gastroesophageal varices
GRADE
Grading of Recommendations
HE
hepatic encephalopathy
HR
hazard ratio
HRQoL
health-related quality of life
HVPG
hepatic venous pressure gradient
ICT
inhibitory control test
IGV
isolated gastric varices
IL
interleukin
ISHEN
International Society for Hepatic Encephalopathy and Nitrogen Metabolism
ISMN
isosorbide-5-mononitrate
KASL
the Korean Association for the Study of the Liver
KPPT
Korean paper and pencil test
LDQoL
Liver Disease Quality of Life
LDSI
Liver Disease Symptom Index
LOLA
L-ornithine-L-aspartate
MCS
mental component summary
MHE
minimal hepatic encephalopathy
MRI
magnetic resonance imaging
NCT
number connection test
NHP
Nottingham Health Profile
NSBBs
nonselective beta-blockers
OHE
overt hepatic encephalopathy
OR
odds ratio
PARTO
vascular plug-assisted retrograde transvenous obliteration
PCS
physical component summary
PEG
polyethylene glycol
PHES
psychometric hepatic encephalopathy score
PPI
proton pump inhibitor
PRBC
packed red blood cell
RCTs
randomized controlled trials
RR
relative risk
SDMT
symbol digit modality test
SF-36
Medical Outcomes Study Short Form-36
SF-LDQOL
Short Form Liver Disease Quality of Life
SIP
Sickness Impact Profile
SVR
sustained virologic response
TIPS
transjugular intrahepatic portosystemic shunt
SUPPLEMENTAL MATERIAL
Supplementary material is available at Clinical and Molecular Hepatology website (http://www.e-cmh.org).
Generic questionnaire assessing health-related quality of life
Disease specific questionnaires for health-related quality of life in chronic liver disease patients