The development of mesenchymal stem cell therapy in the present, and the perspective of cell-free therapy in the future
Article information
Abstract
Cirrhosis is a chronic condition that can lead to liver failure. Currently, the viable option for decreasing mortality is liver transplantation. However, transplant surgery is highly invasive. Therefore, cell-based therapy has been developed as an alternative. Based on promising findings from preclinical research, some new trials have been registered. One of them was autologous bone marrow cell infusion therapy and found that ameliorating liver fibrosis activated liver regeneration. Now, majority of trials focus on low-immunogenicity mesenchymal stem cells (MSCs) appropriate for allogeneic administration. However, despite about 20 years of research, only a limited number of cell-based therapies have entered routine practice. Furthermore, potential shortcomings of cell-based therapy include a limit on the number of cells, which may be administered, as well as their failure to infiltrate target organs. On the other hand, these research show that MSCs act as “conducting cells” and regulate host cells including macrophages via extracellular vesicles (EVs) or exosome signals, leading to ameliorate liver fibrosis and promote regeneration. Therefore, the concept of cell-free therapy, which makes use of cell-derived EVs or exosomes, is attracting attention. Cell-free therapies may be safely administered in large doses and are able to infiltrate target organs. However, development of cell-free therapy exhibits its own set of challenges and such therapy may not be completely curative in the context of liver disease. This review describes the history of cell-based therapy research and recent advances in cell-free therapy, as well as discussing the need for more effective therapies.
INTRODUCTION
Chronic liver disease and cirrhosis are life-threatening conditions —causing approximately 1–1.2 million deaths per year— arising in response to several etiologic factors including hepatitis virus infection, alcohol, nonalcoholic steatohepatitis, autoimmunity, and primary biliary cholangitis [1-3]. In recent years, medical advances have significantly improved the prognosis of liver disease [4]. Since the liver is highly regenerative, fibrosis can be at least partially ameliorated and liver function improved by eliminating the causative factor underlying dysfunction. However, once cirrhosis advances beyond a certain point, elimination of the causative factor will no longer facilitate regression of fibrosis, and cirrhosis may progress to carcinogenesis and/or liver failure [5-7]. Therefore, the number of deaths attributable to cirrhosis remains high [8]. Although liver transplantation is currently the only curative treatment for decompensated cirrhosis [9,10], significant limitations include a shortage of donors, high cost, highly invasive nature of the surgery, use of immunosuppressive agents to prevent graft rejection, and the potential for severe complications [11]. Thus, cell-based therapy is of interest as an alternate and novel strategy to ameliorate liver fibrosis, reduce inflammation, and promote liver regeneration.
A good understanding of the mechanisms underlying liver fibrosis may contribute to therapeutically promoting its reversal. During liver injury, normally-quiescent hepatic stellate cells (HSCs) become activated by several cytokines and trophic factors, including transforming growth factor-β and platelet-derived growth factor, leading to production of excessive extracellular matrix (ECM) constituents by activated myofibroblasts. Once the hepatotoxic stimulus is removed, the number of activated myofibroblasts regresses via apoptosis, senescence, and reversion to an inactive phenotype. Furthermore, matrix metalloproteinases (MMPs) are capable of ECM degradation. Thus, understanding the molecular mechanisms involved in HSC activation, ECM degradation, and myofibroblast involution or deactivation could facilitate manipulation of these processes and may lead to rational development of novel therapies for cirrhosis [12]. Current therapeutic approaches to liver fibrosis are divided into five categories: 1) controlling primary liver disease [13,14], 2) targeting HSC receptor-ligand interactions and intracellular signaling,15-18 3) inhibiting fibrogenesis or matrix degradation [19,20], 4) decreasing the number of activated HSCs [21,22], and 5) cell-based therapy. Although cell-based therapy is mainly concerned with ameliorating liver fibrosis, it is also capable of producing other beneficial effects (including decreasing inflammation and promoting liver regeneration).
The ability of cell-based therapy to promote liver regeneration is also important, however. Regenerative factors including hepatocyte growth factor (HGF), oncostatin M (OSM), and Wnt3A — which induce differentiation and influence phenotypic fate decisions of hepatic stem/progenitor cells— are secreted by macrophages [23,24]. Furthermore, regeneration automatically occurs secondary to the anti-inflammatory and anti-fibrotic effects of cell-based therapy. It has been demonstrated that cell-based therapy, via dampening liver inflammation and ameliorating liver fibrosis, improves liver volume and function, including albumin synthesis and prothrombin time [25].
At the inception of cell-based therapy during the period from 2003, first clinical study of autologous bone marrow cell infusion (ABMi) therapy was started in 2003, in which the patient’s own bone marrow cells are harvested and re-injected [26-28]. In this therapy, ABMi ameliorates liver fibrosis and improves liver function in cirrhotic patients (as indicated by increased serum albumin levels, decreased ascites, and decreased Child-Pugh scores) without inducing any major adverse events. ABMi also promotes a sequential activation of liver inherent regeneration in human livers [29-31]. However, it is invasive, requiring general anesthesia in order to harvest 400 mL of bone marrow. Eligibility for ABMi requires a total bilirubin level of ≤3.0 mg/dL and a platelet count ≥50,000 per µL, representing criteria not met by all cirrhotic patients. Therefore, attention was shifted from bone marrow cells in general to specific stem cell subtypes. Research has focused largely on hematopoietic stem cells [32,33], mesenchymal stem cells (MSCs) [34-36], and bone marrow-derived macrophages [37,38]. In particular, MSCs and macrophages have been the mainstays of cell-based therapy, and their characteristics as well as relevant clinical trials will be described in the first half of this review.
More recently, it was noted that stem cell-derived extracellular vesicles (EVs) or exosomes also exhibit a therapeutic effect and may thus have potential for clinical application [39,40]. While the development of cell-free therapy is still in its infancy, EV and exosome characteristics as well as relevant clinical trials will be described in the latter part of this review. Advantages and limitations of cell-based and cell-free therapies are discussed and future prospects regarding cirrhosis therapy are presented (Fig. 1).
CELL-BASED THERAPY: CHARACTERISTICS OF MSCS AND MACROPHAGES
Research into MSC-mediated cell-based therapy is conducted globally. These cells are relatively easy to obtain, maintain, expand, and cryopreserve, all while maintaining their viability. For example, MSCs can be obtained not only from bone marrow but also from medical wastes such as adipose tissue, umbilical cord tissue, and dental pulp [41-51]. Expression of common pluripotency markers (e.g., CD73, CD90, and CD105) is noted on MSCs, and they are able to differentiate into adipocytes, osteoblasts, and chondroblasts under the appropriate conditions [34,35].
It appears that the major therapeutic (anti-inflammatory, anti-fibrotic, anti-oxidant, and angiogenic) effects of MSCs are due to their soluble products, including chemokines, cytokines, trophic factors, EVs, and exosomes. The most popular application of MSCs is decreasing inflammation [52]. Various MSC products — such as interleukin (IL)-10, tumor necrosis factor-stimulated gen (TSG)-6, nitric oxide, indoleamine2,3-dioxygenase, and prostaglandin E2 (PGE2)— are able to inhibit T-cell activation and expansion, induce regulatory T lymphocyte, alter macrophage polarity towards less inflammatory phenotypes, and modulate natural killer cell, dendritic cell, and B-cell functions [53-56]. By such mechanisms, MSCs are able to remotely influence the activities of many cellular effectors [57]. Since recent studies suggest that MSC-conditioned medium (or exosomes present therein) are as effective as MSCs themselves, soluble factors are currently of great interest regarding their regenerative potential in the therapy of cirrhosis [7,34-36,57-60].
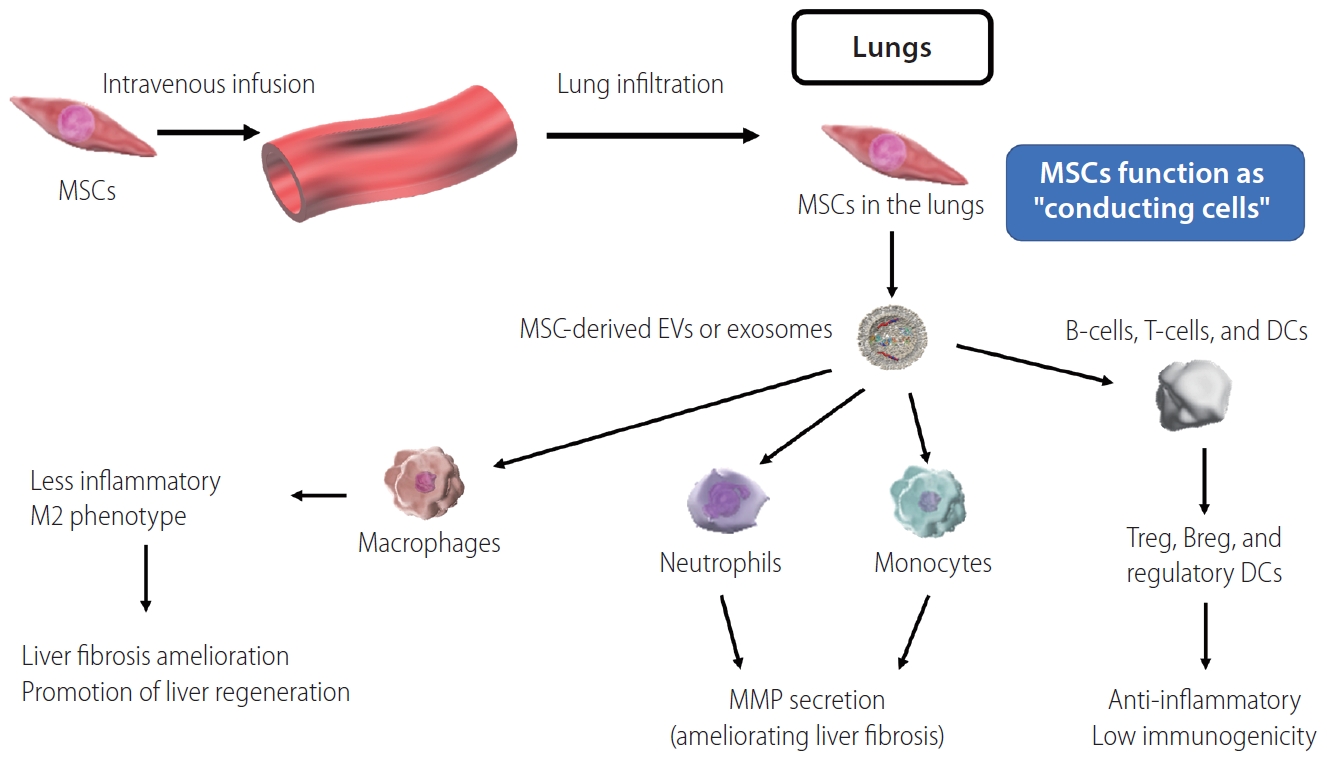
Via soluble factors, MSCs may have therapeutic effects in cirrhosis even if they do not infiltrate the injured liver. Our group was the first to demonstrate in real time (using two-photon excitation microscopy) that peripheral intravenous administration of DsRed-labeled MSCs in a murine cirrhosis model resulted in the majority of MSCs being retained within the pulmonary vasculature, while only a few reached the liver [61]. However, MSC-derived soluble factors (including EVs and exosomes) nonetheless exert a therapeutic effect on target organs (including the liver in context of cirrhosis), supporting the possibility of cell-free therapy.
Another important MSC characteristic is generally low immunogenicity. Since they express relatively low levels of major histocompatibility complex class I molecules and lack expression of major histocompatibility complex class II and co-stimulatory molecules (e.g., CD40, CD80, and CD86), they are not prone to triggering recipient immune responses [57] and are thus suitable for allogeneic (as well as autologous) injection. Clinical studies suggest their therapeutic potential across disorders affecting a wide range of organs, including the liver, nerves, lungs, heart, and intestines.
Macrophages exhibit diverse phenotypes and high plasticity [37], including two major representative phenotypes: “classically activated” (M1) and “alternatively activated” (M2). During liver injury, pro-inflammatory M1 macrophages contribute to fibrosis via activation of HSCs and myofibroblasts. In contrast, after stabilization of the liver injury process, M2 macrophages contribute to resolution of inflammation and reversal of fibrosis, the latter in part via MMPs [38]. Furthermore, as mentioned, macrophages are able to influence differentiation and phenotypic fate decisions of hepatic stem/progenitor cells through production of HGF, OSM, and Wnt3A [23,24].
Bone marrow-derived macrophages cultured in the presence of colony-stimulating factor (CSF)-1 induce regression of carbon tetrachloride-induced liver fibrosis in mice [62]. We have previously reported that a large number of peripherally-administered green fluorescent protein-labeled macrophages are able to infiltrate the liver, where they help promote liver regeneration by phagocytizing hepatocytic debris [61]. Although such findings demonstrate the therapeutic potential of macrophages in the context of cirrhosis, higher immunogenicity limits allogeneic macrophage administration. Instead, autologous macrophage administration can (e.g., via TSG-6, PGE2, and IL-13 [63-65]) help polarize host hepatic macrophages toward the less inflammatory/pro-resolving M2 phenotype, again supporting the potential of macrophages as novel cirrhotic therapeutic agents.
The same factors (TSG-6, PGE2, and IL-13) are also produced by MSCs, and contribute to host macrophages polarization in a similar manner [63-65]. Furthermore, we have previously reported that MSCs promote liver infiltration by host neutrophils and monocytes, which contribute to fibrosis amelioration via production of MMPs [61]. These findings suggest that remote MSCs act as “conducting cells” (Fig. 2), via soluble factors including EVs or exosomes modulating macrophage, neutrophil, and monocyte functions in ways that ameliorate liver fibrosis and promote liver regeneration [53-57].
CELL-BASED THERAPY: CLINICAL TRIALS INVOLVING MSCS AND MACROPHAGES FOR LIVER DISEASE-RELEVANT INDICATIONS
Although both MSCs and macrophages represent promising candidates for use in cell-based therapy, a majority of current clinical trials focus on MSCs. According to ClinicalTrials.gov, over 1,100 MSC-based clinical trials have been registered across a variety of disciplines, including 51 that began investigating cell-based therapy for liver disease after 2006. Thereafter, the number of cell-based therapy clinical trial registrations (incorporating cell sources including bone marrow, adipose tissue, umbilical cord tissue, and dental pulp, and routes of administration including peripheral venous and arterial injection [47-49,66-69]) gradually increased to a peak as the approach gained recognition. In 2017, we ourselves registered a phase I and II clinical trial (first clinical trial involving MSCs in Japan) focused on adipose-derived MSC infusion in cirrhosis patients (NCT: 03254758). Our clinical trial involving allogeneic MSCs (without using immunosuppressive agents) are proceeding into phase II after finishing phase I. However, the number of clinical trials has been declining since 2017 (Fig. 3). This trend is likely due to a decline in novelty, slow progress in translating preclinical research to clinical interventions, and a tendency of shift in focus toward cell-free therapy incorporating EVs or exosomes.

Number of clinical trials involving mesenchymal stem cells. Black bars represent trials focused on cirrhosis in general, while stacked red bars represent trials focused specifically on acute-on-chronic liver failure (ACLF).
Furthermore, to describe recent trends in MSC-oriented clinical trials, we evaluated studies (12 studies) which began or will begin after January 2017 according to ClinicalTrials.gov (Table 1). The majority of these were registered in Asian countries. Studies vary in cell source, autologous versus allogeneic nature, and etiology of cirrhosis. However, in most studies (11 of 12) MSC administration is via peripheral injection of approximately 1×106 MSC/kg per dose. Studies focus not only on cirrhosis, but also on acute-onchronic liver failure (ACLF). Since MSCs exhibit a stronger therapeutic effect during inflammation, they may be particularly effective in the context of ACLF. Altogether, many MSC-oriented clinical trials have not yet progressed beyond phase I or II.
Regarding macrophage-oriented cell-based therapy, clinical trials further elucidating macrophage characteristics have also been reported by Edinburgh University. The first-in-human phase 1 dose-escalation trial of intravenous autologous macrophage therapy incorporated nine adults with cirrhosis and model for end-stage liver disease (MELD) scores of 10–16 [70]. In this trial, macrophages were produced from patient mononuclear cell leukapheresis-derived monocytes cultured in the presence of CSF-1. Despite macrophage potential for immunogenicity, no transfusion reactions, dose-limiting toxicities, or macrophage activation syndromes were reported, and all participants remained alive and transplant-free at 1 year. Furthermore, after 1 year, MELD scores had decreased in seven out of nine patients, and a liver fibrosis marker had decreased in five out of nine patients. Because macrophages are also an essential component in liver regeneration, acceleration of research in this area is anticipated.
CELL-BASED THERAPY: ADVANTAGES AND LIMITATIONS
As alluded to, cell-based therapy has many advantages. Especially so for MSCs, cells are relatively easy to obtain, culture, expand, characterize for quality control, count, and assess for viability [41-51]. In addition, since MSC therapeutic effects are enhanced during inflammation, preconditioning can strengthen their therapeutic effects. Finally, numerous avenues of cell modification are possible. Despite administered cells becoming trapped in the pulmonary vasculature [61], existing clinical trials report few major adverse events, including pulmonary embolism [47]. Although administered macrophages are autologous due to immunogenicity-based constraints, they are now obtainable via less invasive methods [70]. Moreover, the ability to elicit and influence host macrophage development locally within the liver (i.e., without the need for exogenous administration) would be an even more attractive possibility.
However, cell-based therapy also has certain limitations. For example, concerns regarding the risk of pulmonary embolism continue to limit dosage, and administered cells do not access the target organ. Although cells do continue to exert a therapeutic effect remotely, a therapeutic agent acting directly within damaged sites would be preferable. Furthermore, given the propensity of stem cells to differentiate, it is difficult to maintain a stable phenotype during long-term culture. Therefore, commercialization would necessitate rigorous quality control. Such concerns have driven the search for alternate therapeutic strategies and cell-free therapy may overcome some of the limitations associated with cell-based therapy.
CELL-FREE THERAPY IN THE FUTURE: CHARACTERISTICS OF EVS AND EXOSOMES
The generic term for membrane-bound particles naturally released by cells is EVs; exosomes are a subtype of these [71]. Once believed to be “trash bags” for cellular debris, EVs and exosomes are now being explored for their potential as next-generation diagnostic and therapeutic tools [72]. The heterogeneous group of EVs encompasses a variety of particles of different sizes with varying contents. Exosomes, specifically, are 40–100 nm in diameter — corresponding to a density of 1.13–1.19 g/mL in sucrose solution— and can be sedimented by ultracentrifugation at 100,000 ×g [72]. They contain a mixture of proteins, messenger RNA, transfer RNA, micro RNA, genomic DNA, and complementary DNA [73].
Like stem cells, exosomes exhibit many biological activities and have shown therapeutic potential in several organ system and disease contexts. For example, exosomes may: protect against cisplatin-induced renal oxidative stress and renal cell apoptosis [74], enhance myocardial viability and prevent adverse remodeling after ischemic injury [75], promote angiogenesis in the setting of myocardial infarction [76,77], protect the intestines from enterocolitis [78], improve hypoxia-induced pulmonary hypertension [79-81], and promote functional recovery after stroke [82,83]. At least in part, such therapeutic effects of exosomes are attributable to their ability to induce angiogenesis as well as regeneration and proliferation of epithelia [84-86]. Furthermore, exosomes exhibit immunomodulatory (largely anti-inflammatory) effects, some of which —specifically down-regulation of interferon-γ secretion and T-cell polarity alteration— are able to stabilize skin graft survival [87,88]. Many studies have found that the efficacy of exosomes nearly matches that of cell-based therapy, indicating that exosomes have potential as next-generation (i.e., cell-free) therapy [76,89-91].
Specifically regarding liver disease, we previously reported that MSCs exhibit remote therapeutic effects [61], a phenomenon attributable to EVs or exosomes. Furthermore, exosomes are reported to ameliorate liver fibrosis in cirrhotic mice [92-97], promote liver regeneration and hepatocyte proliferation (via up-regulation of Bclxl protein) in a murine liver injury model [92], alleviate acute liver failure by dampening macrophage NLRP3 inflammasome activity (additionally suggesting their potential utility during acute liver injury) [94], and improve survival after D-galactosamine- and tumor necrosis factor-α-induced hepatic failure in mice [95]. The latter study also demonstrated that peripherally-administered fluorescently-labeled exosomes were preferentially detectable at high levels in damaged (relative to normal) liver at 6 hours post-administration [95]. Relatedly, it has been shown that intravenously injected exosomes which infiltrate the liver are internalized by hepatic macrophages [98]. Such results suggest that exosomes do not infiltrate the lungs but accumulate in target organs (e.g., sites of liver damage).
CELL-FREE THERAPY IN THE FUTURE: CLINICAL TRIALS INVOLVING EVS AND EXOSOMES
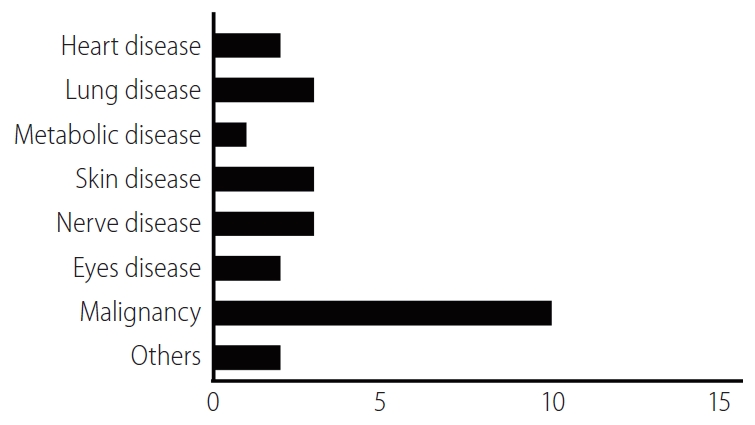
In order to describe relevant recent clinical research trends, we identified EV- or exosome-oriented clinical trials registered on ClinicalTrials.gov. Results included trials focused on both therapeutic applications of exosomes and cancer diagnostic exosome analysis. Excluding observational studies, 26 exosome-oriented therapeutic clinical trials were registered. Of these, 10 focused on cancer diagnostics; three each focused on pulmonary disease, neurological disease, and dermatological disease; two each focused on heart disease, ophthalmological disease, and multiple organ failure, and one focused on metabolic disease (Fig. 4). To the best of our knowledge, no EV- or exosome-oriented clinical trial focused on liver disease was registered at this time. The small number of clinical studies (and the absence of any studies focused on liver disease) is likely attributable to the absence of a simple, standardized method for the production of large quantities of EVs or exosomes; an inadequate understanding of the mechanisms of action of cell-free therapy; and insufficient data regarding the behavior of administered EVs or exosomes in liver disease. However, given the therapeutic potential of EVs and exosomes in liver disease, the number of relevant clinical trials is expected to increase gradually.
CELL-FREE THERAPY IN THE FUTURE: ADVANTAGES AND LIMITATIONS
Given the small diameter of EVs and exosomes, the associated risk of thrombosis after intravenous administration is considered much lower relative to that posed by stem cells (e.g., MSCs). If it is confirmed that cell-free therapeutic effects are dose-dependent, the ability to administer large quantities of EVs or exosomes will be a significant advantage. In addition, the ability of EVs or exosomes to infiltrate damaged target organs directly may result in greater efficacy relative to that of distant stem cells. Some studies have even examined the application of EVs or exosomes to targeted drug delivery [99].
However, even though cell-free therapy overcomes many of the limitations associated with cell-based therapy, the former, too, has its limitations. As mentioned, no standardized method yet exists for the production of large quantities of EVs or exosomes. Furthermore, it is unclear whether using isolated EVs or exosomes excludes an important factor underlying cell-based therapy associated therapeutic effects. Finally, due to the rapid pace of cell-free therapy development, relevant medico-legal norms surrounding cell-free therapy are not yet sufficiently established. Clearly, the field of cell-free therapy is still in its infancy, requiring significant further research.
CONCLUSIONS AND FUTURE PROSPECTS
Cell-based therapy has been developed as an alternative to liver transplantation, but has not yet progressed beyond early-phase clinical trials. Nonetheless, cell-free therapy —which overcomes some of the limitations associated with cell-based therapy— is already under development as a next-generation therapeutic technology, and clinical trials relevant to liver disease are expected to begin in the near future. Cell-free therapy has its own limitations, however. One promising strategy to identify novel and effective curative therapies may be elucidation the mechanisms underlying EV and exosome biological activities, in order to isolate the responsible molecules for direct administration to patients. In conclusion, many challenges remain in developing curative therapies for cirrhosis, but further research into cell-based and cell-free therapies will likely be of significant benefit.
Notes
Authors’ contribution
YW reviewed and interpreted prior literature and case studies/clinical trials. YW, AT, and ST contributed to manuscript preparation and review prior to submission.
Conflicts of Interest: The authors have no conflicts to disclose.
Acknowledgements
This study was supported by the Research Program on Hepatitis from Japan Agency for Medical Research and Development (AMED) (20fk0210070h0001).
Abbreviations
ABMi
autologous bone marrow cell infusion
ACLF
acute-on-chronic liver failure
CSF
colony-stimulating factor
ECM
excessive extracellular matrix
EVs
extracellular vesicles
HGF
hepatocyte growth factor
HSCs
hepatic stellate cells
IL
interleukin
MELD
model for end-stage liver disease
MMPs
matrix metalloproteinases
MSCs
mesenchymal stem cells
OSM
oncostatin M
PGE2
prostaglandin E2
TSG
tumor necrosis factor-stimulated gen