Regulated differentiation of stem cells into an artificial 3D liver as a transplantable source
Article information
Abstract
End-stage liver disease is one of the leading causes of death around the world. Since insufficient sources of transplantable liver and possible immune rejection severely hinder the wide application of conventional liver transplantation therapy, artificial three-dimensional (3D) liver culture and assembly from stem cells have become a new hope for patients with end-stage liver diseases, such as cirrhosis and liver cancer. However, the induced differentiation of single-layer or 3D-structured hepatocytes from stem cells cannot physiologically support essential liver functions due to the lack of formation of blood vessels, immune regulation, storage of vitamins, and other vital hepatic activities. Thus, there is emerging evidence showing that 3D organogenesis of artificial vascularized liver tissue from combined hepatic cell types derived from differentiated stem cells is practical for the treatment of end-stage liver diseases. The optimization of novel biomaterials, such as decellularized matrices and natural macromolecules, also strongly supports the organogenesis of 3D tissue with the desired complex structure. This review summarizes new research updates on novel differentiation protocols of stem cell-derived major hepatic cell types and the application of new supportive biomaterials. Future biological and clinical challenges of this concept are also discussed.
INTRODUCTION
In the clinical context, chronic liver disease is a process in which the liver undergoes progressive destruction and regeneration, which lead to fibrosis and cirrhosis. It is one of the major causes of mortality worldwide. In 2016, chronic liver disease and cirrhosis caused a total of 40,545 deaths, in which alcohol-related liver disease induced nearly half of the deaths [1]. Since damage in the liver often induces inflammation, oxidative stress, cellular necrosis and apoptosis, which causes tissue scarring and fibrosis if occurring repeatedly, developing therapies that can retard or reverse those pathological processes is quite important [2]. When liver disease progresses to the end stage, it becomes irreversible and liver failure is inevitable [3]. At this point, liver transplantation is the only clinical option. However, the lack of transplantable livers and the possibility of immune rejection after the transplant seriously hinders its wide application [4]. In addition, the maximal cold preservation duration of the human liver organ soaked in University of Wisconsin solution before a clinical transplantation operation is only 6–12 hours [5]. Developing novel supercooling techniques to extend this duration to 24–36 hours will dramatically enlarge the donation territory, as well as reduce logistical pressures and facilitate recipient preparation.
To solve the problems hindering successful clinical liver transplants, stem cell medicine provides a brand new solution, particularly those stem cells with the potential to differentiate into liver cells that can then be transplanted directly. For example, a large number of studies have proved that the transplantation of differentiated hepatocytes from human adipose derived stem cells, human amniotic epithelial stem cells, and inducible pluripotent stem cells (iPSCs) effectively ameliorated the complications induced by acute liver failure, including elevated serum aminotransferases, hepatic inflammation, and cell death [6-9]. Stem cell therapy was also reported to be successful in other kinds of chronic/end-stage liver diseases, including nonalcoholic steatohepatitis [10], Wilson’s disease [11], cirrhosis [12], and hepatocellular carcinoma [13]. However, mechanistic studies found that the life support and hepatic protective effects of exogenous stem cells, including mesenchymal stem cells (MSCs) and iPSCs, were primarily from their paracrine actions instead of from direct hepatic occupation [14]. Only hepatic progenitor cells were able to completely and functionally reconstitute a liver that had suffered otherwise lethal damage [15].
Although strategies with differentiated hepatocytes and naïve stem cells demonstrated massive success in the past decades, single cell type-based injection or “in situ transplantation” cannot meet the majority of clinical requirements, which need physiologically functional hepatic tissue to ameliorate damage and support normal liver functions within a short duration. To solve this problem, scientists began to use a complicated but fascinating method – constructing artificial organs from stem cells in three-dimensional (3D) culture. In the current review, we focus on the research updates on liver organogenesis from stem cells, with emphases on the differentiation protocol, biomaterial support, and self-condensation mechanisms.
3D ORGANOGENESIS FROM STEM CELLS
Over the past decade, significant progress has been made in controlling cellular differentiation in stem cell research. For example, it is now possible to force MSCs and iPSCs to differentiate into a large number of specific somatic cell lineages by mimicking the signals presented during embryogenesis. Very importantly, several studies have demonstrated that stem cells have the ability to self-organize into a functional tissue by scattering various somatic cells throughout the tissue [16-19]. For example, Takebe et al. [17] generated a vascularized and functional human liver from human iPSCs by transplanting liver buds created in vitro. Such liver buds were created from a mixture of human iPSCs, human MSCs, and human umbilical vein endothelial cells (HUVECs). The cells then underwent self-organization into a 3D liver bud by recapitulating the organogenetic interactions between the endothelial and mesenchymal cells [19]. The authors tested the hepatic functions of the liver buds both in vitro and in a murine liver failure model, which exhibited satisfactory outcomes. They then expanded this strategy to even more vascularized artificial organs, including kidney, pancreas, intestines, heart, lungs, and brain. It was found that mesenchyme-driven self-condensation on a soft matrix is crucial for organ bud generation [18]. Although those in vitro-grown complex tissue architectures have limited size and survival duration, the ideas raised by this Japanese group extensively inspired other regenerative medicine researchers and clinicians to consider that a mixture of hepatic cell types that are necessary for organ development and self-condensation (e.g., hepatocytes, stellate cells, sinusoidal endothelial cells, and bile duct epithelial cells), together with guides from undifferentiated stem cells, is a promising strategy to generate a clinically transplantable liver or other organ.
DIFFERENTIATION INTO HEPATIC CELL TYPES
Hepatocytes
Hepatocytes are the major cell type of the liver and account for approximately 80% of the hepatic volume and quantity. They are a highly differentiated parenchymal cell type. There are more than 2.5 billion hepatocytes in an adult human liver, and a hepatic lobule is made up of 5 million hepatocytes. Active hepatocyte metabolism can synthesize human essential blood coagulation factors, aliphatic acid, cholesterol, and phospholipids; hepatocytes are also able to store glycogen, proteins, lipids, and vitamins to fulfill the body’s needs.
In the liver, hepatocytes are the very basic cells that support its essential physiological functions. Over the past decade, several studies have introduced protocols to induce hepatocyte-differentiation in stem cells; most methods use chemical and conditioned medium stimulations or coculture with other cell types to expedite hepatic differentiation from human bone marrow mesenchymal stem cells, human umbilical cord mesenchymal stem cells, and human iPSCs [20-23]. Induced hepatocyte-like cells (i-Hep) are shown to possess most of the normal hepatocyte functions (including the secretion of aminotransferases, albumin, and α-fetoprotein), functional biotransformation systems, and the ability to engraft, integrate, and proliferate in an injured liver. Nie et al. [24] used human iPSC to generate a functional liver organoid, which exhibited stronger hepatic functions than human iPSC-derived hepatocyte-like cells (or i-Hep). They found that the functional liver organoid was more susceptible to hepatitis B virus (HBV) infection and could maintain HBV propagation and produce the infectious virus for a prolonged duration, thus causing the hepatic dysfunction of the liver organoid via the down-regulation of hepatic gene expression, induced release of early acute liver failure markers, and altered hepatic ultrastructure. The liver organoid may provide a promising individualized infection model for the development of individualized treatment for hepatitis [24]. Different stem cell types require different concentrations of inducing reagents, including hepatocyte growth factor (HGF), fibroblast growth factor (FGF), oncostatin M (OSM), epidermal growth factor (EGF), and bone morphogenetic protein [25]. Currently, one can even conveniently purchase a commercial one-step direct hepatocyte induction kit.
Although still in the primary stages, a number of studies have tried to induce and culture differentiated hepatocytes in a 3D manner in vitro. The primary interest in developing 3D-structured hepatocytes is because although the 2D in vitro culture of hepatocytes is currently mature, 2D-cultures show a reduction in major liver functions, such as a decreased secretion of albumin and impaired phase I and II enzymatic detoxification abilities [26]. The application of extracellular matrix (ECM) is a major solution for these problems. Thus far, the most common strategy is the sandwich structure in which hepatocytes are placed between two layers of ECM. This model has been proven to provide better hepatocyte cellular functions than 2D monolayer culture conditions since it promotes a polygonal hepatocyte morphology and extended contact surfaces between the cells and the matrix [27].
Hepatic stellate cells (HSCs)
HSCs are the major nonparenchymal cells of the liver. Under physiological conditions, HSCs are in a quiescent state in which their main function is to store and transport vitamin A [28]. When the liver is damaged, under the actions of inflammatory cytokines and stress factors, HSCs change into an activated state (a myofibroblastic phenotype) characterized by increased proliferation, contractility, and chemotaxis. The activation of HSCs will promote the secretion of the ECM involved in liver injury repair. Thus, HSCs play an important role in the occurrence and development of various liver diseases. Moreover, through their interaction with other liver cell types, HSCs are also involved in liver regeneration and differentiation [29].
Research on HSC formation from stem cells is scarce. The main reason for this is that the embryonic origin of HSCs is yet unresolved, with hypotheses of mesenchymal and endodermal origins [30]. Baba et al. [31] proved that in a murine model, HSCs are from the bone marrow since they administered bone marrow cells from green fluorescent protein (GFP) transgenic mice to age-matched mice, and found GFP-positive HSCs in the recipient livers. However, an analytical study of cell lineage demonstrated that HSCs are derived from the mesoderm during liver development; in particular, they are derived from the mesothelium (consisting of mesothelial cells and submesothelial cells) which migrate inward from the liver surface to form HSCs and perivascular mesenchymal cells [32]. Other studies claimed that HSCs are important for the association with hematopoietic sites in the fetal rat liver. HSCs can fulfill some of the functions of MSCs, such as adipocytes or osteocytes differentiation [33,34]. Thus far, only Coll et al. [35] reported that they could obtain HSC-like cells from newborn male fibroblastsderived human iPSC by mimicking embryonic development; those cells closely resemble primary human HSCs at the transcriptional, cellular, and functional levels and possess a gene expression profile between that of quiescent and activated HSCs.
Liver sinusoidal endothelial cells (LSECs)
LSECs consist of the majority of liver nonparenchymal cells (~70% of the total liver nonparenchymal cells). They are functionally unique because of the high activity of receptor-mediated endocytosis, which enables them to clear colloids and soluble waste macromolecules from circulation [36]. LSECs have long been noted to contribute to liver regeneration after liver injury. That is, there is crosstalk between HSCs and LSECs when the activation of HSCs and the formation of fibrosis occur. Healthy LSECs are able to suppress the activation of HSCs and also inactivate already-activated HSCs [37]. Conversely, in the healthy liver, hepatocytes and HSCs can work together to maintain the homeostasis of LSECs through the release of vascular endothelial growth factor (VEGF) [38]. When the Kupffer cells (KCs)’ phagocytic function is damaged, LSECs will remove materials with diameters greater than 1 μm. Furthermore, LSECs take in viruses and submit antigens to T cells. Additionally, they participate in the local immune regulation in the liver and may also play an important role in immune tolerance [39].
Reports of direct differentiation from stem cells to LSECs are extremely limited. Du et al. [40] demonstrated a novel two-step method for that process, that is, a 7-day first stage from human iPSCs that are generated by reprogramming human foreskin fibroblasts to the mesodermal lineage and a 7-day second stage from mesodermal cells to hepatic endothelial cells. The final LSEC-like cells are positive for expressions of endothelial-specific markers such as CD-31 and VE-cadherin [40]. Vunjak-Novakovic et al. [41] claimed that they established a culture condition to promote the differentiation from multiple iPSC cell lines to endothelial cells that coexpress cell surface markers and functional properties (e.g., endothelial nitric oxide synthase production) with an efficiency of approximately 10–40%. In addition, Narmada et al. [42] declared that they had formed endothelial cells from human iPSC that had been derived from IMR90 fibroblasts and BJ fibroblasts by using a chemically defined hepatocyte growth medium (HGM); those cells expressed PECAM-1 and CDH5, could form spontaneous tube structures that represent mature endothelial markers, possessed endothelial nitric oxide synthase, and were capable of taking up acetylated low-density lipoprotein.
Bile duct epithelial cells (BDECs)
BDECs are a kind of distinctive epithelial cell coating on the liver inner tube surface. They are of great importance in bile duct contraction, bile secretion, and liver water electrolyte transport. In addition, BDECs can also mediate the synthesis and secretion of IgA/IgM, cytokines, chemokines, and adhesion molecules involved in immunity, cell interaction, and signal transduction [43].
A study described a differentiation procedure from embryonic stem cells (ESCs) to BDECs on the basis of induced hepatocytes. The authors coincubated the ESCs with HGF/scatter factor, EGF, and transforming growth factor α (TGF-α) in the presence of a new HGM. In addition to the expected morphology, the differentiated BDECs also expressed cytokeratin 19, acidic FGF, and TGF-α, but did not express the hepatocyte markers including albumin and cytochrome P450 IIB1 [44]. Another study confirmed that dexamethasone and transforming growth factor β (TGF-β) contribute to differentiation into BDECs; these compounds are closely connected with the Wnt/β-catenin associated protein, and the Notch and TGF-β signaling transduction pathways [45]
Sampaziotis et al. [46,47] from the University of Cambridge reported a novel and serum-free method for the direct differentiation of cholangiocytes from human skin fibroblasts and peripheral blood derived iPSCs. By using several combinations of recombinant growth factors and inhibitors at different stages, they formed definitive endoderms, foregut progenitors, hepatoblasts, and cholangiocyte progenitors from iPSCs in sequence. After a 26-day differentiation process, the final cholangiocyte-like cells were capable of showing the functional characteristics of normal cholangiocytes, including alkaline phosphatase/γ-glutamy-ltranspeptidase activity, transfer of bile acids, and responses to secretin, somatostatin, and VEGF. The derived cells were also useful in establishing some common biliary disease models (e.g., Alagille syndrome, polycystic liver disease, and cystic fibrosis-associated cholangiopathy) [46,47].
Kupffer cells (KCs)
KCs are hepatic local macrophages which are located on the inner surface of the sinus hepaticus. They are a critical component of the mononuclear phagocytic system, which takes part in the response to pathogens. KCs can remove foreign antigens, antigen-antibody complexes, cell debris, and other substances in the liver and circulation partly through the activation of nuclear factor kappaB and the secretion of a variety of cytokines [48]. The abnormal activation and regulation of KCs contribute to nearly all types of liver diseases, such as acute liver failure, nonalcoholic fatty liver disease, alcohol-related liver disease, genetic liver diseases, cirrhosis, and liver cancer [49].
Although a number of studies have successfully established methods to generate macrophage or dendritic cell from human PSCs [50,51], no report described the differentiation protocol from stem cells to KCs until 2018. Part of the reason is that the derived macrophage is able to migrate into the liver to form KCs and pit cells (intrahepatic lymphocytes). In addition, in the process of establishing an artificial 3D liver, KCs and pit cells are not necessary because 1) they are expected to migrate into the engineered liver grafts from the bone marrow and the circulatory system of the recipients upon transplantation [52], 2) the functions of the assembled LSECs are similar to those of KCs [53], and 3) KCs may secrete nitric oxide to increase their own apoptosis, leading to acute rejection after liver transplantation. Additionally, KCs will induce apoptosis and differentiation of T-cells through the Fas-FasL apoptosis pathway [54]. Thus far, only Tasnim et al. [55] demonstrated that they could obtain mature KCs from human IMR90 fibroblasts derived iPSCs (iKCs). By using a different culture medium and several combinations of recombinant growth factors and inhibitors at different stages, they generated KCs that expressed the macrophage markers CD11, CD14, CD68, CD163, CD32 at 0.3–5 folds of those from primary adult human KCs (pKCs) and KC-specific CLEC-4F, ID1, and ID3. Moreover, iKCs phagocytosed and secreted IL-6 and TNF-α upon stimulation at levels similar to pKCs but different from nonliver macrophages [55]. Although not necessary for 3D liver assembly, KCs should be included in the 3D liver for disease modeling and drug discovery in vitro. The reported differentiation protocols of the above-mentioned hepatic cell types are listed in Table 1. Key markers for differentiated liver cell types were summarized in Figure 1.
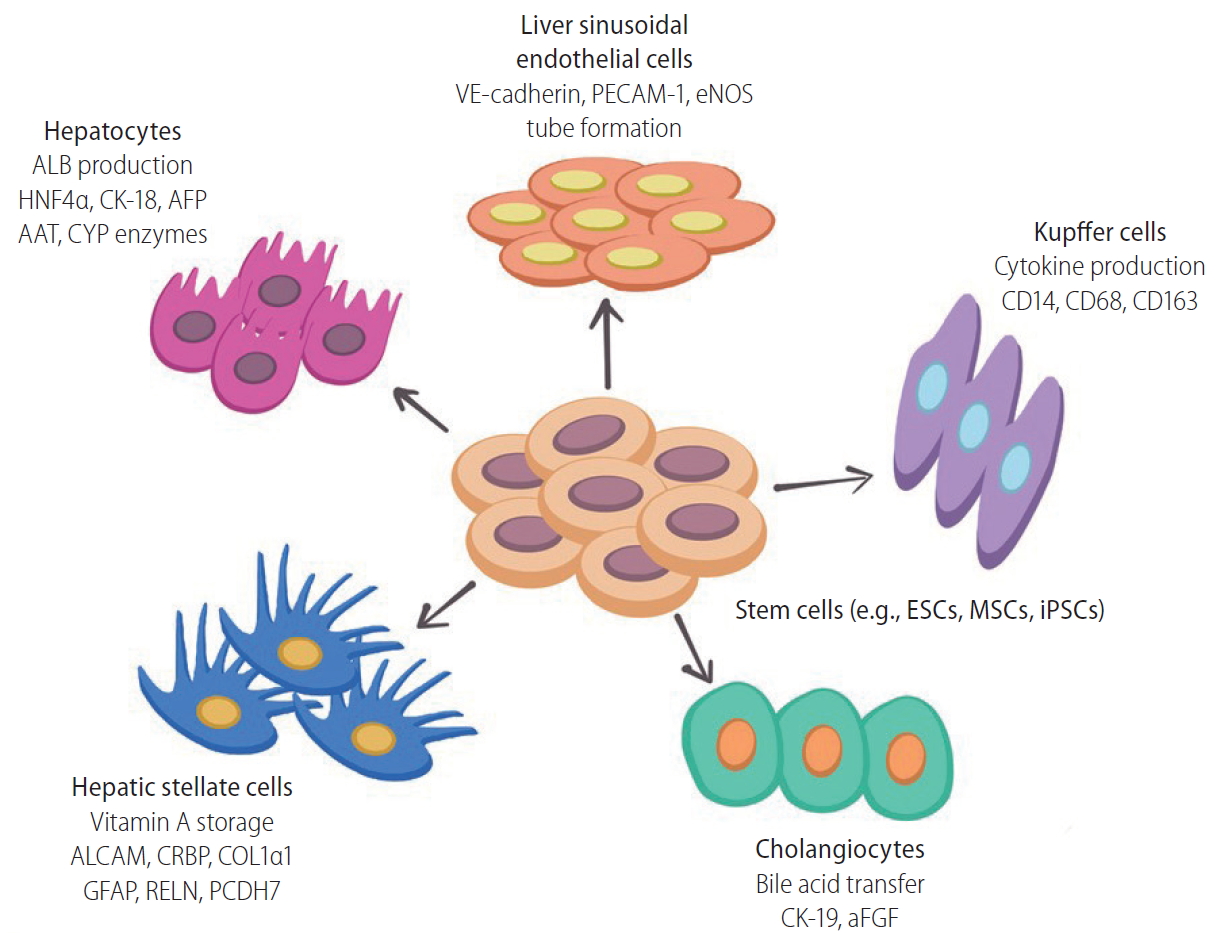
Key functional markers of liver cell types differentiated from stem cells (e.g., embryonic stem cells, mesenchymal stem cells, and inducible pluripotent stem cells). Major liver cell types, including hepatocytes, Kupffer cells, hepatic stellate cells, liver sinusoidal endothelial cells, and cholangiocytes can be reprogrammed from various stem cells using defined medium and chemical compounds during in vitro culture. Key functional markers can be used to validate the successful reprogramming of each cell type. ALB, albumin; HNF4α, hepatocyte nuclear factor 4 α; CK, cytokeratin; AFP, α-fetoprotein; AAT, α-1 antitrypsin; CYP, cytochrome P family enzymes; PECAM, platelet endothelial cell adhesion molecule-1; eNOS, endothelial nitric oxide synthase; ESCs, embryonic stem cells; MSCs, mesenchymal stem cells; iPSCs, inducible pluripotent stem cells; ALCAM, activated leukocyte cell adhesion molecule; CRBP, cellular retinol-binding protein; COL1α1, collagen type 1 α 1; GFAP, glial fibrillary acidic protein; RELN, reelin; PCDH7, protocadherin-7; aFGF, acidic fibroblast growth factor.
ASSEMBLY OF TRANSPLANTABLE 3D LIVER TISSUE
In previous decades, numerous studies have focused on the construction of liver tissue in a 2D planar environment. However, such a strategy is challenging because the actual liver tissue is 3D. Therefore, to address the limitations of 2D cell cultures and achieve the functional recovery of the liver, a 3D microenvironment which aims to mimic the native environment must be fabricated [56]. Recently, scientists focused on exploring artificial 3D ECM by fabricating a scaffold using particular materials or a matrix, including a decellularized matrix, natural macromolecules, and synthetic biodegradable polymers (listed in Table 2) [57]. The engineered scaffolds can provide a 3D environment for cell attachment and proliferation. Moreover, the scaffolds play a role in regulating cell maturation and function by modification with specific physical and biochemical properties [58,59]. Recently, by transdifferentiating hepatocytes, HSCs, and LSECs from human umbilical cord blood stem cells simultaneously, we obtained a self-assembled functional 3D human liver bud in vitro and transplanted it into a murine model of acute liver failure. The liver bud underwent maturation during injury alleviation, after which the cells exhibited a gene expression profile signature similar to that of adult human livers (Fig. 2) [60].
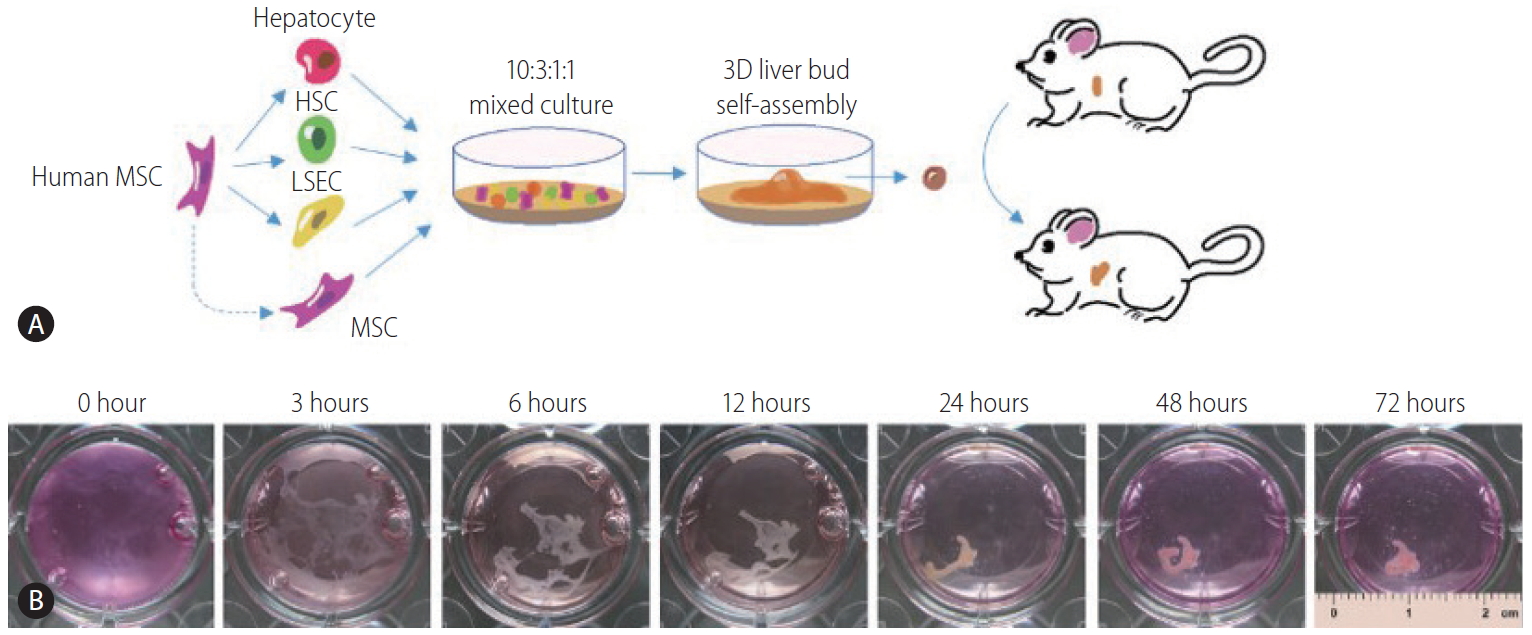
Human 3D liver bud assembly from human umbilical cord blood stem cells (hUCBSCs). (A) Schematic representation of our strategy to assemble human 3D liver buds from naive MSC and MSC-derived hepatocytes, HSC-like cells, and LSEC-like cells. (B) The time-lapse representative images of the self-assembly process. This data and description have been published by Li et al. [60] MSC, mesenchymal stem cell; HSC, hepatic stellate cell; LSEC, liver sinusoidal endothelial cell.
Decellularized matrix
A decellularized matrix scaffold, which is procured by a decellularization technique that removes parenchymal cells from tissues or organs, can potentially retain the skeletal architecture and functional characteristics of the original tissue [61,62]. Decellularized scaffolds derived from animal organs are now being explored as a promising resource for generating transplantable and functional organs. Lin et al. [63] documented that a rat primary hepatocyte culture in a porcine liver-derived biomatrix for up to 45 days showed better liver-specific functions, such as albumin synthesis, urea production, and P450 1A1 activity, than those on adsorbed collagen cultures. Uygun et al. [64] seeded hepatocytes in a decellularized whole liver tissue. In vitro, the recellularized graft supported liver-specific functions including albumin secretion, urea synthesis and cytochrome P450 expression at levels comparable to a normal liver. When transplanted into rats, the recellularized liver grafts could support hepatocyte survival and function with minimal ischemic damage [64]. Park et al. [65] reported that they reseeded hepatocytes derived from iPSCs that had been generated by porcine ear fibroblasts into decellularized liver scaffolds, using a continuous perfusion system to culture the recellularized liver. When the grafts were transplanted into rats for the short-term, they found that the grafts expressed hepatocyte markers and did not rupture [65]. Furthermore, researchers have found that hepatocyte-like cells derived from human amniotic epithelial, bone marrow, and adipose stromal cells display enhanced functionality when cultured on a decellularized liver substrate [66]. To overcome the limitations due to the shortage of donor organs, Xiang et al. [67] utilized the spleen as the source of a decellularized scaffold for liver regeneration. The decellularized spleen scaffold could support the location and survival of BMSCs within the 3D matrix. Moreover, the subcutaneous implantation of the decellularized scaffold presented good histocompatibility [67]. Another study also proved that a bioartificial liver constructed by a heparin-coated decellularized spleen matrix can exert satisfactory short-term anticoagulant effects and can compensate for a certain degree of liver function [68].
However, the strategy based on a decellularized matrix suffers from limitations including a severe shortage of donor organs, high cost of treatment, and lifetime immune suppression [69].
Natural macromolecules
Natural macromolecules that are derived from ECM have been widely investigated as tissue engineering scaffolds for the culture of liver cells, including proteins such as collagen and gelatin as well as polysaccharides such as chitosan and alginate. These macromolecules have shown good biocompatibility both in vitro and in vivo.
Collagen, especially type I collagen, is the main structural protein in the extracellular space in various connective tissues in animal bodies. The collagen matrix serves to mediate cell growth, differentiation, and survival, and tissue organization. A collagen gel could provide a 3D microenvironment for hepatocytes to sustain high liver-specific functions [70]. Khodabandeh et al. [71] cultured human Wharton’s jelly MSCs in 2D collagen films and 3D collagen scaffolds for 21 days. The cells cultured in the 3D collagen scaffolds showed a significant increase in claudin expression compared to a conventional monolayer culture and the 2D collagen scaffold. Their subsequent study showed that the expression of the hepatocyte markers were increased significantly when administering the liver extract from a 3D matrigel/collagen culture of MSCs [72]. Nakai et al. [73] tested the effect of culturing human iPSCs-derived endoderm cells on a collagen vitrigel membrane; they found collagen vitrigel had promoted the differentiation of cells into functional in vitro cells, as shown by the decrease of immature marker alpha-fetoprotein (AFP) levels and the concomitant increase in expression of mature hepatocyte transcription factors (such as ALB and ASGR1 ) and markers involved in liver functions (such as transporters, cytochrome P450 enzymes, and phase II metabolic enzymes). Moreover, collagen gel could also be used in a hepatocyte entrapment culture as an effective model to interpret drug hepatotoxicity and drug metabolism in vivo [74].
Gelatin is the partially hydrolyzed form of collagen with a chemical composition that is, in many respects, very similar to that of its parent collagen. Recently, gelatin-based scaffolds have been documented to increase cell attachment and differentiation. Vasanthan et al. [75] developed galactose-containing physical cross-linked polyvinyl alcohol/gelatin hydrogel scaffolds via the freeze/thaw technique. Such galactose-containing hydrogels promoted cell-cell and cell-hydrogel interactions, aiding cellular aggregation and leading to the formation of spheroids [75]. Another research found that a gelatin cryogel scaffold successfully promoted the differentiation of human adipose-derived MSCs into hepatocyte-like cells with a higher expression of hepatocyte-specific markers and appropriate functional characteristics, such as increased levels of urea biosynthesis and glycogen storage [76]. On this basis, the researchers optimized the aforementioned scaffold and improved it through laminin incorporation; they found that the cells generated on this scaffold were strikingly similar to a standard hepatocyte cell line in terms of the expression of hepatocyte markers [77]. Moreover, gelatin was also employed to fabricate microcarriers with chitosan for hepatocyte culture [78], and improved the performance of hepatocytes for as long as 2 months [79].
Chitosan is a linear polysaccharide which has good biocompatibility and wide applications in biomedical fields. In the study of Verma et al. [80], HepG2 cells cultured on chitosan cast films formed 3D spheroids and exhibited higher amounts of album and urea synthesis compared to a monolayer culture. Rat hepatocytes seeded on an electrospun scaffold of galactosylated chitosan formed immobile, 3D, flat aggregates, and exhibited superior cell bioactivity with high levels of liver-specific albumin secretion, urea synthesis, and cytochrome P450 enzyme expression [81]. Rajendran et al. [82] established a chitosan nanofiber scaffold via an electrospinning technique combined with a coculture system that consisted of hepatocytes and fibroblasts, and thus developed a new 3D liver model for the maintenance of long-term liver functions. To obtain better biocompatibility, chitosan can be further modified with other macromolecules, such as collagen [83], gelatin [78], alginate [84], and heparin [85]. For example, Fan et al. [86] proved that chitosan/galactosylated hyaluronic acid scaffolds were suitable for improving hepatocytes adhesion and maintaining liver function in a previous work. To adjust the interaction of hepatocytes with the scaffold, they further modified the materials by the incorporation of heparin, which induced the formation of cellular aggregates with enhanced liver-specific metabolic activities and increased cell density [85].
Alginate is a widely employed biomaterial in different areas of drug delivery and tissue engineering because of its good biocompatibility and optimal chemical properties. Its gelation with calcium ions provides calcium-alginate scaffolds with mechanical stability and relative permeability. Hepatocytes could organize in aggregates within calcium-alginate scaffolds, which established real 3D hepatocyte architecture with cell polarity, cell junctions, and microvillus-lined channels with abundant bile canaliculi [87]. Lin et al. [88] demonstrated that bone marrow-derived MSCs cultured in alginate scaffolds for several days displayed several liver-specific markers, such as the expression of genes encoding alpha-fetoprotein, ALB, connexin 32, CYP7A1, and were capable of glycogen storage. To fabricate implantable constructs for liver regeneration, Lau et al [89]. encapsulated murine embryoid bodies (EBs) into a 3D alginate microcavitary hydrogel system for both EB formation and subsequent hepatic lineage differentiation. Urea and albumin production were found to be significantly higher compared to a monolayer culture, demonstrating the beneficial effects of the 3D engineered environment [89]. Shteyer et al. [90] reported alginate scaffolds significantly increased animal survival, prolonged alanine aminotransferase and aspartate transaminase serum levels, and were accompanied by improved liver histology after an extended partial hepatectomy. Another study on acute liver failure in rats showed that umbilical cord stem cell-derived in vitro cells encapsulated in alginate scaffolds effectively attenuated biochemical tests, improved liver cytoarchitecture, increased the expression of albumin, and reduced the level of AFP [91]. Additionally, a new study from Shao et al. [92] constructed carbon nanotube nanocomposites via the layer-by-layer assembly of negatively charged multiwalled carbon nanotubes and positively charged poly (dimethydiallylammonium chloride), and proved that they could provide a potent regulatory signal over neural stem cells.
However, natural macromolecules suffer from a limited range of properties due to difficult processing of bulk material and the possibility of inducing potentially dangerous immune responses when used as xenografts or allografts [93]. Moreover, the extraction and purification of natural macromolecules also leads to significant batch-to-batch variations [94], which can lead to unpredictable cell behavior and consequently impact the performance of cell-based tissue-engineered devices.
SYNTHETIC POLYMERS
Biodegradable synthetic polymers have been attractive candidates for hepatocyte culture in vitro and for making implantable scaffolds in vivo [95]. A major advantage of synthetic polymers over natural macromolecules is the easy control of their physiochemical and biological modifications. Some of the widely used synthetic polymers are polylactic acid, poly(lactide-co-glycolide) (PLGA), and poly(ethyleneglycol) (PEG).
The study of Hanada et al. [96] indicated that a 3D culture using poly-L-lactic acid (PLLA) scaffolds with OSM remarkably enhanced the albumin production and cytochrome P450 1A1/2 capacity during the culture time. This observation was in agreement with those of Jiang et al. [97] and Wang et al. [98], where fetal mouse liver cells were cultured on 3D PLLA scaffolds in the presence of nicotinamide, dimethyl sulfoxide, and OSM. In addition, the in vivo implantation of such engineered liver tissue showed a remarkably higher presence of albumin-positive engrafted cells 15 days after the operation when compared to freshly isolated and cultured cells for 1 day [96]. However, researchers are less likely to use pure PLLA because of poor biocompatibility, and because scaffolds fabricated from single polymers display poor mechanical properties and are not easy to handle. To overcome these limitations, many reports have indicated the possible application of PLLA/poly(£-caprolactone) blends [99,100].
Ranucci and Moghe [101] cultured hepatocytes on porous foams of amorphous PLGA with a wide range of controlled pore-size distributions (approximately 1 to 100 microns) and found that foams with supercellular size voids (~67 microns) promoted the kinetics of 3D aggregation with the most rapid and sustained albumin secretory kinetics. Kasuya et al. [102] developed a 3D stacked culture method using biodegradable PLGA membranes. The hepatocyte layers on the PLGA membranes would reorganize into a 3D stacked structure after PLGA degradation. In the study by Liu et al. [103], ESCs were mixed with Matrigel and immediately seeded in a PLGA scaffold with the appropriate inducing factors. The system enabled the spontaneous formation of spheroids, which differentiated into in vitro cells as evidenced by the expression of liver-specific markers and proteins [103].
However, synthetic biodegradable polymers have poor characteristics for cell culture due to their hydrophobic characteristics and lack of bioactive cues. Therefore, their bioactivities should be improved by chemically grafting bioactive cues [104] and/or modifying the surface with natural macromolecules [105]. Kojima et al. [106] developed avidin-biotin binding systems (ABBS) for the initial attachment of biotinylated HepG2 cells to avidin-adsorbed PLLA surfaces. ABBS assisted HepG2 cells to adhere to a PLLA surface, and the proliferation of these attached cells was comparable with those cultured on collagen. Moreover, the hepatic functions of the attached cells were not significantly changed [106]. Brown et al. [107] modified a nanofibrous PLGA electrospun scaffold with a type I collagen coating. The modified scaffold led to 10-fold greater albumin secretion, 4-fold higher urea synthesis, and elevated transcription of hepatocyte-specific CYP450 genes in primary human hepatocytes compared to the unmodified PLGA scaffolds [107]. Similarly, Bierwolf et al. [108] developed a collagen-coated PLLA electrospun nanofibrous scaffold which provide a good in vitro microenvironment for new tissue regeneration of primary rat hepatocytes. Application of novel materials for liver matrix scaffold construction and organogenesis were illustrated in Figure 3.

Assembly of transplantable 3D liver tissue. Natural macromolecules and synthetic polymers can be used to construct a decellularized matrix scaffold. Differentiated liver cells (hepatocytes, Kupffer cells, hepatic stellate cells, liver sinusoidal endothelial cells, and cholangiocytes) are then mixed and transplanted into the matrix scaffold to allow organogenesis for possible transplantation. PLA, polylactic acid; PLGA, poly(lactide-co-glycolide); PEG, poly(ethyleneglycol).
CONCLUDING REMARKS
Establishing a transplantable artificial 3D liver is complicated. Numerous studies have proved that a single hepatocyte component may not be able to fulfill the physiologically required liver functions. Thus, a combination of hepatocytes, HSCs, BDECs, and LSECs is proposed to form vascularized liver tissues providing blood circulation and nutrient supply. Existing researches have shown several ways to force stem cells to differentiate into those kinds of cells, with the proper physical supports from novel biomaterials. Indeed, there are a number of problems that need to be overcome, such as 1) ensuring that the differentiation process occurs properly and completely, 2) constructing 3D biomaterials with proper hepatic structures and with controllable release of growth factors for the self-formation of the liver tissue, and 3) avoiding possible tumorigenicity. Addressing these concerns will definitely promote the basic and clinical development of novel strategies for treating various liver diseases.
Acknowledgements
This work was supported by the Major Science and Technology Projects of Guangdong Province (No. 2015B020225005) and the National Natural Science Foundation of China (No. 81970515, 81873573, 81800525).
Notes
Authors’ contribution
All of the authors were responsible for the interpretation of data and drafting and critically revising the manuscript for important intellectual content.
Conflicts of Interest: The authors have no conflicts to disclose.
Abbreviations
3D
three-dimensional
ABBS
avidin-biotin binding systems
BDEC
bile duct epithelial cells
EBs
embryoid bodies
ECM
extracellular matrix
EGF
epidermal growth factor
ESCs
embryonic stem cells
FGFs
fibroblast growth factors
GFP
green fluorescent protein
HBV
hepatitis B virus
HGF
hepatocyte growth factor
HGM
hepatocyte growth medium
HSCs
hepatic stellate cells
hUCBSCs
human umbilical cord blood stem cells
HUVECs
human umbilical vein endothelial cells
i-Hep
induced in vitro cells
iKCs
induced Kupffer cells
iPSCs
inducible pluripotent stem cells
KCs
Kupffer cells
LSECs
liver sinusoidal endothelial cells
MSCs
mesenchymal stem cells
OSM
oncostatin M
PEG
poly(ethyleneglycol)
pKCs
primary adult human KCs
PLGA
poly(lactide-co-glycolide)
PLLA
poly-L-lactic acid
TGF-α
transforming growth factor α
TGF-β
transforming growth factor β
VEGF
vascular endothelial growth factor