| Clin Mol Hepatol > Volume 18(4); 2012 > Article |
ABSTRACT
Background/Aims
The early diagnosis of acute hepatitis A (AHA) is hindered because serum IgM against hepatitis A virus (HAV) can yield false-negative results during the window period. This study evaluated the diagnostic accuracy of a polymerase chain reaction (PCR) kit for HAV RNA for the diagnosis of AHA.
Methods
Samples were collected from 136 patients with acute severe hepatitis at their admission to Asan Medical Center between June 2010 and July 2010. Samples were analyzed for serum IgM anti-HAV using an immunoassay test and for qualitative HAV RNA using the Magicplex HepaTrio PCR test kit. The diagnostic accuracies of these methods were tested on the basis of clinical and laboratory diagnoses of AHA.
Results
The concordance rate and kappa value between IgM anti-HAV and HAV RNA PCR were 88.2% and 0.707, respectively. For the diagnosis of AHA, the sensitivity and specificity of IgM anti-HAV were 90.7% and 100%, respectively, when an "equivocal" result was regarded as positive; and 79.1% and 100%, respectively, when an "equivocal" result was regarded as negative. The sensitivity and specificity of HAV RNA PCR were 81.4% and 100%, respectively. All four patients with negative IgM anti-HAV and positive HAV RNA PCR results and all four patients with equivocal IgM anti-HAV RNA and positive HAV RNA PCR results were eventually diagnosed with AHA.
Early etiologic diagnosis of acute hepatitis is urgent, especially when the severe necroinflammation proceeds, because the etiology is most important predictive factor of clinical course in acute liver failure.1 As a major cause of acute liver failure, infection by hepatitis A virus (HAV) has a better prognosis than infection by hepatitis B virus (HBV) or toxin.2 In rapidly developing countries, including Korea, the incidence of symptomatic HAV infection in adults is increasing, due to the shifting seroprevalence of HAV with improvement in socioeconomic conditions.3
Since early epidemiologic studies characterised hepatitis A as an infectious hepatitis that is transmitted via a faecal-to-oral route, several diagnostic tests for HAV have been developed, including serologic and molecular approaches. Although immunoglobulin M (IgM) anti-HAV is used as the primary marker of acute hepatitis A (AHA) infection, the serologic test based on this marker is associated with a false-negative rate of 4-13% during the window period.4,5 On the other hand, serum HAV ribonucleic acid (RNA) is generally detected before IgM anti-HAV.6 This finding suggests that HAV RNA reverse transcriptase-polymerase chain reaction (RT-PCR) may be more sensitive than serologic tests based on IgM anti-HAV during the early phase of HAV infection, especially during the window period before antibody to HAV can be detected.
The purpose of this study was to evaluate the diagnostic accuracy of a polymerase chain reaction kit for HAV RNA for the diagnosis of AHA infection.
This prospective study enrolled consecutive patients who underwent IgM anti-HAV and HAV RNA PCR sampling to evaluate acute hepatitis at Asan Medical Center from June 1, 2010 to July 30, 2010. Of the 729 subjects who were sampled for IgM anti-HAV, 136 patients showed an alanine aminotransferase (ALT) level >300 IU/L during the disease period or peak ALT >100 IU/L in the case suspecied acute HAV infection. These patients were selected as severe acute hepatitis patients. When the initial anti-HAV was negative, the subsequent IgM anti-HAV was performed at 3-7 day after initial serologic test to detect the case of AHA in which initial negative IgM anti-HAV was shown during window period. The disease phase of all patients was assessed on the basis of their medical records (Fig. 1).
AHA was defined when the peak ALT level of the disease period was >300 IU/L, the first or subsequent serologic test provided a positive IgM anti-HAV result, and other causes of acute hepatitis could be excluded (e.g., HBV, hepatitis C virus [HCV], drug, toxin, autoimmune hepatitis, and Wilson disease).
Overt phase of AHA was considered when the ALT level was above the upper-normal range, the initial IgM anti-HAV result was positive, and the duration from clinical onset to initial IgM anti-HAV sampling was equal to or shorter than the duration from clinical onset to peak ALT.
Recovery phase of AHA was considered when the initial IgM anti-HAV result was positive, and the duration from clinical onset to initial IgM anti-HAV sampling was longer than the duration from clinical onset to peak ALT.
Relapse phase of AHA was considered when recent recovery from AHA infection was confirmed, and the ALT level was >100 IU/L without other known cause.
Early phase of AHA was considered when the initial IgM anti-HAV result was negative or equivocal, but positive on subsequent serologic testing, and the duration from clinical onset to initial sampling was equal to or shorter than the duration from clinical onset to peak ALT.
This study was approved by the Institutional Review Board of Asan Medical Center.
The IgM anti-HAV result was measured with a commercially available chemiluminescent microparticle immunoassay (Abbott Laboratories, Abbott Park, IL, USA) according to the manufacturer's instructions. The results were expressed based on the S/CO ratio: >1.2 was reported as positive, 0.8-1.2 as equivocal, and <0.8 as negative.
The serum at the time of sampling for initial IgM anti-HAV was used to detect HAV RNA. As advised by the manufacturer, nucleic acid was extracted from serum with the SEEPREP12 (Cat. No. SPN1200) and SEEPREP12 Viral NA kits (Cat. No. SPN1004, Seegene, Seoul, Korea). Briefly, 10 ┬ĄL of proteinase K (20 mg/mL, Sigma) were added to 240 ┬ĄL of serum. After lysis, nucleic acid was bound to beads, washed and automatically eluted into 60 ┬ĄL elution buffer. To prevent contamination, RNA extraction and amplification were performed in separate rooms.
The RT-PCR process was carried out with Magicplex HepaTrio Amplification (Cat. No. HT8000X) and Magicplex HepaTrio Real-time Detection kits (Cat. No. HT8301X, Seegene). This procedure was developed as a qualitative test to detect HAV, HBV, and HCV simultaneously.
For amplification, 36 ┬ĄL of extracted nucleic acid were mixed with PCR Master Mix (5 ┬ĄL of 10├Ś HepaTrio RM, 5 ┬ĄL of 10├Ś Onestep RT-PCR buffer, and 4 ┬ĄL of Onestep RT-PCR Enzyme Mix). The first round of amplification was performed with a GeneAmp PCR System 9700 (Applied Biosystems, Life Technologies, Carlsbad, CA, USA) with the following protocol: 50Ōäā for 20 min, 94Ōäā for 15 min, 10 cycles of (94Ōäā for 0.5 min, 55Ōäā for 1.5 min, and 72Ōäā for 1.0 min), 35 cycles of (94Ōäā for 30 sec, 55Ōäā for 30 sec, and 72Ōäā for 30 sec), and 72Ōäā for 2 min. Then, 2 ┬ĄL of the product from the first round of PCR were used as the template for the second round of PCR. The template was mixed with PCR Master Mix (4 ┬ĄL of 5├Ś HepaTrio DOM, 4 ┬ĄL of RNase-free water, and 10 ┬ĄL of 2├Ś Detection mix). Finally, RT-PCR was performed with a CFX96 RT-PCR System (Bio-Rad, Hercules, USA) at 95Ōäā for 2 min and 20 cycles of 95Ōäā for 20 sec and 55Ōäā for 40 sec. The HAV RNA result was interpreted as positive when the Ct value was Ōēż20 and as negative when the Ct value was not available.
Between-group comparisons were performed with independent t-test for continuous variables and the chi-square test or Fisher's exact test for categorised variables, as appropriate. For concordance analysis between the IgM anti-HAV and HAV RNA RT-PCR, the concordance rate and weighted overall kappa value were calculated using 3├Ś3 crosstable. Statistical analyses were performed with SPSS, version 19.0 (SPSS, Chicago, IL, USA). Statistical hypothesis tests with P<0.05 were considered significant.
Among the total of 136 patients, 43 patients (31.6%) were identified as having AHA (HAV group). Patients in the HAV group were younger than those in the non-HAV group (33.2┬▒8.30 years vs. 48.6┬▒19.9 years; P<0.001). The ALT level at the time of initial IgM anti-HAV sampling (2389┬▒2201 IU/L vs. 667┬▒696 IU/L; P<0.001) and peak ALT level during the disease period (3135┬▒2255 IU/L vs. 963┬▒790 IU/L); P<0.001) in the HAV group were significantly higher than those in the non-HAV group (Table 1).
Of the 38 patients with acute hepatitis A excluding 5 equivocal results in initial IgM anti-HAV, 34 patients (89.5%) showed positive IgM anti-HAV in initial IgM anti-HAV test. All the parameters were not statistically significant difference between initial positive and negative IgM anti-HAV group (Table 2).
When the patients with AHA were classified according to the definition of disease phase, the number of the patient was 8 in early phase, 18 in overt phase, 14 in recovery phase, and 2 in relapse phase, respectively. One patient could not be allocated because her clinical onset data was not available.
The concordance rate and kappa value between IgM anti-HAV and HAV PCR were 88.2% and 0.707, respectively (Table 3).
If an "equivocal" result IgM anti-HAV was regarded as positive, then the sensitivity and specificity of IgM anti-HAV were 90.7% and 100%, respectively, and the positive predictive value (PPV) and negative predictive value (NPV) were 100% and 95.9%, respectively. If an "equivocal" IgM anti-HAV result was regarded as negative, then the sensitivity and specificity of IgM anti-HAV were 79.1% and 100%, respectively, and the PPV and NPV were 100% and 91.1%, respectively (Table 4). The sensitivity and specificity of HAV RNA were 81.4% and 100%, respectively, and the PPV and NPV were 100% and 92.1%, respectively (Table 5). When the inclusion criteria restricted as more severe acute hepatitis with peak ALT >1,000 IU/L, The sensitivity and specificity of HAV RNA were 80.0% and 100%, respectively, and the PPV and NPV were 100% and 83.3%, respectively.
All four patients with negative anti-HAV IgM and positive HAV RNA results and all four patients with equivocal anti-HAV IgM and positive HAV RNA results were identified as being in the early phase of AHA. Of the seven patients with positive anti-HAV IgM and negative HAV RNA results, four patients were in the recovery phase, one patient was in the overt phase, one patient was in the relapse phase of AHA and one patient was diagnosed with acute calculous cholecystitis. One patient with equivocal anti-HAV IgM and negative HAV RNA results was in the relapse phase of AHA.
All eight of the negative or equivocal HAV IgM results and positive HAV RNA results in confirmed AHA cases were distributed such that the initial sampling was performed within 6 days from the clinical onset of symptoms. As the time from clinical onset increased, the number of negative HAV RNA results increased (Fig. 2).
Previous studies have defined severe acute hepatitis by an ALT level >5-10 times the upper limit of the normal range.7-9 This term is important because patients with severe acute hepatitis should be carefully evaluated to identify whether the cause is treatable and to determine whether the clinical course will proceed to acute liver failure.
In this study, 30.9% of 136 patients with severe acute hepatitis were confirmed as having HAV infection on the basis of serologic testing. Patients in the HAV group were significantly younger and had higher initial and peak ALT levels than those in the non-HAV group. These results are relevant to the trend of increasing incidence of AHA in Korean adults. Kim et al reported that the age-specific seroprevalence of protective IgG anti-HAV dramatically decreased among all age groups from 1982 to 2006. Consequently, several outbreaks of HAV have occurred in Korean young adults during the past two decades.3 In countries (such as Korea) where sanitation conditions have recently improved with increased socioeconomic development, it is necessary to ascertain whether HAV is the cause of severe acute hepatitis.
Initial negative IgM anti-HAV in acute onset of AHA means that this is the early stage of clinical course. The study by Hyun et al10 showed the interval from peak-ALT day to the first HAV-test day was associated with the initial result of IgM anti-HAV. The patients with negative initial IgM anti-HAV showed the negative value of the interval from peak ALT to the first HAV test (mean┬▒SD, -1.5┬▒1.5 days).10 This trend also was shown in our data (mean┬▒SD, -1.3┬▒1.3 days), and this suggests that IgM anti-HAV has limitation to early diagnosis of AHA during window period.
In this study, the detection of HAV RNA through RT-PCR was concordant with detection of IgM anti-HAV, with a good kappa value (0.707). When an equivocal result of anti-HAV IgM was regarded as negative, the sensitivity and specificity of HAV RNA were similarly high between the serologic test and HAV RNA detection by RT-PCR. Therefore, the molecular approach of HAV RNA detection was comparable to the serologic test of anti-HAV IgM HAV.
When we analysed the discordant sample cases between the two tests, all eight patients with negative or equivocal anti-HAV IgM and positive HAV RNA results were confirmed as having AHA by subsequent serologic tests. Because HAV is a major etiologic cause of severe acute hepatitis, IgM anti-HAV is usually included in first-line laboratory examinations in such cases. However, serologic tests with IgM anti-HAV may provide a false-negative result during the window period.
In a study of 195 children with IgM anti-HAV negative results during an HAV outbreak in a public school and a child care center, de Paula et al reported that a considerably high (12-13%) proportion showed positive HAV RNA results.4 Among 143 patients confirmed as having acute hepatitis on the basis of IgM anti-HAV by commercially available solid-phase radioimmunoassays during an outbreak, Liaw et al demonstrated that six patients (4.2%) were initially negative when tested within three days after onset of symptoms.5 Bower et al found that HAV RNA was detected an average of 14.4 days before IgM antibody in experimentally infected chimpanzees, with similar data in humans.6 These findings show that serum HAV RNA is generally detected before IgM anti-HAV, and they suggest that HAV RNA RT-PCR may be more sensitive during the early phase of HAV infection, especially during the window period before antibody to HAV is detected.
Bower et al reported that the duration of HAV viremia was 95 days (range, 36-391 days), and viremia persisted for an average 79 days (range, 18-383 days) after the liver enzyme peak.6 Normann et al reported a case in which 4├Ś104 HAV genome equivalents per mL were detected up to 490 days after the onset of jaundice.11 By contrast, in our study, six cases of confirmed AHA among seven cases with negative HAV RNA and positive IgM anti-HAV results showed negative HAV RNA results within 6-16 days after clinical onset. Most of these patients (4 of 6) were categorised in the recovery phase of the disease. This discrepancy between the present study and others may be due to inaccuracies of the reported time of clinical onset of symptoms. Moreover, the cut-off level of qualitative HAV RNA RT-PCR in this study may be relatively high. Also, this finding suggests that HAV RNA RT-PCR may be less sensitive than IgM anti-HAV after the overt phase of the disease. Figure 2 demonstrates the trend of high sensitivity of HAV RNA RT-PCR in the early phase and low sensitivity in the late phase among confirmed AHA patients. And, this result suggests that HAV RNA RT-PCR is probably more useful in early diagnosis of AHA at least in the patients of whom the duration from clinical onset to admission is within 7 days.
This commercially available multiplex PCR method uses the standardized kit, and performs the automated procedure from nucleic acid extraction to RT-PCR process, can monitor real-time results compared to conventional PCR method using agalose gel to see the results. Therefore, this multiplex PCR method is likely to produce the more rapid and reliable results than conventional PCR method. Also, originally it is developed in order to detect the nucleic acid of HAV, HBV, and HCV simultaneously. Thus, it is capable to diagnose the acute viral hepatitis in window and overt period earlier than serologic test. And it also has advantage in the early accurate diagnosis of overt viral hepatitis in immunosuppressant patients who may not produce the serologic markers.
In summary, 30.9% of 136 patients with severe acute hepatitis were confirmed as having HAV infection, and the concordance between IgM anti-HAV and HAV PCR was 88.2%. The sensitivity and specificity of HAV RNA PCR were equivalent to those of IgM anti-HAV for the diagnosis of AHA. Some cases (19%) of AHA were diagnosed by HAV PCR before the appearance of IgM anti-HAV. In the recovery or relapse phase of AHA, some cases showed negative HAV RNA results. We conclude that the qualitative HAV RNA PCR test has equivalent diagnostic accuracy for the diagnosis of AHA as compared with IgM anti-HAV, and may be more sensitive during the window period.
Abbreviations
AHA
acute hepatitis A
ALT
alanine aminotransferase
HAV
hepatitis A virus
HBV
hepatitis B virus
HCV
hepatitis C virus
IgM
immunoglobulin M
INR
international normalized ration
NPV
negative predictive value
PPV
positive predictive value
RNA
ribonucleic acid
RT-PCR
reverse transcriptase polymerase chain reaction
REFERENCES
1. Ostapowicz G, Fontana RJ, Schi├Ėdt FV, Larson A, Davern TJ, Han SH, et al. Results of a prospective study of acute liver failure at 17 tertiary care centers in the United States. Ann Intern Med 2002;137:947-954. 12484709.


2. Lee WM, Squires RH Jr, Nyberg SL, Doo E, Hoofnagle JH. Acute liver failure: Summary of a workshop. Hepatology 2008;47:1401-1415. 18318440.



3. Kim YJ, Lee HS. Increasing incidence of hepatitis A in Korean adults. Intervirology 2010;53:10-14. 20068335.


4. de Paula VS, Villar LM, Morais LM, Lewis-Ximenez LL, Niel C, Gaspar AM. Detection of hepatitis A virus RNA in serum during the window period of infection. J Clin Virol 2004;29:254-259. 15018853.


5. Liaw YF, Yang CY, Chu CM, Huang MJ. Appearance and persistence of hepatitis A IgM antibody in acute clinical hepatitis A observed in an outbreak. Infection 1986;14:156-158. 3759243.


6. Bower WA, Nainan OV, Han X, Margolis HS. Duration of viremia in hepatitis A virus infection. J Infect Dis 2000;182:12-17. 10882576.



7. Yu JW, Sun LJ, Zhao YH, Kang P, Li SC. The study of efficacy of lamivudine in patients with severe acute hepatitis B. Dig Dis Sci 2010;55:775-783. 19957031.


8. Wong VW, Wong GL, Yiu KK, Chim AM, Chu SH, Chan HY, et al. Entecavir treatment in patients with severe acute exacerbation of chronic hepatitis B. J Hepatol 2011;54:236-242. 21030105.


9. Pratt DS. In: Feldman M, Friedman LS, Brandt LJ, eds. Liver chemistry and function tests. Feldman: Sleisenger and Fordtran's Gastrointestinal and Liver Disease. 2010. Vol II: 9th ed. Philadelphia: Saunders/Elsevier; p. 1230-1231.

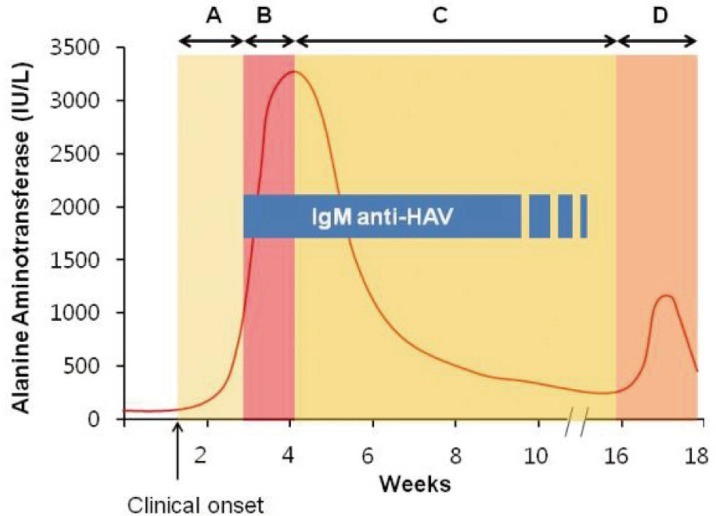
Figure┬Ā1
Disease phase of AHA relative to the time of clinical onset, the initial IgM anti-HAV, and peak serum alanine aminotranferase. A, early phase. B, overt phase. C, recovery phase. D, relapse phase.
Figure┬Ā2
Distribution of HAV IgM and HAV RNA relative to time from clinical onset to initial sampling among confirmed cases of AHA.  , Positive IgM anti-HAV and positive HAV RNA;
, Positive IgM anti-HAV and positive HAV RNA;  , negative or equivocal IgM anti-HAV and positive HAV RNA;
, negative or equivocal IgM anti-HAV and positive HAV RNA;  , positive or equivocal IgM anti-HAV and negative HAV RNA. One patient with positive IgM anti-HAV and positive HAV RNA was not described due to the nonavailability of clinical data.
, positive or equivocal IgM anti-HAV and negative HAV RNA. One patient with positive IgM anti-HAV and positive HAV RNA was not described due to the nonavailability of clinical data.
 , Positive IgM anti-HAV and positive HAV RNA;
, Positive IgM anti-HAV and positive HAV RNA;  , negative or equivocal IgM anti-HAV and positive HAV RNA;
, negative or equivocal IgM anti-HAV and positive HAV RNA;  , positive or equivocal IgM anti-HAV and negative HAV RNA. One patient with positive IgM anti-HAV and positive HAV RNA was not described due to the nonavailability of clinical data.
, positive or equivocal IgM anti-HAV and negative HAV RNA. One patient with positive IgM anti-HAV and positive HAV RNA was not described due to the nonavailability of clinical data.
Table┬Ā1.
Clinical and laboratory characteristics of severe acute hepatitis between HAV and non-HAV infection groups
| Characteristics | HAV group (n=43) | Non-HAV group (n=93) | P value |
|---|---|---|---|
| Age (years) | 33.2┬▒8.30 | 48.6┬▒19.9 | <0.001 |
| Gender (M/F) | 27/16 | 52/41 | 0.450 |
| Initial ALT (IU/L) | 2,389┬▒2,201 | 667┬▒696 | <0.001 |
| Initial albumin (g/dL) | 3.4┬▒0.50 | 3.3┬▒0.69 | 0.490 |
| Initial total bilirubin (mg/dL) | 4.3┬▒3.4 | 5.1┬▒7.6 | 0.406 |
| Initial INR | 1.43┬▒0.64 | 1.50┬▒1.78 | 0.800 |
| Peak ALT (IU/L) | 3,135┬▒2,255 | 893┬▒790 | <0.001 |
| Peak total bilirubin (mg/dL) | 7.3┬▒5.7 | 7.0┬▒10.1 | 0.833 |
| Duration from clinical onset to initial IgM anti-HAV sampling (days)* | 9.3┬▒11.4 | ||
| Duration from clinical onset to peak ALT (days)* | 8.2┬▒9.3 |
Table┬Ā2.
Clinical and laboratory characteristics of AHA between the initially positive and negative IgM anti-HAV groups*
| Characteristics | Positive IgM anti-HAV group (n=34) | Negative IgM anti-HAV group (n=4) | P value |
|---|---|---|---|
| Age (years)ŌĆĀ | 33.9┬▒9.0 | 28.3┬▒1.3 | 0.221 |
| Gender (M/F) | 19/15 | 4/0 | 0.138 |
| Initial ALT (IU/L)ŌĆĀ | 2561┬▒2335 | 2202┬▒2102 | 0.771 |
| Initial albumin (g/dL)ŌĆĀ | 3.3┬▒0.51 | 3.7┬▒0.34 | 0.255 |
| Initial total bilirubin (mg/dL)ŌĆĀ | 4.9┬▒3.5 | 2.3┬▒1.1 | 0.140 |
| Initial INRŌĆĀ | 1.49┬▒0.71 | 1.27┬▒0.19 | 0.547 |
| Peak ALT (IU/L)ŌĆĀ | 3181┬▒2326 | 886┬▒1555 | 0.807 |
| Peak total bilirubin (mg/dL)ŌĆĀ | 7.9┬▒6.0 | 6.5┬▒3.6 | 0.644 |
| Duration from clinical onset to initial IgM anti-HAV sampling (days)ŌĆĀ,ŌĆĪ | 10.6┬▒12.5 | 4.0┬▒1.4 | 0.309 |
| Duration from clinical onset to peak ALT (days)ŌĆĀ,ŌĆĪ | 8.9┬▒10.3 | 5.3┬▒2.1 | 0.489 |
Table┬Ā3.
Concordance analysis between IgM anti-HAV and HAV RNA



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print




