Noninvasive serum biomarkers for liver steatosis in nonalcoholic fatty liver disease: Current and future developments
Article information
Abstract
Nonalcoholic fatty liver disease (NAFLD) affects approximately 30% of the population worldwide and includes nonalcoholic fatty liver, nonalcoholic steatohepatitis (NASH), and cirrhosis. Since NAFLD-associated diseases begin with steatosis, the early diagnosis of steatosis helps to prevent the progression of NASH and fibrosis. In addition, more convenient and easily diagnosable serum biomarkers are becoming crucial in disease diagnosis. In this report, we summarize the known serum biomarkers for liver steatosis and provide guidance for their application in clinical practice.
INTRODUCTION
Nonalcoholic fatty liver disease (NAFLD) is defined as the presence of >5% hepatic steatosis without evidence of liver injury [1,2]. However, the pathophysiology of NAFLD is complex and multifactorial. The most widely known mechanism is accumulated oxidative stress from insulin resistance, and others include an unhealthy diet, lifestyle, genetic factors, and the individual’s microbiome. NAFLDs, regardless of their causative factors, are due to hepatic fat deposition, also known as steatosis; detecting hepatic steatosis is the first step in diagnosing NAFLD.
Liver biopsy, the gold standard for diagnosing NAFLD, is invasive, difficult to interpret, and expensive [3]. Moreover, only a limited range (1/50,000) of the entire liver can be assessed in this manner. Due to the limitations of liver biopsy, other non-invasive methods are being implemented. Ultrasonography is commonly used due to its low cost and wide availability. Recently, with the development of imaging technology, the diagnostic rate of fatty liver by ultrasound has increased to 83.4% [4]. Moreover, the accuracy of ultrasound has been improved by using the differences in the scatter and attenuation of ultrasound waves according to tissue type [5]. Further, the controlled attenuation parameter has the advantage of good feasibility for detecting steatosis and is widely used for steatosis evaluation. However, it cannot reliably differentiate between steatosis grades. Other techniques (such as computed tomography) carry risks associated with radiation exposure, and magnetic resonance imaging is not routinely used due to its cost.
Research into the noninvasive evaluation of hepatic steatosis is ongoing [6]. It is predicted that more than half of the population will be diagnosed with fatty liver in the future, making it critical to find a simple and easy-to-use serum test [7]. Therefore, noninvasive tests have been developed to overcome these limitations, and their use is gradually increasing in clinical practice. This article aims to discuss the existing methods available for classifying steatosis using serum biomarkers.
NAFLD BIOMARKERS
Currently, the most commonly used serum markers are aminotransferase and γ-glutamyl transferase (GGT). Alanine aminotransferase (ALT) has long been used as a marker of liver fat accumulation; in 1986, Nanji et al. first reported the association between liver enzymes (i.e., the ALT-to-aspartate aminotransferase [AST] ratio) and fatty liver in obese patients [8]. As a marker of steatosis, the sensitivity and specificity of ALT are limited [9]; however, there is usually an absence of elevation in aminotransferase levels in steatosis-only conditions [10,11]. Moreover, patients with advanced liver disease show decreased aminotransferase levels [12,13]. GGT is often elevated in NAFLD patients and may be associated with advanced fibrosis and increased mortality rates [14]. However, GGT levels alone cannot identify the degree of steatosis.
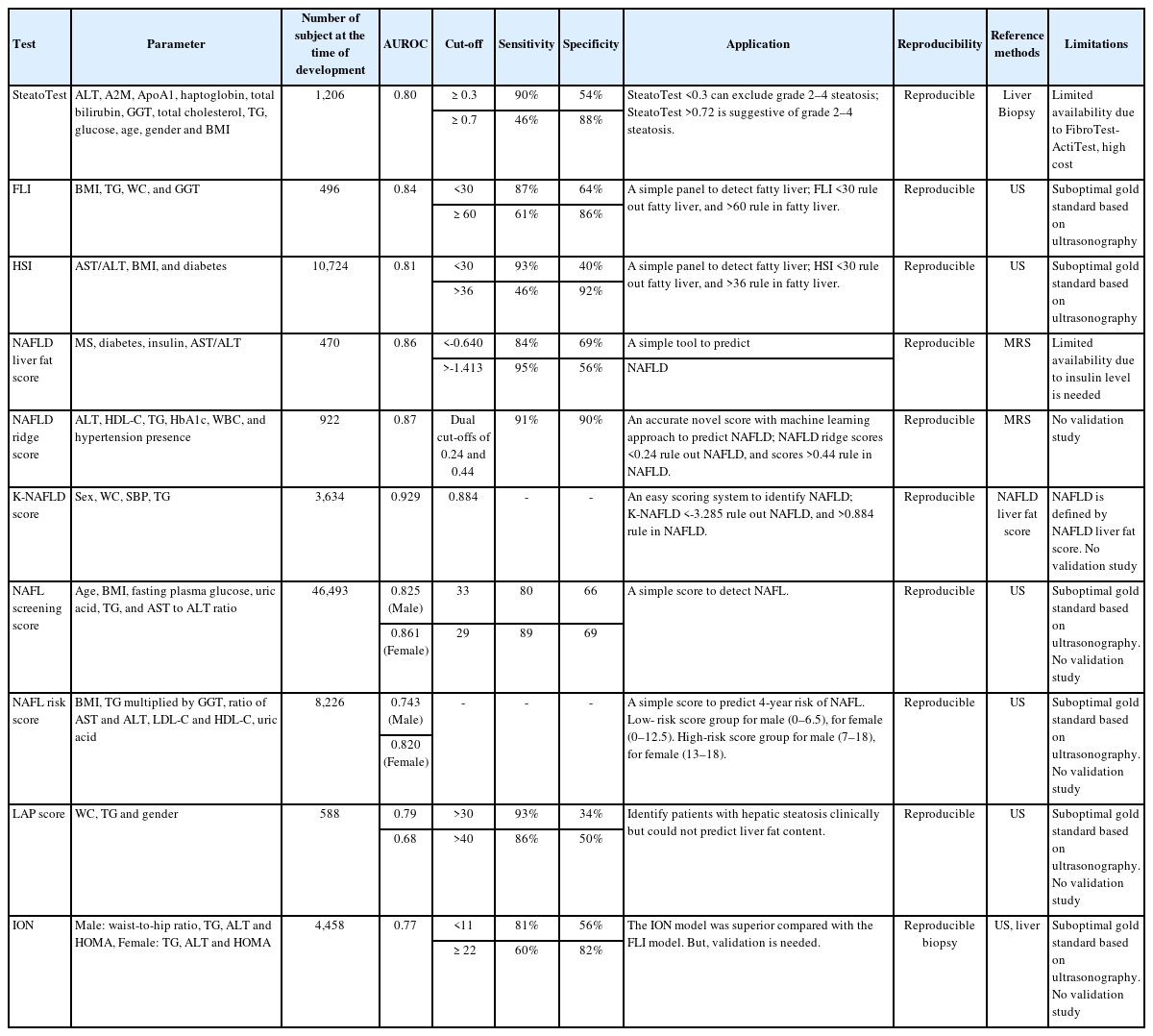
The SteatoTest
The SteatoTest was developed using a combination of the six components of the FibroTest-ActiTest plus the body mass index (BMI), serum cholesterol, triglycerides (TG), and glucose after adjusting for age and sex [15]. It is known to have moderate accuracy in diagnosing liver steatosis (the area under the curve of the receiver operating characteristic [AUROC]: 0.79–0.80; sensitivity: 80–100%; specificity: 83–100%). The patients were classified according to hepatitis C treatment and alcoholic liver disease, and analyzed by dividing them into a training group and three validation groups. For the diagnosis of Grade 2–4 steatosis, the sensitivity values of the SteatoTest at the 0.30 cut-off value were 0.91, 0.98, 1.00, and 0.85, while the specificity data at the 0.70 cut-off were 0.89, 0.83, 0.92, and 1.00, respectively.
The SteatoTest has better predictive power than ALT and GGT serum markers: a meta-analysis has shown an AUROC of 0.80 for diagnosing steatosis >33% [16]. The disadvantage of this biomarker is that it is difficult to use in clinical practice and is expensive; it is also unable to discriminate between different levels of steatosis, and it cannot be used if the FibroTest-ActiTest is not available. (Table 1).
The fatty liver index (FLI)
The FLI utilizes four components: BMI, waist circumference, serum TG, and serum GGT. Based on abdominal ultrasonography studies, the FLI is moderately accurate (AUROC: 0.84; sensitivity: 87%; specificity: 64%) [17]. An FLI <30 (negative likelihood ratio=0.2) rules out and an FLI ≥60 (positive likelihood ratio=4.3) confirms fatty liver. Another study has suggested that the FLI is associated with insulin resistance and all-cause, liver-related, and cancer mortality [18].
The FLI uses information that can be easily obtained in clinical practice and is moderately accurate; however, ultrasonography, not liver biopsy, was used as a reference standard.
The hepatic steatosis index (HSI)
The HSI involves four components: the AST/ALT ratio, BMI, sex, and the presence of diabetes mellitus [19]. At values of <30.0 or >36.0, the HSI rules out NAFLD with a sensitivity of 93.1% or detects NAFLD with a specificity of 92.4%, respectively. The HSI was shown to have an AUROC of 0.81 in a large cohort study (n=10,724) of Korean patients. However, ultrasonography was used as a reference standard, and validation studies in other populations are required.
The nonalcoholic fatty liver disease liver fat score
The NAFLD liver fat score involves five components: the presence of metabolic syndrome or type 2 diabetes mellitus, the fasting serum insulin, the serum AST, and the AST/ALT ratio [20]. A study based on magnetic resonance spectroscopy has shown high accuracy (AUROC, 0.86–0.87; sensitivity, 86%; specificity, 71% [cut-off point of -0.640]). NAFLD liver fat scoring was validated using magnetic resonance spectroscopy as a reference standard and showed relatively good diagnostic performance. The downside of this biomarker is that it requires fasting serum insulin test results, which are not yet standard.
The nonalcoholic fatty liver disease ridge score
The NAFLD ridge score is a machine learning-based method that utilizes seven components: serum ALT, high-density lipoprotein cholesterol, TG, hemoglobin A1c, leukocyte count, comorbidity data, and the presence of hypertension. NAFLD ridge scoring uses proton magnetic resonance spectroscopy as a reference standard. By using dual cut-offs of 0.24 and 0.44, the NAFLD ridge score achieved 92% (86–96%) sensitivity and 90% (86–93%) specificity. This method showed good accuracy levels (AUROC: 0.87; sensitivity: 92%; specificity: 90%) and excellent negative predictive values (96% to exclude NALFD) [21]. The downside of this method is that there are no subsequent validation studies.
The K-nonalcoholic fatty liver disease score
This scoring system was created based on a sample of 3,634 patients and includes four components: sex, waist circumference, systolic blood pressure, and serum TG. A cut-off value for NAFLD was set at 0.884 [22]. K-NAFLD scores <-3.285 and >0.884 were set as the cut-off values for no NAFLD and NAFLD. The K-NAFLD scoring method is based on data from a large cohort of patients, and it showed the most accurate (AUROC=0.929) predictive power compared to other biomarkers (FLI [AUROC=0.870]; LAP [AUROC=0.841]; and body mass index, age, alanine aminotransferase, and TG [BAAT] [AUROC=0.782]). However, the scoring system was created without using a liver biopsy or imaging study as a reference standard and therefore requires validation using other populations.
The nonalcoholic fatty liver screening score
The nonalcoholic fatty liver screening score (NSS) was based on a large cohort study of >40,000 people that utilized a total of six components: age, fasting plasma glucose, urinalysis, the ALT/AST ratio, BMI, and TG. A total score >29 correlates to a high risk for NAFL. For males, at the cut-off point of 33, the NSS had a sensitivity of 79.86% and a specificity of 66.13%. For females, at a value of 29, the sensitivity and specificity values of the NAFL screening score were 89.39% and 68.98%, respectively. This scoring system showed a higher accuracy than other NAFL models (male AUROC: 0.825 [0.806–0.843], and female AUROC: 0.861 [0.820–0.896], compared with the HSI: 0.791 [0.770–0.810] and the FLI: 0.805 [0.785–0.82]) [23].
The NAFL screening score was created based on a large cohort of patients and was particularly accurate for men, demonstrating higher AUROC values than other steatosis markers. However, ultrasonography was used as a reference standard, and validation studies have not yet been conducted.
The nonalcoholic fatty liver risk score
This scoring system was developed to predict the future four-year risk of NAFLD. The outcome is a score between 0 and 18 points that is based on five measurements: BMI, TG×GGT, ALT/AST ratio, low-density lipoprotein/high-density lipoprotein cholesterol ratio, and uric acid levels.
The advantage of this marker is that the NAFL risk score was relatively discriminative (AUROC=0.739 for males and 0.823 for females) [24]. However, ultrasonography was used as a reference standard, and validation studies were not conducted.
The lipid accumulation product score
The LAP score uses three variables (waist circumference, TG, and sex) with moderate accuracy (AUROC: 0.79) to diagnose >30% of steatosis [25]. The degree of steatosis can be evaluated using this method, which has been validated as moderately accurate (AUROC: 0.79) in diagnosing >5% of steatosis [26]. Again, ultrasonography was used as a reference standard, and validation studies in other ethnic groups are warranted.
The index of nonalcoholic steatohepatitis (ION)
The ION Model was created using the data from 4,458 NAFLD patients from the National Health and Nutrition Examination Survey III and 152 patients with biopsy-proven NAFLD [27]. This model uses different variables that are calculated using the sex/waist-to-hip ratio, TG, ALT, and the Homeostatic Model Assessment for Insulin Resistance (HOMA) in males and the TG, ALT, and HOMA in females. The ION had an AUROC of 0.77, a sensitivity of 81% for ruling out steatosis at a cut-off <11, and a specificity of 82% for ruling in steatosis at a cut-off >22. The ION model was superior in predicting NASH and mortality compared with the FLI model; however, ultrasonography was used as a reference standard.
DISCUSSION AND CONCLUSIONS
We investigated the biomarkers currently used in evaluating hepatic steatosis. Serum markers have several limitations in evaluating steatosis alone and are thus commonly combined with other markers including sex, age, BMI, and waist circumference.
Limitations exist when making direct comparisons between the methods mentioned above. First, the models were compared to different standards when assessing accuracy, such as liver biopsy, ultrasonography, and magnetic resonance spectroscopy. The FLI, NAFLD liver fat score, and HSI were obtained from the same cohort of patients and were hence directly comparable; however, the AUROC values between the methods were similar (0.83, 0.80, and 0.81, respectively). A previous study externally validated the involved hepatic steatosis formulas. In this study, the NAFLD liver fat score showed the best diagnostic performance and similar diagnostic agreement with ultrasonography [28].
Novel serum markers to evaluate steatosis are being developed; however, a reliable method has not been widely validated, and further research is required. Since the long-term prognosis of NAFLD is more likely to be associated with fibrosis than steatosis, the focus on steatosis could be lessened. It is crucial to make an early diagnosis of steatosis to prevent the progression of NASH and fibrosis; this can be challenging since NAFLD and NASH are usually asymptomatic until patients reach the advanced stages. The high applicability, reproducibility, and widespread availability of serum biomarkers gives them an advantage over other methods. There is a demand for easy and precise methods, not only for diagnostic purposes but also to evaluate treatment outcomes. The lack of noninvasive methods to evaluate steatosis in both the clinical and research fields hinders the enrollment of new patient study objects. Thus, the development of a noninvasive steatosis marker is warranted for future pharmaceutical research and development.
While liver steatosis can be an effective measure of liver disease, this condition can also diminish during the progression of NAFLD to liver cirrhosis, which is known as the “burnout” effect. For patients with later-stage NAFLD, assessing the severity of NASH and fibrosis could be more critical than steatosis. Thus, it is crucial to identify high-risk groups that are likely to develop liver fibrosis to ensure that these groups are followed up regularly. Recognizing the limitations of serum markers is important; integrating imaging studies and patient information during the diagnosis process results in better outcomes. Furthermore, circulating biomarkers, such as microRNA and cell-free nuclear material DNA/RNA, and “omics” studies are yet to be developed for commercialization but may be critical in future clinical and research practices [29].
Abbreviations
ALT
alanine aminotransferase
AST
aspartate aminotransferase
AUC
area under the curve
AUROC
area under the curve of the receiver operating characteristic
BMI
body mass index
FLI
fatty liver index
GGT
γ-glutamyl transferase
HSI
hepatic steatosis index
LAP
lipid accumulation product
NAFL
nonalcoholic fatty liver
NAFLD
nonalcoholic fatty liver disease
NASH
nonalcoholic steatohepatitis
TG
triglyceride