Combined treatment with silibinin and either sorafenib or gefitinib enhances their growth-inhibiting effects in hepatocellular carcinoma cells
Article information
Abstract
Background/Aims
Silibinin, the main component of silymarin, is used as a hepatoprotectant and exhibits anticancer effects against various cancer cells. This study evaluated the effects of a combination of silibinin with either gefitinib or sorafenib on hepatocellular carcinoma (HCC) cells.
Methods
Several different human HCC cell lines were used to test the growth-inhibiting effects and cell toxicity of silibinin both alone and in combination with either gefitinib or sorafenib. The cell viability and growth inhibition were assessed using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay, trypan blue staining, and a colony-forming assay. Furthermore, changes in epidermal growth factor receptor (EGFR)-related signals were evaluated by Western blot analysis.
Results
Gefitinib, sorafenib, and silibinin individually exhibited dose-dependent antiproliferative effects on HCC cells. Combined treatment with silibinin enhanced the gefitinib-induced growth-inhibiting effects in some HCC cell lines. The combination effect of gefitinib and silibinin was synergistic in the SNU761 cell line, but was only additive in the Huh-BAT cell line. The combination effect may be attributable to inhibition of EGFR-dependent Akt signaling. Enhanced growth-inhibiting effects were also observed in HCC cells treated with a combination of sorafenib and silibinin.
Conclusions
Combined treatment with silibinin enhanced the growth-inhibiting effects of both gefitinib and sorafenib. Therefore, the combination of silibinin with either sorafenib or gefitinib could be a useful treatment approach for HCC in the future.
INTRODUCTION
Liver cancer is the sixth most common neoplasm in the world, and its poor prognosis makes it the third leading cause of cancer-related mortality.1 Because of early angioinvasion and metastasis, only a small proportion of patients with hepatocellular carcinoma (HCC) receives surgical resection or liver transplantation as a curative treatment.2 Except for sorafenib, a multikinase inhibitor, there are no effective systemic treatments of HCC. Two large phase III randomized controlled trials showed that the median overall survival was 10.7 months3 and 6.5 months4 in a sorafenib-treated group. Therefore, sorafenib has been used as a salvage therapy for advanced HCC. However, in the treatment of HCC, sorafenib alone has demonstrated insufficient responses and has resulted in several adverse effects, such as hand foot skin reaction, diarrhea, and jaundice.3,4 Recently, there are several studies that address the multidisciplinary management of HCC, which includes sorafenib treatment,5,6,7,8,9 to improve overall survival as well as drug combination therapy based on sorafenib.10,11,12 Unfortunately, there has been no definite combination treatment that improves the overall survival of HCC patients.
Milk thistle (Silybum marianum) has been used widely as an herbal remedy for liver disease. Milk thistle extract is composed almost completely of silymarin, and silibinin (silybin) is the major active compound of silymarin.13,14 Many studies have revealed the antitumor effects of silibinin on multiple cancer cells, such as prostate,15,16,17,18 colon,19,20 skin,21,22 bladder,23,24,25 and lung cancers,26 Moreover, Varghese et al. and Lah et al. identified the efficacy of silibinin in HCC cells.27,28 Recently, Rho et al reported that combined treatment with silibinin and epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors (TKIs) overcame drug resistance caused by the T790M mutation in non-small cell lung cancer cell lines.29 These results demonstrated the combined synergistic effect of gefitinib and silibinin.
We investigated the efficacy of combined treatment with silibinin and either sorafenib or gefitinib on HCC cells and suggest a new strategy in the treatment of HCC.
MATERIALS AND METHODS
Study design
To assess the combination effects, we compared the cell growth after combined treatment with silibinin and either gefitinib or sorafenib with the single treatment of each drug in the following human HCC cell lines: Huh7, HepG2, Huh-BAT, Hep3B, HepG2, PLC/PRF5, SNU387, SNU398, SNU449, SNU475, and SNU761.
We examined the cell growth by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, trypan blue staining, and a colony-forming assay then determined alterations in intracellular signals using Western blot analysis.
Materials
The human HCC lines (Hep3B, HepG2, Huh7, PLC/PRF5, SNU387, SNU398, SNU449, SNU475, and SNU761) were purchased from the Korean Cell Line Bank. Huh-BAT cells were obtained from the Liver Research Institute, Seoul National University. Gefitinib was kindly provided by AstraZeneca Korea. Sorafenib and silibinin were purchased from Selleck Chemicals (Houston, TX, USA) and Sigma-Aldrich Co. (St. Louis, MO, USA), respectively.
Cell lines and cell culture
The human HCC cell lines were cultured in DMEM, MEM, and RPMI media supplemented with 10% fetal bovine serum and 100 U/mL penicillin in 5% CO2 at 37℃.
MTT assay
Cells were plated in 96-well plates at a density of 5,000 cells/well and cultured overnight. Then, the cells were exposed to varying concentrations of either gefitinib or sorafenib or silibinin for 72 h. Subsequently, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution was added to the media containing the various drugs; after incubating for 3 h, a 10% SDS solution was added to the samples. The samples were cultured for 24 h, and the color absorbance was read at a wavelength of 595 nm.
Colony-forming assay
Cells were plated in 60-mm plates at a density of 1,000 cells/well and cultured overnight. Then, the cells were treated with either gefitinib or sorafenib and/or silibinin for 72 h. Thereafter, the cells were incubated in drug-free media for 2 weeks and then stained using methylene blue. The colonies were photographed and then counted.
Trypan blue staining
Cells were plated in 60-mm plates at a density of 1,000 cells/well and cultured overnight. Then, the cells were treated with either gefitinib or sorafenib and/or silibinin for 72 h. Thereafter, the cells were incubated in drug-free media for 10 days and then stained using trypan blue to count cell numbers.
Combination index (CI) analysis
Combined at a constant ratio, gefitinib or sorafenib:silibinin (1:100 µM) were measured by an MTT assay. CI plots were computationally generated by CalcuSyn software (Biosoft, Cambridge, UK)
Western blot
Cell lysates were prepared in non-denaturing lysis buffer (20 mmol/L Tris-HCl; pH 7.4; 150 mmol/L NaCl; 1 mmol/L EDTA; 1 mmol/L EGTA; 1 mmol/L ß-glycerophosphate; 1 mmol/L sodium vanadate; 1 µg/mL leupeptin; 1 mmol/L PMSF). The protein was collected after centrifugation at 13,000 × g for 10 min. The resulting supernatant was separated on a 10% SDS-PAGE gel and transferred to Immobilon-P membrane (Millipore, Bedford, MA, USA). The membrane was blocked with 5% non-fat skim milk-PBS-T (137 mmol/L NaCl; 2.7 mmol/L KCl; 10 mmol/L phosphate buffer; 0.01% Tween-20) for 1 hr at room temperature before being incubated overnight with a primary antibody against p-EGFR, EGFR, IGF-1R, p-Akt, Akt and Erk (all from Santa Cruz Biotechnolongy [Santa Cruz, CA, USA]), and p-IGF-1R and p-Erk (from Cell Signaling Technology [Beverly, MA, USA]), and β-actin (from Sigma-Aldrich [St. Louis, MO, USA]). Then, the membrane was washed three times in PBS-T for 5 min and incubated with horseradish peroxidase-conjugated secondary antibody for 1 h and washed three times in PBS-T for 10 min. After washing, the membrane was developed using chemiluminescence and exposed to X-ray film to visualize the protein bands.
RESULTS
Effects of gefitinib or silibinin treatment depend on the HCC cell line
Cell viability was evaluated by MTT assay in the following gefitinib- or silibinin-treated HCC cell lines: Huh7, Huh-BAT, HepG2, SNU475, and SNU761. Gefitinib treatment resulted in the dose-dependent inhibition of Huh7, Huh-BAT, HepG2, and SNU761 cell growth (Fig. 1A). Silibinin treatment also significantly inhibited the cell growth of Huh7, Huh-BAT, and SNU761 cells; however, the cell growth of HepG2 and SNU475 cells increased with low doses of silibinin and decreased in high doses of silibinin.

Growth-inhibiting effects of gefitinib and/or silibinin in hepatocellular carcinoma cells (HCCs). A and B: Huh7, Huh-BAT, HepG2, SNU475, and SNU761 HCC cell lines were cultured in medium containing 5% fetal bovine serum (FBS) and treated with gefitinib (A, left), silibinin (A, right), or the two in combination (B) for 72 hr. The resulting cell viability was determined using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. C and D: The combination effects of gefitinib (1 µM) and silibinin (100 µM) in SNU761 (C) and Huh-BAT (D) cells were measured using the MTT assay. Combination index (CI) plots were generated using computer software; CI values of <1, 1, and >1 indicate synergism, an additive effect, and antagonism, respectively. G, gefitinib; S, silibinin; G+S, gefitinib+silibinin.
Combined treatment of HCC cells with gefitinib and silibinin has a synergistic effect and the effect is associated with the inhibition of EGFR via the downregulation of Akt
The combined-treatment effect of gefitinib and silibinin is displayed in Figure 1B. The combined-treatment effects of gefitinib and silibinin were assessed in SNU475, SNU761, Huh7, Huh-BAT, and HepG2 cells and demonstrated a significant inhibiting effect in SNU761 and Huh-BAT cells. The CI in the SNU761 cells was below 1, which indicates a synergistic effect of gefitinib and silibinin combination treatment. However, in Huh-BAT cells, the CI was below 1 at some doses, which indicates an additive effect rather than a synergistic effect (Fig. 1C and 1D).
In the SNU761 and Huh-BAT cell lines, the cell numbers were decreased more in the combined treatment (gefitinib and silibinin) compared with the single treatment (gefitinib or silibinin) (Fig. 2A and 2B). In addition, in the colony-forming assay, the colony numbers were decreased more in the combination treatment (gefitinib and silibinin) compared with the single treatment (gefitinib or silibinin) (Fig. 2C and 2D). These results established that the combined-treatment effects of gefitinib and silibinin produce significant inhibition of cellular growth in HCC cells.
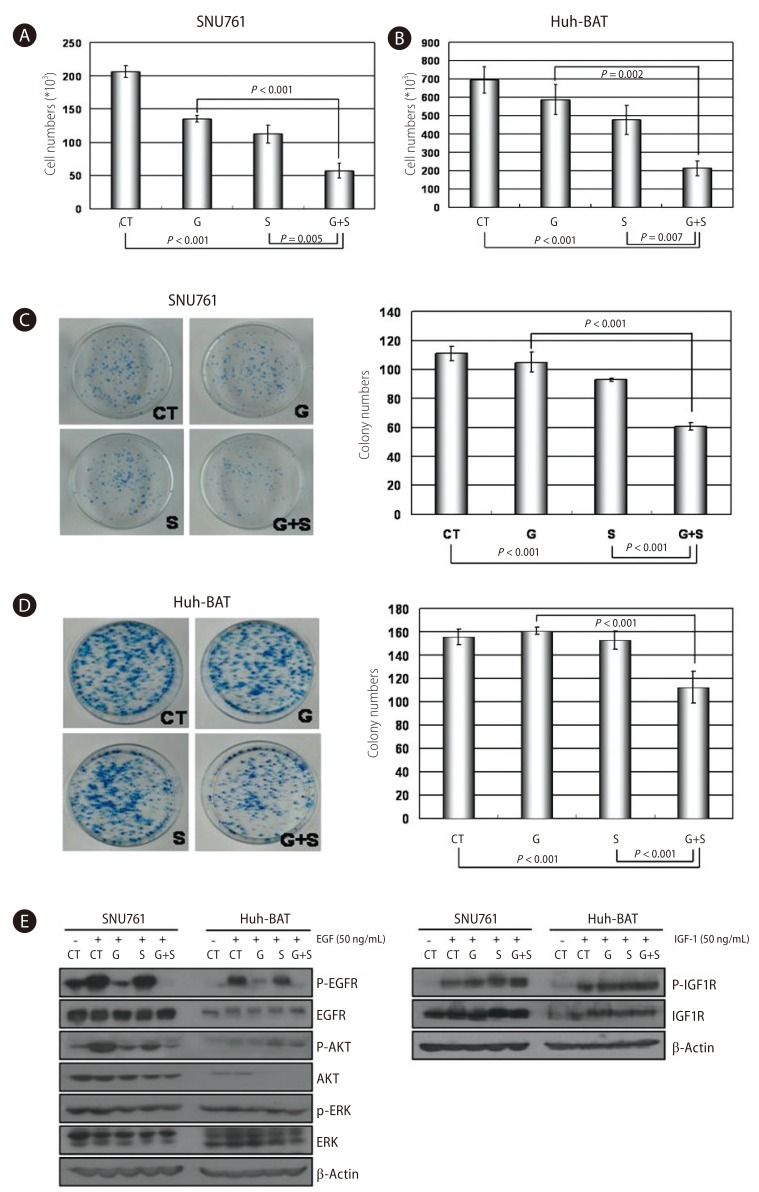
Combined treatment with gefitinib and silibinin inhibited cell proliferation through the inhibition of Akt signaling in HCC cells. A and B: SNU761 (A) and Huh-BAT (B) cell lines were treated with gefitinib, silibinin, or the two in combination for 72 hr, and viable cells were counted after staining with trypan blue. C and D: The combination effects of gefitinib and silibinin on HCC cells were evaluated using a colony-forming assay in SNU761 (C) and Huh-BAT (D) cells. E: SNU761 and Huh-BAT cells were treated with silibinin (100 µM) and gefitinib (1 µM) alone or in combination. After treating HCC cells with epidermal growth factor (EGF) or insulin-like growth factor 1, molecules of EGF receptor-related signaling were detected by Western blot analysis. CT, control; G, gefitinib (1 µM); S, silibinin (100 µM); G+S, gefitinib (1 µM)+silibinin (100 µM).
To clarify the mechanism of the anti-proliferative effects, the activity of EGFR, IGF1R, and the downstream molecules of IGF1R were tested. The activity of IGF1R and IGF1R-related molecules were not altered, but the activity of EGFR was decreased in both SNU761 and Huh-BAT cells. In SNU761 cell lines, the activity of Akt, the downstream signal of the EGFR pathway, was decreased, but in Huh-BAT cell lines did not. These results are illustrated in Figure 2E.
The inhibiting effects of sorafenib are different in each HCC cell line
To confirm the anticancer effect of the multikinase inhibitor sorafenib on HCC, we examined the cell viability of various HCC cells using an MTT assay. In all of the HCC cell lines tested, sorafenib inhibited cell growth in a dose-dependent manner. The half-maximal inhibiting concentrations (IC50) were approximately 1.77-2.64 µM in Huh7, Huh-BAT, HepG2, Hep3B, and PLC/PRF5 cells, which indicates that the anticancer effects of sorafenib on HCC are effective at a relative low dose; yet, the IC50 in the SNU387, SNU398, SNU475, and SNU761 cells were approximately 8.31-13.31 µM. The inhibiting effects of sorafenib were dependent on each HCC cell line (Fig. 3A).
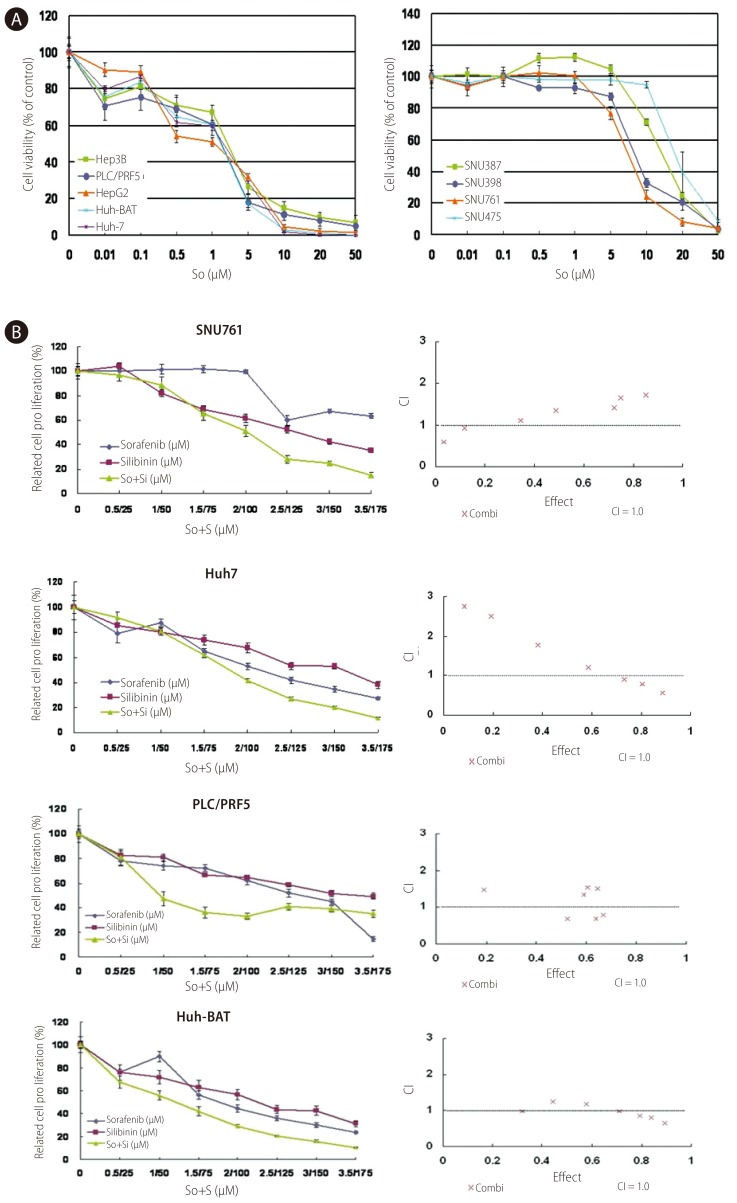
Growth inhibition of HCC cells by sorafenib alone or in combination with silibinin. A: Huh7, Huh-BAT, HepG2, Hep3B, PLC/PRF5, SNU387, SNU398, SNU449, SNU475, and SNU761 HCC cell lines were cultured in medium containing 5% FBS and treated with the indicated doses of sorafenib for 72 hr. The resulting cell viability was determined using the MTT assay. B: SNU761, Huh7, Huh-BAT, and PLC/PRF5 cell lines were cultured in medium containing 5% FBS and treated with the indicated amounts of sorafenib, silibinin, or the two in combination for 72 hr. The resulting cell viability was measured using the MTT assay. Cell viability curves (left) and CI plots (right) were generated using computer software. So, sorafenib; S, silibinin.
The addition of silibinin to sorafenib inhibited the growth of some HCC cell lines
The combination of silibinin and sorafenib demonstrated an additional inhibiting effect in SNU761, Huh7, PLC/PFR and Huh-BAT cells. The CIs indicated a synergistic effect of combined silibinin and sorafenib treatment at some doses. Cell growth and CIs are displayed in Figure 3B.
In SNU761 and Huh7 HCC cell lines, the cell numbers were more decreased with combined treatment than the single treatment of sorafenib or silibinin (Fig. 4A and 4B). The enhanced growth-inhibiting effect of combination treatment (sorafenib and silibinin) was confirmed again in colony forming assay by reduced colony numbers in combined treatment of sorafenib and silibinin (Fig. 4C and 4D).
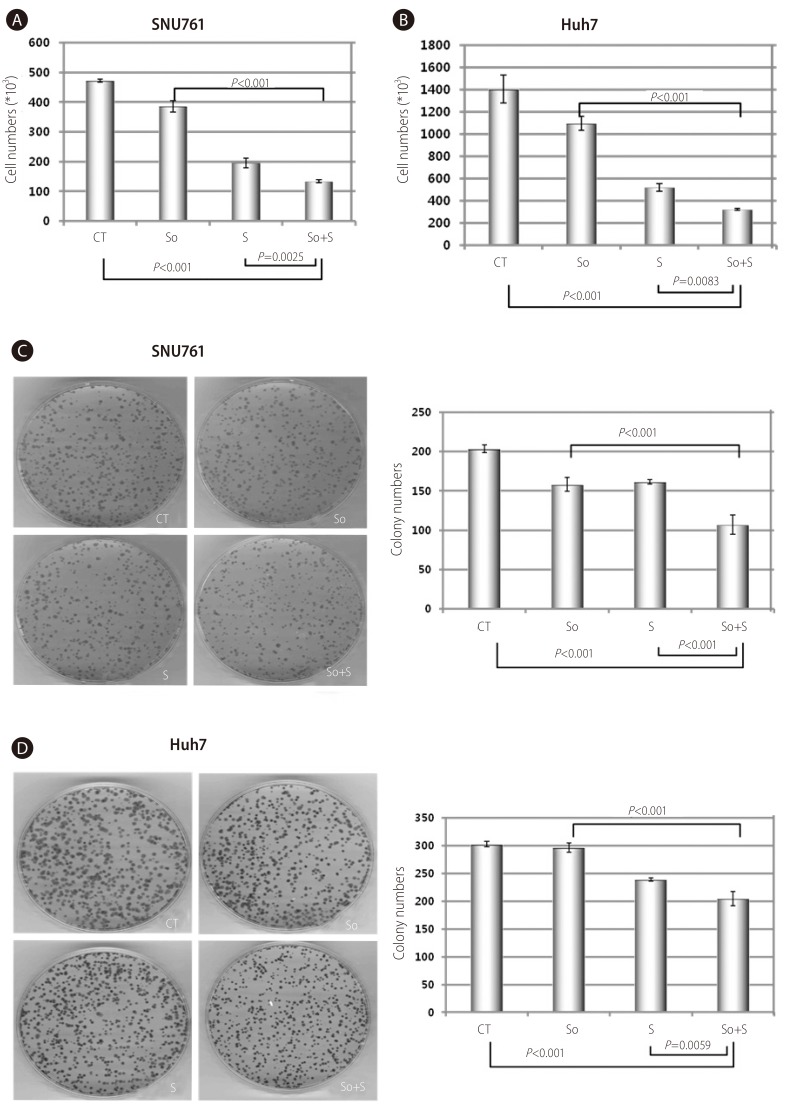
Combined treatment of sorafenib with silibinin conferred an additional growth-inhibiting effect in SNU761 and Huh7 HCC cell lines. A and B: SNU761 (A) and Huh-7 (B) cell lines were treated with sorafenib (2 µM), silibinin (100 µM), or a combination of the two for 72 hr, and viable cells were counted after staining with trypan blue. C and D: The combination effects of sorafenib (2 µM) and silibinin (100 µM) on SNU761 (C) and Huh-7 (D) HCC cells were evaluated using a colony-forming assay. CT, control; So, sorafenib (2 µM); S, silibinin (100 µM); So+S, sorafenib (2 µM)+silibinin (100 µM).
DISCUSSION
HCC has a poor prognosis due to early vascular invasion, rapid growth rate, underlying chronic hepatitis or hepatic cirrhosis. Furthermore many HCC patients are diagnosed at advanced stage, so they cannot receive curative therapy.30,31 Sorafenib is approved as the standard of palliative therapy in patients with advanced HCC, but the clinical benefits have been limited.32 Therefore, the development of more effective and less hepatotoxic treatments with fewer side effects is necessary.
Empirically, silibinin has been used as a hepatoprotectant for more than 2000 years. Many studies have established the anti-cancer effect of silibinin against various cancers.15,16,17,18,19,20,21,22,23,24,25,26 The anti-tumor effect exhibited by silibinin is facilitated by the inhibition of EGFR activation, which is the first step of the signaling pathway of cell growth associated with tumor cell growth.19,20,25,27,28,33,34,35,36,37,38 Silibinin induces apoptosis via the downregulation of CDK expression, which regulates cell cycles, and the increased expression of p21 and p27, which inhibits CDK and arrests the cell cycle at G1 and G2.25,27,37 It was reported that the mechanism of EGFR-activation inhibition occurred via interruptions of ligand synthesis37, ligand binding39, and receptor dimerization29. Because silibinin is lipophilic, it can easily move on cell membranes and directly insert into membrane lipids and can have multiple actions. Moreover, silibinin suppresses angiogenesis and metastasis.34,40
This study investigated the effect of target agents combined with silibinin, which inhibits EGFR activation and regulates cellular growth factors. We obtained results by testing the target agents on many human HCC cell lines. Sorafenib, gefitinib, and silibinin could reduce the cell growth of many HCC cell lines. The combined treatment of gefitinib and silibinin significantly suppressed cell growth in some HCC cell lines, such as SNU761 and Huh-BAT cells, compared with gefitinib treatment alone. These results are attributed to alterations in the intracellular EGFR signaling pathway via the downregulation of Akt signaling. In some HCC cell lines, the combined treatment with silibinin and sorafenib significantly attenuated the cell growth compared with sorafenib treatment alone.
Varghese et al. and Lah et al. reported that silibinin significantly reduced the cell growth of many HCC cell lines.27,28 They showed the inhibiting effect of silibinin in a dose-dependent manner. In our study, we observed similar results. Moreover, silibinin has other, more common effects, such as antioxidant, anti-inflammatory, anti-fibrotic, and metabolic. Silibinin may offer protection from acute liver damage; therefore, many clinicians prescribe silibinin to prevent severe liver injury and to resolve liver damage. Additionally, silibinin is used for the treatment of chronic liver diseases, such as chronic hepatitis C and alcoholic hepatitis. In our study, we observed that the cell viability of the HepG2 and SNU475 cells was increased when treated with the low dose of silibinin. These results may explain the reasons for silibinin use in liver damage. While, silibinin showed anticancer effects in most of the HCC cell lines at a dose over 100 µM. Recently, Flaig et al employed large doses (2.5-20 g) of silibin-phosphatidylcholine.41 When treated with over 15 g of silibin-phosphatidylcholine, certain patients showed only asymptomatic hyperbilirubinemia; however, in all of the patients, hyperbilirubinemia was reversed after the discontinuation of silibin-phosphatidylcholine. This study provided evidence that silibinin can be administered at high doses. However, further investigations are warranted to investigate whether higher doses can induce a higher blood concentration and whether there is a relationship between the blood concentration and the tissue concentration of silibinin.
Gefitinib, an EGFR-TKI, is a potent anticancer agent on EGFR positive non-small cell lung cancer, especially adenocarcinoma.42 Some reports have stated that EGFRs are expressed frequently in HCC.43,44 Hopfner et al. described gefitinib-induced growth inhibition, apoptosis, and cell cycle arrest in HCC cells.45 Additionally, in our study, gefitinib reduced cell growth in each HCC cell line, except in SNU475 cells, in dose-dependent manner. Moreover, the combined treatment of HCC cells with silibinin and gefitinib showed a synergistic effect at all concentrations tested in SNU761 cells and at some concentrations tested in Huh-BAT cells; other HCC cell lines showed a significant reduction of cell growth at higher concentrations of silibinin and gefitinib treatment. In the trypan blue staining assay and the colony-forming assay, cell numbers and colony numbers were more decreased in HCC cell lines exposed to the combined treatment compared with the single treatment of gefitinib or silibinin. Furthermore, Western blot analysis revealed the decreased activity of EGFR and Akt in SNU761 cells. However, in Huh-BAT cells that demonstrated a synergistic effect of the combination treatment at partial concentrations, the activity of EGFR was declined, but the activity of Akt was not declined. These different effects indicate the heterogeneity of HCC in each HCC cell line.
Silibinin is a safe drug with a few mild adverse effects, including gastrointestinal symptoms such as abdominal distention, indigestion, nausea, and diarrhea, which are all common and reversible. The effective anticancer dosage of silibinin, as stated previously, could be higher than the current dosages used; this issue needs to be addressed in future investigations.
Sorafenib, which is used widely as an effective target agent for HCC treatment, was shown to possess anti-proliferative effects in all of HCC cell lines tested, although the IC50 in each of the cell line was different. The combined effects of silibinin and sorafenib on the HCC cell lines tested indicated a synergistic effect in a dose-dependent manner. Many ongoing studies are addressing which drug is most effective when combined with sorafenib.46,47 However, there are not any drugs available. Although this study was performed in vitro, it is promising that silibinin produces a synergistic effect with target agents, especially sorafenib. We suggest that combination treatment with target agents and silibinin could be a useful approach for future HCC treatments.
Acknowledgements
This research was supported by a grant of the Radiological Translational Research Program, Korea Institute of Radiological and Medical Sciences (50351-2009) and the GlaxoSmithKline Research Fund of the Korean Association for the Study of the Liver.
Notes
Conflicts of Interest: The authors have no conflicts to disclose.
Abbreviations
EGFR
epidermal growth factor receptor
HCC
hepatocellular carcinoma
MTT
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
TKIs
tyrosine kinase inhibitors