| Clin Mol Hepatol > Volume 23(2); 2017 > Article |
|
ABSTRACT
Background/Aims
Mongolia has one of the highest hepatitis A, C, B and D infection incidences worldwide. We sought to investigate changes in the proportion of acute viral hepatitis types in Mongolia over the last decade.
Methods
The cohort comprised 546 consecutive patients clinically diagnosed with acute viral hepatitis from January 2012 to December 2014 in Ulaanbaatar Hospital, Mongolia. A time trend analysis investigating the change in proportion of acute hepatitis A virus, hepatitis C virus (HCV), hepatitis B virus (HBV) and hepatitis delta virus (HDV) infection among the cohort with respect to a previous published study was undertaken.
Results
Acute hepatitis A, B and C was diagnosed in 50.9%, 26.2% and 6.0% of the cohort. Notably, 16.8% of the cohort had a dual infection. The etiologies of acute viral hepatitis were varied by age groups. The most common cause of acute viral hepatitis among 2-19 year olds was hepatitis A, HBV and superinfection with HDV among 20-40 year olds, and HCV among 40-49 year olds. Patients with more than one hepatitis virus infection were significantly older, more likely to be male and had a higher prevalence of all risk factors for disease acquisition. These patients also had more severe liver disease at presentation compared to those with mono-infection.
According to the World Health Organization, 350 and 170 million are chronically infected with hepatitis B virus (HBV) and hepatitis C virus (HCV) infections, respectively [1,2]. A further 15-20 million people with HBV infection are co-infected with hepatitis delta virus (HDV). Central and Northern Asia are amongst the regions with the highest prevalence of HDV, which may infect up to 90% of HBV carriers in this region [3].
Mongolia in central Asia is a unique country since all three chronic hepatitis viruses are highly endemic, as is infection from hepatitis A. As of July 2013, Mongolia had a population of just 3 million [4], a very low population density (1.8 people per km2) and a relatively young population age demographic with only 3.5% of people aged over 65 years [5]. The country is classified geographically into regions comprising either urban or rural locals [4,5]. Ulaanbaatar, the capital, is the largest city and is home to thirty-two percent of the nationŌĆÖs population [4].
In a previous study [6], we reported on the prevalence of HBV, HDV and HCV infections in 110 consecutive patients presenting with acute hepatitis at eight city hospitals in Ulaanbaatar. In that study, 16.4%, 32.7%, 6.4%, 1.8%, and 27.3% of the patients were diagnosed as having acute hepatitis due to hepatitis A virus (HAV), HBV, HCV, HBV/HDV coinfection, and superinfection, respectively.
Given the high prevalence of chronic viral hepatitis in Mongolia, not surprisingly, end stage liver disease and liver cancer are major cause of morbidity and mortality. Thus, the incidence of hepatocellular carcinoma is six times higher than the global average. Mortality in males in Mongolia is mainly due to cancers of the liver and stomach; and in females, it is most commonly due to liver and cervical cancers [7-9].
As Mongolia develops, with improvements in housing and sanitation, universal HBV vaccination, uniform screening of blood products, as well as improvements in infection control practices in hospitals and health centers, it would be expected that the numbers and prevalence of acute viral hepatitis should decline. In the present study conducted 10 years after our first report, the prevalence of acute HAV, HBV, HCV and HDV infections and their time trend changes were investigated.
Consecutive patients clinically diagnosed as having acute viral hepatitis from January 2012 to December 2014, at the Department of Infectious Diseases, National Center for Communicable Diseases, Mongolia, were studied. Patients were included if they fulfilled the following inclusion criteria: (1) an acute illness presenting with signs or symptoms such as jaundice, dark urine, fatigue, anorexia, nausea, vomiting and fever, and (2) a serum alanine aminotransferase (ALT) level that was at least two and onehalf times the upper limit of normal. Patients were excluded if they had alcohol-related liver disease (determined by questioning of the patient and family members) and those with a history of exposure to hepatotoxic drugs or chemicals. Autoimmune liver disease was not positively excluded since testing for autoimmune markers was not available.
Approval of the study was obtained from the ethics committees of the Ministry of Health of Mongolia and from the Human Research Ethics Committees of the Health Sciences University of Mongolia, Ulaanbaatar, Mongolia. The study was conducted in accordance with the international ethical guidelines of the International Conference on Harmonization Guidelines for Good Clinical Practice [10].
The following data were collected at the index diagnostic visit: sex, age, risk factor and routine laboratory tests. The latter, including ALT, aspartate aminotransferase (AST) and bilirubin levels were determined by routine laboratory techniques.
Sera from the patients were tested for IgM antibody against HAV (anti-HAV IgM) and for total antibodies against HAV (anti-HAV [total]) by enzyme-linked immunosorbentassay (ELISA) (HAM(N)-EIA and HAT-EIA, respectively; Denkaseiken, Tokyo, Japan). HBsAg was assessed by ELISA (IMxHBsAg Assay System; Abbott Japan, Tokyo, Japan); antibodies to HBsAg (anti-HBs) and antibodies to HBV core (anti-HBc [total]) was by passive hemagglutination with commercial assay kits (Mycell II anti-HBs and Mycell anti-HBc, respectively; Institute of Immunology Co. Ltd, Tokyo, Japan). Anti-HCV antibodies were assayed by the hemagglutination method (Abbott HCV-PHA; Abbott Japan). The presence of antibodies to HDV-IgM antibodies was determined by ELISA. Due to resource constraints, no other serological test or viral quantification was available. Serum HCV RNA was measured by a COBAS Amplicor HCV Test (Roche Diagnostics, Branchburg, NJ, USA) (<600 IU/mL).
Acute hepatitis A infection was defined as positive anti-HAV IgM Ab, and acute hepatitis B was defined by a positive IgM anti-HBc and circulating levels of HBsAg. Acute HCV infection was identified by positive HCV RNA in serum with either negative or positive anti-HCV antibody. Patients with negative anti-HCV antibody and positive HCV RNA were considered as acute hepatitis C cases. Patients with positive anti-HCV antibody and positive HCV RNA were considered acute hepatitis C cases only if ALT values were >10 times upper limit of normal and there was a recent history of possible high-risk exposure to HCV (e.g., surgical procedure). HBV/HDV co-infection was diagnosed by positive HDV-IgM antibodies in patient with acute hepatitis B (positive IgM anti-HBc and circulating levels of HBsAg), and acute HDV superinfection was defined by positive HDV-IgM antibodies in patients chronically infected with hepatitis B (positive IgG anti-HBc and HBsAg >6 months).
We conducted a time trend analysis investigating the change in proportion of acute HCV, HBV and HDV infection among the cohort with respect to our previous study published a decade earlier [6].
Quantitative data are expressed as mean┬▒standard deviation (SD) and categorical data by number (percentage) of patients. For skewed variables, the data are presented as median and range. The Student t -test or non-parametric, i.e. Wilcoxon-Mann-Whitney U -test or Kruskal-Wallis tests were used to compare quantitative data, while appropriate. Žć2 and Fisher-exact tests were used for the comparison of frequency data and to evaluate the relationships between groups. All tests were two-tailed and P -values <0.05 were considered significant. All analyses were carried out using the statistical software package SPSS for Windows, version 21 (SPSS Inc., Chicago, IL, USA).
The cohort comprised 546 consecutive patients with acute viral hepatitis. The mean age was 17.6 (SD, 10.3) years with 249 (54.8%) male. The mean ALT at presentation was 1,800.6 IU/L (SD, 902.3), AST 1,152.4 IU/L (SD, 518.3) and total bilirubin 6.8 mg/dL (SD, 3.1). The characteristics of the cohort are shown in Table 1.
Acute hepatitis A (anti-HAV IgM-positive) was diagnosed in 278 (50.9%), acute hepatitis B (anti-HBc IgM-positive and 6 months follow-up HBsAg negative) in 143 (29.2%) and acute hepatitis C in 33 (6.0%) patients. Ninety two (16.8%) patients had either coinfection or super-infection with two hepatitis viruses.
The mean age of patients infected with acute hepatitis A was lower than those with acute hepatitis B or C (11.6┬▒5.9, 22.9┬▒5.2 and 32.2┬▒10.3, respectively, P<0.001) (Table 1). Hepatitis A was the most prevalent cause of acute viral hepatitis among 2-19 year olds, while acute hepatitis B and superinfection with HDV were the most prevalent causes among the 20-40 year age group. Interestingly, hepatitis C was the most prevalent acute viral hepatitis among 40-49 year old patients (Table 2). We did not observe differences in the prevalence of acute hepatitis when stratified according to gender.
The AST, ALT and bilirubin levels at presentation were significantly higher among patients with acute hepatitis B than among those with acute hepatitis C or A (P<0.001, for all) (Table 1).
Blood transfusion and surgery were significantly more frequent as possible risk factors for acute hepatitis C acquisition (P<0.001), while patients with acute hepatitis B were more likely to have one or more of the possible risk factors for acquiring blood-borne hepatitis viruses, including tattooing, dental care, or folk remedies such as acupuncture and bloodletting within 6 months before the onset of disease or being a health care worker. Patients with acute hepatitis B were significantly more likely to have tattooing and family histories of hepatitis as risk factors than those with acute hepatitis C or A (P<0.001, for both) (Table 1).
Of the overall cohort, 92 (16.8%) patients had either co-infection or super-infection with two hepatitis viruses. There were 59 (10.8%) patients who had detectable HBsAg and anti-HDV but who were negative for both IgM anti-HAV and IgM anti-HBc, suggesting that they had developed superinfection with HDV. Unfortunately, HBeAg status was not known, due to lack of testing resources, nor was HDV RNA testing available. Thus, in this group, it is not possible to definitely exclude HBV reactivation in a HBV-HDV co-infected patient.
Patients with more than one hepatitis virus infection were significantly older, more likely to be male and had a higher prevalence of all risk factors for disease acquisition. These patients also had more severe liver disease at presentation with higher AST, ALT and bilirubin levels compared to those with mono-infection (Table 3).
Those with superinfection of HDV had higher bilirubin levels than all other types of co- and superinfection, and were significantly more likely to be male. No difference in risk factors was observed between different groups (Table 4).
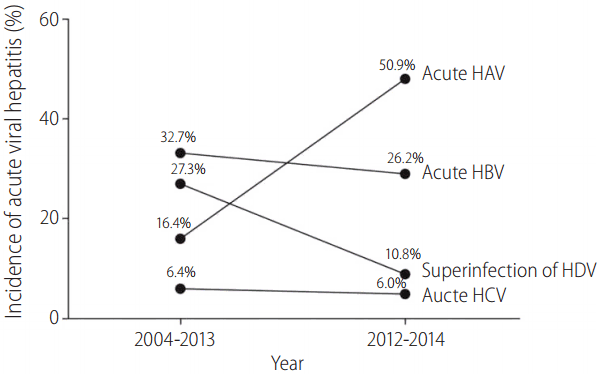
We undertook time trend analysis between our initial study published in 2006, and the present cohort (2012-2014). As shown in Fig. 1, we observed a significant decline in the proportion of HDV superinfection (from 27% to 11%, P=0.001), while those of acute HCV and HBV infection changed from 6% to 6% and from 33% to 26%, respectively. There was a significant increase in the incidence of acute hepatitis A (from 16% to 26%, P<0.001).
We have described the epidemiology of acute viral hepatitis in Mongolia between 2012 and 2014. We also undertook time trend analysis with similar data obtained a decade earlier and previously published [6]. The most important outcome of the present study is the knowledge that Mongolia still has an ongoing epidemic of acute viral hepatitis. Despite significant changes in socioeconomic parameters, for example, increasing school enrollment by 117% from 1994 to 2012 and an increase in Gross National Income per capita, from 0.3 to 3.77 US dollars [11], the epidemic of acute viral hepatitis remains largely not decreased. Thus, apart from HDV infection, there was no significant decline in the number of cases with acute hepatitis due to the other viruses, and there was a significant increase in HAV infection.
The clinical illness in Mongolia is manifested across essentially all population groups, attesting to the pervasive nature of the epidemic, something that is by and large nonexistent in most other parts of the world. Notably, 16.8% of patients presented with either co-infection or super-infection with two hepatitis viruses, a clinical scenario that is rare in developed nations. As expected, those with more than one infection had clinically more severe disease at presentation manifested by higher levels of ALT, AST and serum bilirubin.
Despite being a major cause of acute hepatitis in many countries [11], the relative contribution of injecting drug use (IDU) to viral hepatitis burden in Mongolia was significantly lower than in other regions. In an international study including 57 countries, HCV prevalence was reported to be at least 50% among IDUs in 49 countries. However, available regional estimates varied widely; it was up to 96% in Eastern Europe and Central Asia, 93% in East-Asia and the Pacific, between 25 to 88% in Australia and New Zealand, and from 2 to 93% in Western Europe [12]. In our study, IDU was virtually nonexistent. In Mongolia, viral hepatitis transmission is mainly associated with medical exposures (dental care, surgery and blood transfusion), which were responsible for 40% of all infections. Blood transfusion and surgery were more prevalent risk factors among patients with acute HCV, while tattooing, dental care, acupuncture and bloodletting were the main risk factors for acute hepatitis B (Table 1). No difference in risk factors was observed among the different categories of patients with more than one infection (Table 4).
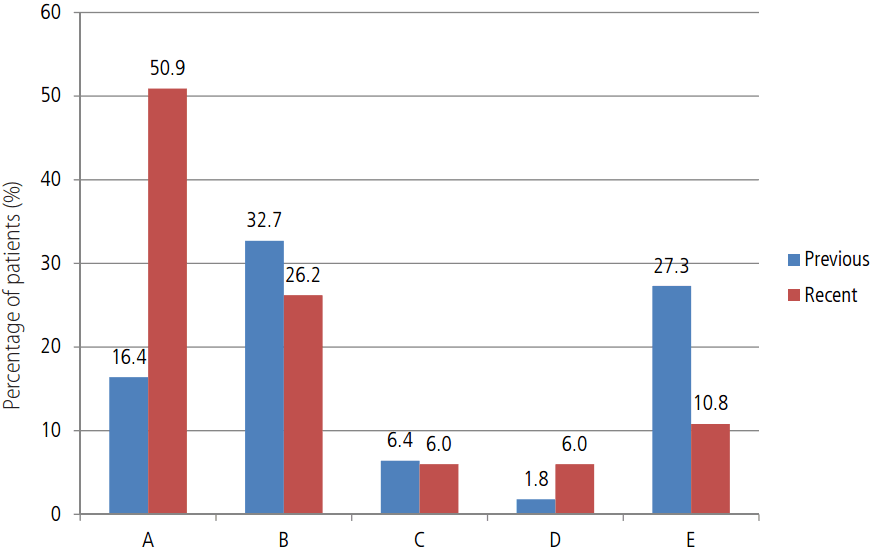
The results of our time trend analysis suggest that there are varying rates of decline of the three blood-borne hepatitis viruses (HBV, HCV, and HDV). While there was a dramatic decline in HDV superinfection (27.3% to 10.8%, P=0.001) over the last decade, contrary to expectations, there appeared to only be a small decline (32.7% to 26.2%, and 6.4% to 6.0%, respectively) in HCV and HBV incidence over time (Fig. 2). There were just 26 cases of acute HBV infection in the 0-19 age group in the present cohort. In Mongolia, mass hepatitis B vaccination program was initiated in 1991 with 98-100% of all live babies receiving vaccination within 24-48 hours after birth. However, even today, there is no universal testing of blood in all centers since patients have to pay for blood screening. Thus, the influx of uninfected birth cohorts, and improved medical hygiene may partially explain the small decline in HCV and HBV incidence.
Unexpectedly, there was a significant increase in the incidence of acute HAV infection compared to our previous study (from 16.4% to 50.9%, P<0.001). This observation is not completely explained; however, it may reflect community outbreaks as previously reported in 2007 [13]. Another possibility is that parents are now more aware when children develop acute hepatitis and therefore seek medical advice. Thus, the increase in the proportion of cases attributable to HAV may represent a change in the percentage of cases seeking medical attention rather than changes in overall disease incidence. This is also consistent with data from a large survey of 520 apparently healthy 7-12 year olds, suggesting that the prevalence of anti-HAV antibodies in Mongolia is 85% [14].
This study has also limitations as an epidemiologic study. In addition, it is sometimes hard to recognize the exact timing of infection such as HCV infection. Our findings clearly suggest that much more rigorous and stringent infection control public health policies and measures are required across Mongolia to eliminate incident acute viral hepatitis. Many of these policies are easy to implement including basic infection control practices across medical and dental facilities, as well as, for example, the use of single use needles, syringes and equipment during acupuncture or tattooing. While local resource limitations are an issue, international health agencies can provide much-needed expertise and help to construct and implement policies to reduce viral hepatitis transmission.
We agree that only a national epidemiologic study can provide a definitive answer regarding the overall prevalence and incidence of viral hepatitis and dynamic trends over time in Mongolia. Lacking such a national sample survey, our study from the national viral hepatitis treatment center provides a useful secondary measure.
In conclusion, acute viral hepatitis is still prevalent in Mongolia. There is an urgent need for efforts to improve infection control measures in the medical system. The results from this large cohort of patients with acute hepatitis in the National Center for Communicable Diseases in Ulaanbaatar, Mongolia, will improve our understanding of current measures for hepatitis control, and suggest where new resources need to be deployed.
FOOTNOTES
Financial support
JG is supported by the Robert W. Storr Bequest to the Sydney Medical Foundation, University of Sydney; a National Health and Medical Research Council of Australia (NHMRC) Program Grant (1053206), and a Project grant (1047417), and the Sydney West Translational Cancer Research Centre Partner Program funded by the Cancer Institute NSW. ME is supported by an International Postgraduate Research Scholarships (IPRS) and an Australian Postgraduate Award (APA) of the University of Sydney. This study was also supported by Yonsei Liver Blood Bank (YLBB) funded by Sanofi Aventis Korea.
Figure┬Ā1.
Changes in the proportion of acute hepatitis A, B, C and D in Mongolia. HAV, hepatitis A virus; HBV, hepatitis B virus; HDV, hepatitis delta virus; HCV, hepatitis C virus.
Figure┬Ā2.
The comparison of proportion in acute viral hepatitis with previous data. (A) acute hepatitis A, (B) acute hepatitis B, (C) acute hepatitis C, (D) co-infection with HBV and HDV, (E) HDV superinfection.
Table┬Ā1.
Comparison of the characteristics of the different acute hepatitis (A, B and C) virus infection
| Feature | Total (n=454) | Acute hepatitis A (n=278) | Acute hepatitis B (n=143) | Acute hepatitis C (n=33) | P-value |
|---|---|---|---|---|---|
| Age (years) | 17.6┬▒10.3 | 11.6┬▒5.9 | 22.9┬▒5.2 | 32.2┬▒10.3 | <0.001 |
| Male | 249 (54.8) | 149 (53.6) | 84 (58.7) | 16 (48.5) | 0.311 |
| Risk factors* | |||||
| ŌĆāBlood transfusion | 16 (3.5) | 1 (0.4) | 9 (6.3) | 6 (18.2) | <0.001 |
| ŌĆāTattooing | 53 (11.7) | 3 (1.1) | 48 (33.6) | 2 (6.1) | <0.001 |
| ŌĆāSurgery | 74 (16.3) | 8 (2.9) | 47 (32.9) | 19 (57.6) | <0.001 |
| ŌĆāDental care | 102 (22.5) | 7 (2.5) | 78 (54.5) | 17 (51.5) | <0.001 |
| ŌĆāAcupuncture | 9 (2.0) | 0 | 9 (6.3) | 0 | <0.001 |
| ŌĆāFamily history | 17 (3.7) | 3 (1.1) | 13 (9.1) | 1 (3.0) | 0.004 |
| Laboratory data | |||||
| ŌĆāTotal bilirubin (mg/dL) | 6.8┬▒3.1 | 4.5┬▒2.4 | 7.3┬▒3.1 | 6.3┬▒3.9 | <0.001 |
| ŌĆāALT (IU/L) | 1800.6┬▒902.3 | 1537┬▒651.7 | 2250.5┬▒782.0 | 1719.3┬▒997.2 | <0.001 |
| ŌĆāAST (IU/L) | 1152.4┬▒518.3 | 973.5┬▒450.8 | 1160.1┬▒412.8 | 1021.4┬▒621.5 | <0.001 |
Table┬Ā2.
Age-dependent prevalence of acute viral hepatitis in Mongolia
Table┬Ā3.
Comparison of baseline characteristics of patients with mono-infection and those with coinfection or superinfection
Table┬Ā4.
Comparison of characteristics between co-infection and superinfection of HBV and HDV
Abbreviations
ALT
alanine aminotransferase
anti-HAV IgM
IgM antibody against HAV
anti-HBc
antibodies to HBV core
anti-HBs
antibodies to HBsAg
AST
aspartate aminotransferase
ELISA
enzyme-linked immunosorbentassay
HAV
hepatitis A virus
HBV
hepatitis B virus
HCV
hepatitis C virus
HDV
hepatitis D virus
IDU
injecting drug use
SD
standard deviation
REFERENCES
1. World Health Organization (WHO). Hepatitis B. Fact sheet number 204, revised October 2000. WHO web site, <http://www.who.int/mediacentre/factsheets/fs204_Jul2014/en/>. Accessed 2013.07.06.
2. World Health Organization (WHO). Hepatitis C. Fact sheet number 164, revised October 2000. WHO web site, <http://www.who.int/mediacentre/factsheets/fs164_apr2014/en/>. Accessed 2013.07.06.
4. Baatarkhuu O, Kim DY, Ahn SH, Nymadawa P, Dahgwahdorj Y, Shagdarsuren M, et al. Prevalence and genotype distribution of hepatitis C virus among apparently healthy individuals in Mongolia: a population-based nationwide study. Liver Int 2008;28:1389-1395.


5. World Health Organization (WHO). The Work of WHO in the Western Pacific Region 2003ŌĆō2004. Report of the Regional Director to the Regional Committee for the Western Pacific, Statistical Annex. Manila: World Health Organization, Regional Office for the Western Pacific; 2003. p. 190-205.
6. Tsatsralt-Od B, Takahashi M, Endo K, Buyankhuu O, Baatarkhuu O, Nishizawa T, et al. Infection with hepatitis A, B, C, and delta viruses among patients with acute hepatitis in Mongolia. J Med Virol 2006;78:542-550.


7. Baatarkhuu O, Kim DY, Bat-Ireedui P, Han KH. Current situation of hepatocellular carcinoma in Mongolia. Oncology 2011;81 Suppl 1:148-151.


8. Jazag A, Puntsagdulam N, Chinburen J. Status quo of chronic liver diseases, including hepatocellular carcinoma, in Mongolia. Korean J Intern Med 2012;27:121-127.



9. Baatarkhuu O, Kim DY, Nymadawa P, Kim SU, Han KH, Amarsanaa J, et al. Clinical features and prognosis of hepatocellular carcinoma in Mongolia: a multicentre study. Hepatol Int 2012;6:763-769.


10. Dixon JR Jr. The international conference on harmonization good clinical practice guideline. Qual Assur 1998;6:65-74.


11. The World Bank. GDP (current US$). The World Bank web site <http://data.worldbank.org/indicator/NY.GDP.MKTP.CD>. Accessed 2014.09.19.
12. Aceijas C, Rhodes T. Global estimates of prevalence of HCV infection among injecting drug users. Int J Drug Policy 2007;18:352-358.


- TOOLS



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




