INTRODUCTION
Hepatic hydrothorax is defined as a significant pleural effusion, usually greater than 500 mL, in a patient with liver cirrhosis but no primary cardiac or pulmonary diseases.
1,
2 In patients with hepatic hydrothorax, even a modest volume of pleural fluid can cause respiratory symptoms, in contrast to ascites, where a large volume can be well tolerated.
Hepatic hydrothorax is a relatively uncommon complication of end-stage liver with an estimated prevalence of 5-10% among cirrhotic patients.
1-
4 In spite of the numerous case reports describing clinical features and treatments for hepatic hydrothorax the current knowledge of this complication of cirrhosis is limited. Unfortunately the pathogenesis and therapy of hepatic hydrothorax have not been appropriately studied, therefore, proper guidelines on therapy based on good evidence have been difficult to establish.
The aim of current study is to examine the treatment outcomes of the 11 patients with refractory hepatic hydrothorax who underwent chemical pleurodesis with or without video-assisted thoracoscopic surgery (VATS).
PATIENTS AND METHODS
Patients
Eleven consecutive patients with refractory hepatic hydrothorax and decompensated liver cirrhosis who underwent chemical pleurodesis with or without VATS in our institution between July 2007 and February 2011 were included. The Child-Turcotte-Pugh (CTP) class and etiology of cirrhosis was documented in all patients. All 11 patients had failed medical treatment because diuretics and other modes of therapy had not led to symptomatic improvement or caused electrolytes disturbances or impairment in renal function. Average diuretic dosages in the study population were spironolactone 200 mg/day and furosemide 80 mg/day. Of 11 patients, 10 (90.9%) had coexisting ascites and the amount of ascites examined by abdominal ultrasonography were small in 9 patients and moderate in 1 patient, respectively. The diuretic treatment applied to these patients for the management of symptomatic hydrothorax was sufficient for the control of ascites. A medical history, physical examination, laboratory findings, and radiologic imagings including chest X-ray, and computed tomographic (CT) scans of the chest and abdomen were thoroughly reviewed. Echocardiography was performed to measure left ventricular function in all 11 patients. Abdominal ultrasound or CT scans were performed in all 11 patients to examine the liver for size and structure and to determine the presence of ascites. Simultaneously with abdominal ultrasound, a Doppler study for patency of, and flow in, the hepatic and portal venous systems was also performed. Prior to the chemical pleurodesis with or without VATS, a diagnostic thoracentesis (approximately 40-50 mL of pleural fluid) was performed in all 11 patients for confirmation of diagnosis and to exclude alternative diagnoses. Diagnostic tests of pleural fluid to be ordered included cell count, Gram stain and culture of the pleural fluid in blood culture bottles and fluid protein, albumin, lactative dehydrogenase and bilirubin. Other tests in pleural fluid that would be appropriate depending on the clinical circumstance include triglyceride, pleural pH, adenosine deaminase, and polymerase chain reaction (PCR) for mycobacterium, amylase, and cytology to exclude chylothorax, empyema, tuberculosis, pancreatitis and malignancy, respectively. This study was conducted in accordance with the principles of the Declaration of Helsinki. Our study protocol obtained approval of the Ethics Committee of Kangbuk Samsung Hospital.
VATS and chemical pleurodesis
VATS was performed in two patients with the intent to provide chemical pleurodesis. The remaining 9 patients underwent only chemical peurodesis by tube thoracostomy. The agents used in chemical peurodesis were talc in 3 patients (27.3%), taurolidine in 2 patients (18.2%) and viscum album in 6 patients (54.25%). The processes of chemical pelurodesis were performed as previously defined.
5 VATS operative procedure was as follows. Patients received general anesthesia. The rigid pleural effusion was drained using by a 5-mm rigid thoracoscopy. It was sent for body fluid chemistry, cytology and culture. If the fenestrations of hemidiaphragm were noticed during the thoracoscopic observation, they were closed using thoracoscopic technique with suture. The insufflated slcerosing agents used in VATS were talc (20 g)+bleomycin (30 mg) in one patient and taurolidine (10 g)+bleomycin (30 mg) in another patient.
Definitions
Successful pelurodesis was defined by a patient who had no longer dyspnea symptoms and had a chest roentgenogram that did not show pleural effusion at 1 month after the termination of VATS with chemical pleurodesis or chemical pleurodesis only. The definition for refractory hepatic hydrothorax has not been properly established. We adopted the definition of refractory hepatic hydrothorax as a symptomatic hepatic hydrothorax which was refractory to conventional treatments for hepatic hydrothorax, such as salt restriction, maximum tolerable dosage of diuretics, repeated thoracentesis and pleural drainage. Procedure related morbidity and mortality were defined as the occurrences of any complication and/or death within 1 month relevant to the adoption of chemical pleurodesis with or without VATS for the management of refractory hepatic hydrothorax in our enrolled patients.
Statistical methods
Mann-Whitney U-test for continuous variables and Fisher's exact test for categorical variables were performed. The comparison of the cumulative survival rate between success and non-success group was performed using a log-rank test with Kaplan-Meier analysis. Data were analyzed using PASW software package (version 18.0; SPSS, Inc., Chicago, IL, USA). P-values less than 0.05 were considered significant.
RESULTS
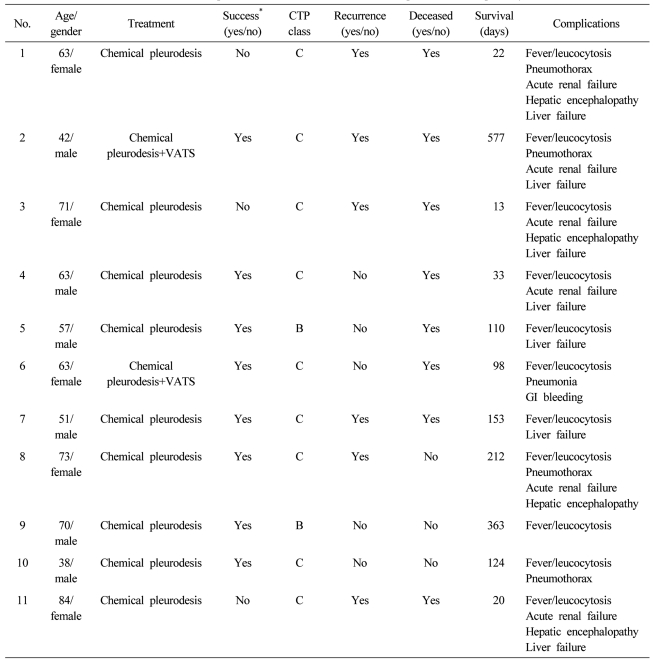
A total of 11 patients with refractory hepatic hydrothorax were included. Median (range) age of the patients was 63 (38-84) years and 6 (54.5%) patients were male. The etiologies of liver cirrhosis were HBV (n=4, 36.2%), HCV (n=4, 36.4%), alcoholic (n=1, 9.1%) and cryptogenic (n=2, 18.2%). Nine (81.8%) and 2 patient were CTP class C and B, respectively. All the patients have right sided pleural effusion refractory to medical therapy. The median (range) sessions of chemical pleurodesis were 3 (2-10) times. Nine patients underwent only chemical pleurodesis via chest tube and two patients underwent chemical pleurodesis with VATS (
Table 1). During the chemical pleurodesis with or without VATS, tolerable medical treatments such as low salt diet, diuretics and chest tube drainage were maintained. Diaphragmatic defects were not observed in the patients during the VATS procedure.
Successful pleurosdesis was achieved in 8 of 11 patients (72.7%,
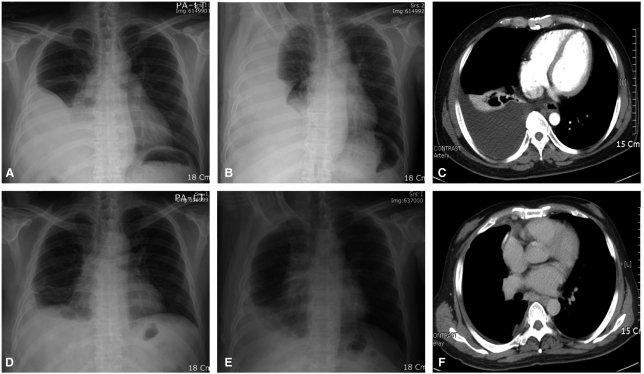
Table 1). Representative radiologic findings of chest X-ray and CT were shown in
Figure 1. By Kaplan-Meier analysis with log rank comparison, the overall survival was significantly longer in the success group than the non-success group (
P<0.01). Five (62.5%) of the patients in the success group remained asymptomatic and hydrothorax free at a median follow-up of 16 (2-52) weeks after the procedure. However, three patients experienced symptomatic and delayed recurrence of hydrothorax 16, 30, and 32 weeks after the initial procedure. Complications occurred were low grade fever/leukocytosis (n=11, 100.0%), pneumonia (n=1, 9.1%), pneumothorax (n=4, 36.4%), azotemia/acute renal failure (n=6, 54.6%) and hepatic encephalopathy (n=4, 36.4%). Chest pain and percutaneous drainage (PCD) site pain and chest pain occurred in all the 11 patients during the chemical pelurodesis. Five patients (45.5%) were suspected to have procedure-related mortality due to the occurrence of acute renal failure (
Table 2).
DISCUSSION
This study was as retrospective observational results in the patients with refractory hydrothorax and cirrhosis. The current study adds a report of 11 patients to the previously published series of chemical pleurodesis
6-
8 that examined the role of chemical pleurodesis with or without VATS for the management of refractory hepatic hydrothorax. The initial success of treatment was achieved in 8 of 11 patients (72.7%) which was comparable to the treatment result of previously published case series.
8 Notably, the cumulative survival rate was significantly higher in patients who achieved the initial success of treatment than patients without the initial success of treatment. The patients with initial treatment failure naturally underwent lesser sessions of chemical pleurodesis compared to the patients with successful treatment (albeit statistically insignificant) because the condition of a patient deteriorated, further session of chemical pleurodesis could not be performed.
Ablation of the space between the parietal and visceral pleura with a sclerosing agent injected by tube thoracostomy has been used to treat hepatic hydrothorax.
3 The procedure is generally unsuccessful, probably because fluid is formed too rapidly to allow the visceral and parietal pleural surfaces to approximate and adhere.
6 Often, incomplete adhesions form between the parietal and visceral pleura, resulting in the formation of loculated pleural effusions. Thoracentesis does not evacuate all the fluid, because all locules cannot be aspirated. In addition, several complications may be associated with placement of a chest tube, including subcutaneous emphysema; lung, spleen, liver, and stomach laceration; unilateral pulmonary edema from rapid removal of fluid; and placement of the tube into the abdominal cavity. In the current study, complications associated with placement of a chest tube occurred in all enrolled patients, and most devastating complications were severe fluid and electrolytes imbalance and protein depletion leading the azotemia/acute renal failure. Five patients (45.5%) were suspected to have procedure-related mortality due to the occurrence of these devastating complications (
Table 2). Milder complications such as fever/leucocytosis occurred in all enrolled patients, and a significant proportion of patients with refractory hepatic hydrothorax who had treated with chemical pleurodesis only via a chest tube and chemical pleurodesis+VATS experienced more severe complications such as pneumonia (n=1, 9.1%), pneumothorax (n=4, 36.4%), azotemia/acute renal failure (n=6, 54.6%) and hepatic encephalopathy (n=4, 36.4%,
Table 2). Morbidity including PCD site pain and chest pain during chemical pleurodesis occurred in all the patients. Because of low success rates and procedure-related complications, chest tube drainage of the pleural space for the routine treatment of hepatic hydrothorax is not recommended. However, the patients with advanced liver cirrhosis presenting with a unilateral pleural effusion, most commonly in the right side, usually present with symptoms related to shortness of breath, cough, hypoxemia and/or chest discomfort. Unlike the ascites, relief of symptoms and prevention of pulmonary complications and infections are critical for the patients with refractory hepatic hydrothorax who are potential candidates for liver transplantation. Conventional treatment modalities for hepatic hydrothorax such as sodium restriction and diuretics, and intermittent thoracentesis cannot achieve the goals of treatment in many patients. Increasing the doses of diuretics to achieve the negative sodium balance may increase in the serum creatinine level, which indicates a decrease in the glomerular filtration rate.
9 Moreover, increasing the doses of diuretics may precipitate hepatic encephalopathy. Although, thoracentesis for hepatic hydrothorax in clinical practice is both useful and safe, thoracentesis dependence may be associated with deteriorating clinical status and impaired quality of life. Actually, when thoracentesis is required every 2-3 weeks, alternative strategies were needed to be considered.
Transjugular intrahepatic portosystemic shunt (TIPS) were reported to have encouraging results in patients of hepatic hydrothorax with or without ascites.
10-
12 However, most of patients with TIPS for the treatment of hepatic hydrothorax developed hepatic encephalopathy and had the deterioration of liver function and subsequently died within 6 weeks of shunt placement.
12 Another study also evaluated the clinical efficacy of TIPS in patients with hepatic hydrothorax and this study reported poor prognosis in patients with hepatic hydrothorax who underwent TIPS.
13 In our institution, TIPS was not available and we did not consider this treatment modality for our enrolled patients with refractory hepatic hydrothorax. Hence, we could not compare the treatment results between chemical pleurodesis with or without VATS and TIPS in patients with refractory hepatic hydrothorax.
There are a few reports of successful primary repair of diaphragmatic defects.
8,
14 Evaluation for defects in the diaphragm formerly required a thoracotomy, which carries substantial risks. Recently, VATS was used to identify diaphragmatic defects in patients with refractory hepatic hydrothorax, and biologic glue or sutures were used to close the defects.
15 However, VATS also required a general anesthesia, which carries substantial risks for the patients with advanced liver cirrhosis. Additionally, in contrast to the series by Mouroux et al,
15 we did not identify any visible diaphragmatic defects in two patients who underwent chemical pelurodesis combined with VATS.
In a real clinical situation, where advanced liver cirrhosis patients with refractory hepatic hydrothorax who need symptomatic relief and the control of pulmonary complications are in the care of a physician who has a few available treatment modalities, chemical pleurodesis by tube thoracostomy may be a considerable treatment option for these intractable patients.
In the current study, success of treatment was identified as a significant prognostic indicator for patients with advanced liver cirrhosis and refractory hepatic hydrothorax. Previous study by Ferrante et al
8 also adopted the concept of success of treatment defined as control of symptomatic hydrothorax in the first 30 days after the VATS with talc pleurodesis. However, they did not figure out any prognostic significance of this concept "success of treatment". However, in the current study, the patient who achieved the success of treatment showed significantly better outcomes than the patients who did not achieve the success of treatment.
The current study has limitations to consider. First, the number of enrolled patients with refractory hepatic hydrothorax is too small to draw any statistical significance. Due to the rare frequency of hepatic hydrothorax, previously reported case series
6-
8 rarely included more than 20 patients with hepatic hydrothorax. Second, we could enroll only 2 cases with chemical pleurodesis with VATS due to the practical and psychological burdens of general anesthesia in patients with decompensated liver diseases. Hence, we could not evaluate the clinical efficacy of chemical pleurodesis with VATS to reduce the performed sessions of pleurodesis compared to the procedure without VATS.
In summary, this series suggests that refractory hepatic hydrothorax can be controlled with multiple sessions of chemical pleurodesis via chest tube with or without VATS in as many as 72.7% of patients. Additionally, the patients who achieved the success of treatment defined as symptoms free and no pleural effusion at 1 month after the termination of chemical pleurodesis and/or VATS, showed significantly better outcomes than those who did not achieve the success of treatment. However, due to a significantly high occurrence rate of procedure-related morbidity and mortality hindered the routine application of this procedure for patients with this debilitating complication of end stage liver disease. Further options of management, such as TIPS or chemical pleurodesis combined with nasal continuous positive airway pressure (CPAP)
16 may be required for effective treatment for these patients. However, warrant future validation is needed.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



