| Korean J Hepatol > Volume 16(4); 2010 > Article |
ABSTRACT
Background/Aims
Few studies have investigated hepatitis A virus (HAV) seroepidemiology in Koreans with chronic liver disease (CLD). This study compared the prevalence of IgG anti-HAV between the general healthy population and patients with hepatitis B virus-related CLD (HBV-CLD), with the aim of identifying predictors of HAV prior exposure.
Methods
In total, 1,319 patients were recruited between June 2008 and April 2010. All patients were tested for IgG anti-HAV, hepatitis B surface antigen (HBsAg), and antibodies to hepatitis C virus. The patients were divided into the general healthy population group and the HBV-CLD group based on the presence of HBsAg. The seroprevalence of IgG anti-HAV was compared between these two groups.
Results
The age-standardized seroprevalence rates of IgG anti-HAV in the general healthy population and patients with HBV-CLD were 52.5% and 49.1%, respectively. The age-stratified IgG anti-HAV seroprevalence rates for ages ≤19, 20-29, 30-39, 40-49, 50-59, and ≥60 years were 14.3%, 11.2%, 45.5%, 90.5%, 97.6% and 98.3%, respectively, in the general healthy population, and 0%, 9.8%, 46.3%, 91.1%, 97.7%, and 100% in the HBV-CLD group. In multivariate analysis, age (<30 vs. 30-59 years: OR=19.339, 95% CI=12.504-29.911, P<0.001; <30 vs. ≥60 years: OR=1060.5, 95% CI=142.233-7907.964, P<0.001) and advanced status of HBV-CLD (OR=19.180, 95% CI=4.550-80.856, P<0.001) were independent predictors of HAV prior exposure.
Hepatitis A is usually self-limited and has a benign clinical course. It is usually asymptomatic in children, whereas it causes clinically apparent disease in the majority of adults, rarely progressing to fulminant hepatic failure.1 In particular, the old age and chronic liver disease (CLD) have been known to be risk factors for fulminant hepatic failure.2 Therefore, hepatitis A virus (HAV) vaccination is recommended for patients with CLD.
Improvements in the socioeconomic status and general public health of Korea have led to a shift in the seroprevalence of hepatitis A from hyperendemic region to lower one.3 Paradoxically, the number of children and young adults who are susceptible to HAV infection has been gradually increased, resulting in the recent rapid rise of symptomatic hepatitis A. Currently, hepatitis A has become one of the most common causes of acute viral infection in Korean adults.3,4 Although the prevalence of chronic hepatitis B virus (HBV) infection has been declined after the introduction of universal vaccination, it is still the most important cause of CLD in Korea.5 Accordingly, the seroprevalence of hepatitis A in patients with HBV-related CLD has been of interest. However, there are few studies about HAV seroepidemiology in Korean population with CLD,6,7 and no studies have compared the seroprevalence of IgG anti-HAV between general healthy population and patients with HBV-related CLD.
This study was aimed to evaluate the seroprevalence of IgG anti-HAV in Korean patients with HBV-related CLD and analyzed it as compared with general healthy population. Thus, we attempted to provide the objective data about the HAV vaccination strategy in patients with HBV-related CLD and to identify the predictors of prior HAV exposure.
We retrospectively analyzed the medical records of 1,319 patients aged 15 years or older who underwent tests for hepatitis B surface antigen (HBsAg), anti-hepatitis C virus antibody (anti-HCV) and IgG anti-HAV, between June 2008 and April 2010. Of them, 622 patients who were negative to both HBsAg and anti-HCV and had no past history of other liver diseases such as alcoholic hepatitis and autoimmune hepatitis were classified as general healthy population. Remaining 697 patients who were positive to HBsAg regardless of the presence of anti-HCV were classified as those with HBV-related CLD. The seroprevalence of IgG anti-HAV was evaluated according to age and sex at the time of laboratory tests and compared between the general healthy population and the patients with HBV-related CLD.
The severity of liver disease was classified as follows: 1) inactive HBsAg carrier was defined as those who were positive to HBsAg for more than six months, were negative to hepatitis B e antigen, had a serum level of HBV DNA<104 copies/mL and persistently had normal levels of serum ALT for more than one year, 2) chronic hepatitis B (CHB) patient was defined as those who had a serum level of HBV DNA>104 copies/mL, had serum ALT level elevated persistently or intermittently and had a lack of evidences demonstrating the presence of liver cirrhosis and hepatocellular carcinoma,8 3) liver cirrhosis was diagnosed by histology or ultrasonographic/CT imaging features, supplemented with clinically relevant portal hypertension (esophageal and/or gastric varices, ascites, splenomegaly with a platelet count of<100,000/mm3) or hepatic encephalopathy, 4) hepatocellular carcinoma was made according to the guidelines of American Society for the Study of Liver Diseases, which was based on the imaging study and serum alpha-fetoprotein levels.9
The current study was conducted in compliance with the declaration of Helsinki and approved by the Institutional Review Board (IRB) of our medical institution.
Student's t-test for continuous variables, and chi squared analysis, Fisher's exact test for categorical variables were used where appropriate. Age standardization was done using the direct methods where the 2005's statistics of Korean population were served as the standard population. Then, the prevalence of IgG anti-HAV was evaluated using Cochran-Mantel-Haenszel methods. To identify the factors affecting the HAV prior exposure, a multivariate logistic regression analysis was performed using variables such as age, sex, the presence of HBsAg and the severity of liver diseases. On multivariate analysis, the age of the patients were divided into three groups (<30, 30~59, ≥60) and the severity of liver diseases were divided into two groups. Patients with liver cirrhosis or hepatocellular carcinoma were classified as severe cases. General healthy population, inactive HBsAg carrier and CHB patients were classified as mild cases. A value of P<0.05 was considered statistically significant. Statistical analysis was performed using SPSS version 18.0 (Chicago, IL, USA).
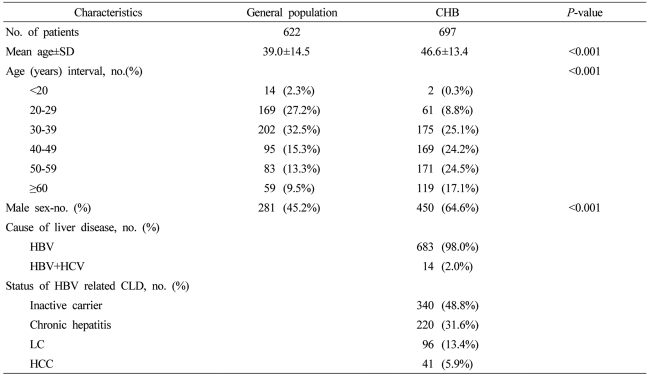
The number of general healthy population and that of patients with HBV-related CLD were 622 and 697, respectively. Mean age was 39 years (14.5 years, S.D.) and 46.6 years (13.4 years, S.D.), respectively. The proportion of male patients was 45.2% (281/622) and 64.6% (450/697), respectively. There were significant differences in age and sex between the two groups (P<0.001). Of patients with HBV-related CLD, a total of 14 patients had a superinfection with chronic hepatitis C (Table 1).
The overall positive rate for IgG anti-HAV was 54.3% (338/622) in general healthy population and 75.6% (527/697) in patients with HBV-related CLD with a significant difference (P<0.001). Following the age standardization, however, it was found to be 52.5% and 49.1%, respectively. This difference did not reach a statistical significance (OR 1.026, 95% CI 0.735~1.433, P=0.880). The age-stratified IgG anti-HAV seroprevalence in general healthy population for ages≤19, 20~29, 30~39, 40~49, 50~59, ≥60 was 14.3% (2/14), 11.2% (19/169), 45.5% (92/202), 90.5% (86/95), 97.6% (81/83), and 98.3% (58/59), respectively. It was 0% (0/2), 9.8% (6/61), 46.3% (81/175), 91.1% (154/169), 97.7% (167/171), and 100.0% (119/119), respectively, in patients with HBV-related CLD (Fig. 1).
The sex-related positive rate for IgG anti-HAV was 68.8% (503/731) in male patients and 61.6% (362/588) in female patients (P=0.006). After the age standardization, however, it was found to be 54.1% and 48.7%, respectively. This difference did not reach a statistical significance (OR 1.072, 95% CI 0.772~1.487, P=0.680). Therefore, there was no significant difference in the age-stratified seroprevalce of IgG anti-HAV according to sex (Fig. 2). In general healthy population, the positive rate for IgG anti-HAV between male patients (60.5%, 170/281) and female patients (49.3%, 168/341) was significantly different (P=0.006), however, this difference did not reach a statistical significance (55.1% in male vs. 48.0% in female; OR 1.324, 95% CI 0.848~2.069, P=0.217) after the age standardization. In patients with HBV-related CLD, it was found to be 74.0% (333/450) and 78.5% (194/247), respectively. It was not significantly different (P=0.197), and following the age standardization, it was also not changed (48.6% in male vs. 49.9% in female; OR 0.817, 95% CI 0.494-1.352, P=0.433).
To identify the factors affecting the positive rate for IgG anti-HAV, a multivariate analysis was performed using variables such as age, sex, the presence of HBsAg, and the severity of liver diseases. Age (<30 vs. 30-59: OR 19.339, 95% CI 12.504~29.911, P<0.001; <30 vs. ≥60: OR 1060.5, 95% CI 142.233~7907.964, P<0.001) and the severity of liver diseases (OR 19.180, 95% CI 4.550-80.856, P<0.001) were identified as independent factors for HAV prior exposure. Sex (OR 1.018, 95% CI 0.762~1.360, P=0.906) and the presence of HBsAg (OR 1.246, 95% CI 0.931~1.668, P=0.139) were not associated with HAV prior exposure (Table 2).
Improvements in the socioeconomic status and general public health of Korea have led to a shift in the seroprevalence of hepatitis A.3 In the early 1980s, the positive rate of antibody against HAV has been reported to exceed 90% in adults aged 20 years or older.10 Thereafter, it has been gradually decreased. Paradoxically, the prevalence of apparent hepatitis A infection has been increased in adolescents and young adults. Symptomatic hepatitis A has been rapidly increasing on a nationwide scale since 2000, and it has become a legally-designated infectious disease. At the present, as an infectious disease for sentinel surveillance, the status of its occurrence has been collected by the Korea Centers for Disease Control and Prevention (KCDC).4 According to epidemiologic data by the KCDC, there were 320 cases of acute hepatitis A in 2002, 307 cases in 2003 and 357 cases in 2004. Since 2005, however, it has begun to rise strikingly, showing 826 cases in 2005, 2,130 cases in 2006, 2,168 cases in 2007 and 7,878 cases in 2008. Then, it reached 15,041 cases in 2009, showing the explosive increase in the incidence of acute hepatitis A.4 These results indicate that the incidence of acute hepatitis A occurred at an incidence of almost 30 cases per 100,000 Korean people, which urges to promote HAV vaccination for childhood and high risk groups.
The superinfection of hepatitis virus infection in patients with CLD may aggravate the underlying liver disease. Korea is an endemic area of HBV infection.11 and superinfection with HAV in HBV-related CLD remains problematic in these days. In Thai, it has been reported that the incidence and mortality of fulminant hepatic failure were 55% and 25% in cases of acute hepatitis A superinfection in HBsAg carriers.12 Chu et al. from Taiwan reported that risks of developing fulminant hepatic failure were approximately nine times higher in HBsAg carriers than noncarriers.13 According to a study about the prevalence of hepatitis A, which was conducted in Shanghai and the USA, Keeffe reported that the mortality was approximately 6-58 times higher in HBsAg-positive patients as compared with HBsAg-negative patients.14 In a Korean study,15 it was demonstrated that HBV-related CLD is related to the poor prognosis of acute hepatitis. As mentioned above, considering the marked increase in the incidence of apparent hepatitis A due to the epidemiologic shift and a poor prognosis in patients with HBV-related CLD who have HAV superinfection, it is imperative that the vaccination policy for hepatitis A be established in patients with HBV-related CLD. To date, however, studies of HAV seroepidemiology in populations including HBV-related CLD in Korea are scarce.
According to the current study, the positive rate for IgG anti-HAV was relatively higher in patients with HBV-related CLD and male patients as compared with general healthy population and female patients, respectively. Following the age standardization, however, there was no significant difference in the positive rate according to the presence of HBsAg and sex. This might be because elderly people accounted for a greater part of patients with HBV-related CLD and male patients. There was no significant difference in the age-specific seroprevalence of IgG anti-HAV between the two groups and multivariate analysis showed that sex and the presence of HBsAg were not associated with HAV prior exposure. These results were in agreement with reports, made by Hindman et al.,16 that hepatitis A and hepatitis B are mutually exclusive to each other. Besides, the above results were comparable with reports of Saab et al.,17 that the positive rate for IgG anti-HAV was higher in patients with HBV-related CLD as compared with general healthy population in univariate analysis, but there was no significant correlation in multivariate analysis. Also in Korea, Cho et al.18 also reported that the age was the independent factor affecting HAV prior exposure, which was consistent with our findings.
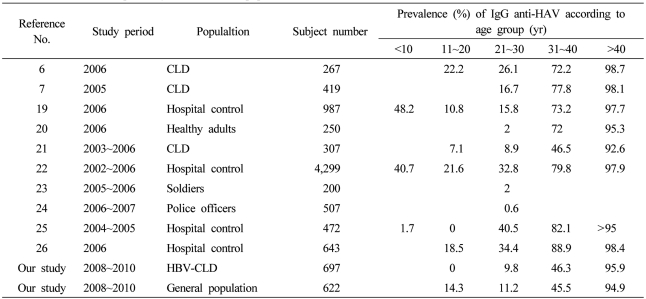
In respect to the positive rate for IgG anti-HAV, our study showed similar results to other Korean reports (Table 3).6,7,19-26 The positive rate for IgG anti-HAV was found to be higher than 90% in patients aged 45 years or older. Most of these patients were exposed to HAV previously and then spontaneously acquired an immunity against hepatitis A. In patients aged 29 years or younger, the positive rate for IgG anti-HAV was found to be lower than 15%. These results suggest that most of these patients were devoid of immunity. In patients aged between 30 and 44 years old, the positive rate for IgG anti-HAV varied ranging from 28 to 86%. These results indicate that the selective vaccination strategy would be warranted for these age groups. According to Saab et al.,27 in cases in which the positive rate for IgG anti-HAV was higher than 35%, the selective vaccination had an excellent profile of cost-effectiveness as compared with the universal vaccination. According to the current study, the prevalence of IgG anti-HAV in general healthy population aged between 30 and 34 years old and those aged between 35 and 39 years old was found to be 28.8% (32/111) and 65.9% (60/91), respectively. And, the prevalence of IgG anti-HAV in patients with HBV-related CLD aged between 30 and 34 years old and those aged between 35 and 39 years old was found to be 27.4% (23/84) and 63.7% (58/91), respectively. Based on our results, it can be inferred that the universal vaccination should be used for patients aged 34 years or younger and the selective vaccination should be used for patients aged 35 years or older. This might be beneficial from a cost-effective perspective.
Our results also showed that not only the age but also the severity of liver disease had a prognostic value for a past history of exposure to hepatitis A infection. It is therefore imperative that an active vaccination be performed for younger patients aged 35 years or younger, especially, patients with HBV-related CLD who had no advanced liver diseases such as liver cirrhosis or hepatocellular carcinoma, because they are expected to have a lack of the immunity against hepatitis A. Besides, considering that patients with HBV-related CLD who have a superinfection with acute hepatitis A have a poor clinical course as compared with general healthy population, the vaccination based on an active screening test should be performed for patients with HBV-related CLD who were aged 35 years or older. Our results might present the baseline data for establishing the appropriate vaccination strategy for general healthy population and patients with HBV-related CLD.
The limitations of the current study are that it was difficult to identify a statistical significance in young age group because the number of enrolled patients aged 19 years or younger was smaller than those of other age groups. Also, because the current study was conducted in a single center in Seoul and did not include patients with non-HBV-related CLD, it could not be representative of total Korean people and could not be generally applied to all the Korean patients with CLD. Considering not only that there was no significant difference in the seroprevalence of IgG anti-HAV between the current study and other previous Korean reports but also that HBV was the most common cause of CLD in Korea, however, our results might present the baseline epidemiologic data for IgG anti-HAV in Korean patients with CLD.
In conclusion, our results showed that there was an epidemiologic shift where the positive rate for IgG anti-HAV was decreased as compared with the past in adults in their 20s and 30s. It was also confirmed that the prevalence of IgG anti-HAV was not increased in patients with HBV-related CLD as compared with general healthy population. This might be used as a baseline epidemiologic data for estimating the prevalence of IgG anti-HAV in general healthy population and patients with CLD in Korea. This should also be followed by a nationwide epidemiological study. Besides, the age and the severity of hepatic diseases were found to be variables affecting the HAV prior exposure, based on which the HAV vaccination strategy should be established.
REFERENCES
1. Lemon SM. Type A viral hepatitis. New developments in an old disease. N Engl J Med 1985;313:1059-1067. 2413356.


2. Jung YK, Kim JH. Epidemiology and clinical features of acute hepatitis A: from the domestic perspective. Korean J Hepatol 2009;15:438-445. 20037262.


3. Kwon SY. Current status of liver diseases in Korea: hepatitis A. Korean J Hepatol 2009;15(Suppl 6):S7-S12. 20037282.


4. Jeong SH. Current status and vaccine indication for hepatitis A virus infection in Korea. Korean J Gastroenterol 2008;51:331-337. 18604134.

5. Lee CH, Kim JH, Paik SH, Jo KH. In: Lee CH, ed. Prospective study about natural history of chronic hepatitis B in Korea. The status and development of new therapy and prevention about chronic hepatitis B and C in Korea. Final report about intensive cooperative study (HMP-99-M-01-0008). 2002. Seoul: Department of health and welfare; p. 28-62.
6. Kim do Y, Ahn SH, Lee HW, Kim SU, Kim JK, Paik YH, et al. Anti-hepatitis A virus seroprevalence among patients with chronic viral liver disease in Korea. Eur J Gastroenterol Hepatol 2007;19:923-926. 18049159.


7. Song HJ, Kim TH, Song JH, Oh HJ, Ryu KH, Yeom HJ, et al. Emerging need for vaccination against hepatitis A virus in patients with chronic liver disease in Korea. J Korean Med Sci 2007;22:218-222. 17449927.



9. Bruix J, Sherman M. Practice Guidelines Committee, American Association for the Study of Liver Diseases. Management of Hepatocellular Carcinoma. Hepatology 2005;42:1208-1236. 16250051.


10. Kim JY, Hong WS. Seroepilemiology of Type A and Type B Hepatitis in Seoul Area. Korean J Intern Med 1982;25:19-27.
11. Chae HB, Kim JH, Kim JK, Yim HJ. Current status of liver diseases in Korea: hepatitis B. Korean J Hepatol 2009;15(Suppl 6):S13-S24. 20037275.


12. Pramoolsinsap C. Acute hepatitis A and acquired immunity to hepatitis A virus in hepatitis B virus (HBV) carriers and in HBV- or hepatitis C virus-related chronic liver diseases in Thailand. J Viral Hepat 2000;7(suppl 1):11-12. 10866839.


13. Chu CM, Liaw YF. Increased incidence of fulminant hepatic failure in previously unrecognized HBsAg carriers with acute hepatitis independent of etiology. Infection 2005;33:136-139. 15940414.


14. Keeffe EB. Is hepatitis A more severe in patients with chronic hepatitis B and other chronic liver diseases? Am J Gastroenterol 1995;90:201-205. 7847285.

15. Kim JI, Kim YS, Jung YK, Kwon OS, Kim YS, Ku YS, et al. Factors influencing the severity of acute viral hepatitis A. Korean J Hepatol 2010;16:295-300. 20924212.



16. Hindman SH, Maynard JE, Bradley DW, Berquist KR, Denes AE. Simultaneous infection with type A and B hepatitis viruses. Am J Epidemiol 1977;105:135-139. 189599.


17. Saab S, Lee C, Shpaner A, Ibrahim AB. Seroepidemiology of hepatitis A in patients with chronic liver disease. J Viral Hepat 2005;12:101-105. 15655056.


18. Cho HJ, Choi MS, Lee JH, Koh KC, Yoo BC, Paik SW. Prevalence of hepatitis a virus antibody in paitients with chronic liver disease. Korean J Hepatol 2010;16(Suppl 3):S196 [Abstract].
19. Kim JH. Recent Epidemiological Status and Vaccination of Hepatitis A in Korea. J Korean Med Assoc 2008;51:110-118.

20. Song YB, Lee JH, Choi MS, Koh KC, Paik SW, Yoo BC, et al. The age-specific seroprevalence of hepatitis A virus antibody in Korea. Korean J Hepatol 2007;13:27-33. 17380072.

21. Lee D, Cho YA, Park Y, Hwang JH, Kim JW, Kim NY, et al. Hepatitis a in Korea: epidemiological shift and call for vaccine strategy. Intervirology 2008;51:70-74. 18434743.

22. Kim TY, Sohn JH, Ahn SB, Son BK, Lee HL, Eun CS, et al. Comparison of recent IgG anti-HAV prevalence between two hospitals in Seoul and Gyeonggi area. Korean J Hepatol 2007;13:363-369. 17898552.


23. Kang CI, Choi CM, Park TS, Lee DJ, Oh MD, Choe KW. Incidence and seroprevalence of hepatitis A virus infections among young Korean soldiers. J Korean Med Sci 2007;22:546-548. 17596668.



24. Lee JM, Park HK, Choi KY, Kim HK, Jung JO, Suh SO, et al. Incidence and seroprevalence of hepatitis a virus among Korean auxiliary police officers. Infect Chemother 2007;39:292-295.
25. Kwon YO, Choi IJ, Jung JW, Park JH. An epidemiologic study on the seropositive rate of hepatitis A virus among a selected group of children and adults in Busan. Korean J Pediatr 2007;50:262-267.

26. Yoo HJ, Yoon BC, Han BH, Lee SU. The clinical feature of acute hepatitis A and IgG anti-HAV seroprevalence of the Busan: Single center experience. Korean J Hepatol 2007;13(Supple 3):S173 [Abstract].
Figure 1
Age-stratified IgG anti-HAV prevalence rates in the general healthy population and patients with HBV-CLD. The prevalence rates in the various decade age groups did not differ significantly between these two groups.
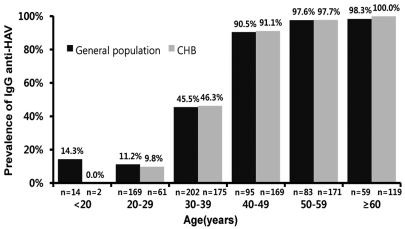
Figure 2
Age-stratified IgG anti-HAV prevalence rates in male and female subjects. The prevalence did not differ significantly with sex.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




