| Clin Mol Hepatol > Volume 20(3); 2014 > Article |
The sarcomatoid variant of hepatocellular carcinoma (HCC) is a rare histologic variant comprising malignant spindle cells and typical HCC. The proportion of the spindle cell component varies; when the tumor fully consists of malignant spindle cells, it is difficult to distinguish from various primary sarcomas. The 2010 World Health Organization classification of gastrointestinal tract tumors classifies sarcomatoid HCC under the special HCC types with fibrolamellar carcinoma, scirrhous HCC, undifferentiated carcinoma, and lymphoepithelioma-like carcinoma.1 Carcinosarcoma, which is synonymously used with sarcomatoid carcinoma, is separately described under "malignancies of mixed or uncertain origin" of the liver encompassing calcifying nested epithelial stromal tumor, combined hepatocellular-cholangiocarcinoma, hepatoblastoma, mixed epithelial-mesenchymal type, and malignant rhabdoid tumor. Here a recent case of sarcomatoid HCC is reported and relevant literature is reviewed.
A 64-year-old man was admitted for the resection of a hepatic mass. He had hepatitis B virus-associated chronic liver disease and a several-year history of medical treatments for liver cancer that was first detected 5 years prior. He underwent transarterial embolization (TAE) three times for a 1.4-cm multinodular mass in segment 6 first and percutaneous ethanol injection (PEI) and TAE of a new lesion in segment 6 after 4 years. Seven months after the last treatment, marginal recurrence of a segment 6 mass around the previous PEI defect site was suspected. Magnetic resonance imaging showed a 5-cm lobulating soft tissue mass with an internal hemorrhagic component. Except for the first event showing mild elevation of serum ╬▒-fetoprotein (AFP) of 36.1 ng/mL, serum AFP and proteins induced by vitamin K absence (PIVKA) were within the normal range. A peripheral segmentectomy of segment 6 was then performed.
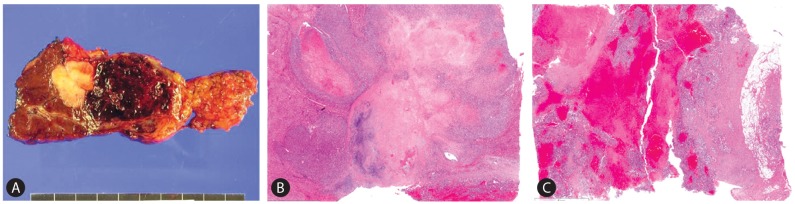
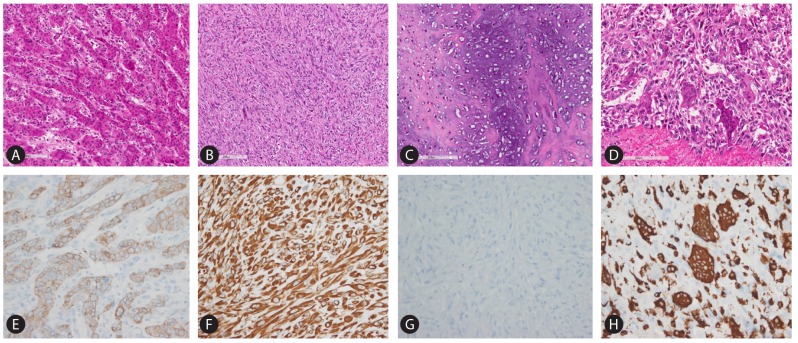
On gross examination, a 6.0├Ś4.0├Ś2.2 cm solid mass was identified in the cirrhotic hepatic parenchyma. The mass consisted of a mixture of two different solid components abutting each other (Fig. 1A). One was a 2.5├Ś2.0├Ś1.2 cm white glistening hard lobulating mass (Fig. 1B) and the other was a 4.0├Ś4.0├Ś2.0 cm red friable sponge-like mass extending into the perihepatic adipose tissue that resembled a hemangioma or ruptured hepatoma (Fig. 1C). On microscopy, the white lobulating mass was composed of a central hypocellular stroma and peripheral hypercellular tumor nodules (Fig. 1B). Two tumor cell types were seen. The first type was classical hepatocyte-like cells arranged in a microtrabecular pattern with fibrotic stroma or blood-filled sinusoid compatible with classical HCC (Fig. 2A). These cells had hyperchromatic round nuclei, plump eosinophilic cytoplasm, and a distinct cellular membrane, and some mitosis. Edmondson-Steiner's nuclear grade of most of these cells was grade III. The second type was spindle cells showing a storiform pattern. These cells had indistinct cellular membranes, short spindle hyperchromatic nuclei that frequently displayed mitosis (9 per 10 high-powered fields), and some small multinucleated giant cells (Fig. 2B). The hypocellular stroma in the center consisted of a pink hyalinized collagen matrix and a blue chondroid matrix containing viable cells within the lacunae, implying chondroid differentiation (Fig. 2C). The red sponge-like mass showed some cellular components in blood pools (Fig. 1C). The tumor cells were not in an organoid pattern and had ovoid or short spindle-shaped cytoplasm. Hyperchromatic nuclei displayed membrane folding and prominent nuclei, and frequently displayed mitosis (up to 22 per 10 HPF). Large multinucleated giant cells having 10-25 small nuclei within the cytoplasm, the so-called osteoclast-like giant cells (OGC) (Fig. 2D), were occasionally identified in these areas.
Immunohistochemical staining revealed that the HCC component was reactive for pan-cytokeratin (Fig. 2E) but not for AFP, antihepatocyte antigen, glypican-3. The spindle cell components revealed a positive reaction for vimentin (Fig. 2F) but not cytokeratin (Fig. 2G) and no positive reaction for desmin, smooth muscle actin, CD34, or C-kit, suggesting no specific cellular differentiation. Multinucleated cells were positive for CD68 (Fig. 2H).
The sarcomatoid variant of carcinoma is rare but can be observed in any organs in which carcinomas can occur, such as the lung, breast, prostate, skin, gastrointestinal tract, and female genital organs.2 Terminology for these tumors are various among organs, including sarcomatoid carcinoma, spindle cell carcinoma, metaplastic carcinoma, pleomorphic carcinoma, and carcinosarcoma. By definition, the sarcomatoid variant of carcinoma has to have both epithelial and mesenchymal differentiation in the same lesion. Although sarcomatoid carcinoma and carcinosarcoma are synonymously used, sarcomatoid carcinoma is preferred when the spindle cells have remaining epithelial differentiation confirmed by immunohistochemical staining for epithelial markers such as cytokeratin and no specific type of mesenchymal lineage, while the term carcinosarcoma is used when the spindle cells have the characteristic of specific lineages of sarcoma such as chondrosarcoma, malignant fibrous histiocytoma, fibrosarcoma, or rhabdomyosarcoma as well as no epithelial differentiation.3
Because sarcomatoid carcinoma in the liver is so rare, its general incidence is not described in the text; however, in Japan, the incidence of the sarcomatoid variant of HCC was reportedly 1.8-2.0% of surgical cases and 3.9-9.4% of autopsy cases and higher in cases of HCC with repeated preoperative treatment like TAE or chemotherapy than in cases of non-treated HCC.4,5
In a recent literature review of 92 cases of sarcomatoid carcinoma in the liver, 79% cases had HCC in the epithelial compartment and accompanying sarcomatoid components of osteosarcoma, fibrosarcoma, malignant fibrous histiocytoma, rhabdomyosarcoma, chondrosarcoma, embryonal sarcoma, and leiomyosarcoma; however, a specific sarcoma type was not reported for approximately one-third of the cases.6 Another comparative study between sarcomatoid carcinoma and carcinosarcoma reported that the two types of biphasic tumors did not differ significantly in terms of several clinicopathological features including survival, stage, and background liver disease except for the retention of epithelial differentiation.3 However, considering the scarcity of these tumors, whether a sarcomatous component with specific lineage will influence the clinical course remains unclear.
Several hypotheses have been reported regarding the pathogenesis or histogenesis of sarcomatous lesions in sarcomatoid carcinoma: 1) transdifferentiation or dedifferentiation from the original carcinoma cells, 2) biphasic differentiation from pluripotent stem cells, 3) metaplastic process of carcinoma, and 4) redifferentiation of immature multipotent carcinoma cells transformed from carcinoma cells.3,7,8,9
OGC in carcinomas are reported in many organs including the pancreas, breast, and thyroid; among them, the pancreas is predominant.10 The incidence of OGC in liver cancer was reviewed in 2009 by Tanahashi et al, who summarized 15 earlier studies, of which one-third of 16 reports including their case reported the presence of overt HCC.10 OGC in carcinoma are generally considered reactive histiocytic cells rather than true malignant tumor cells10 because OGC react positively for CD68 and have a low Ki-67 index. About histogenesis of OGC in liver, OGC in liver cancer had a similar expression of almost all osteoclast markers of bone: tartrate-resistant acid phosphatase, CD68, CD51, CD54, matrix metalloprotease-9, receptor activator of nuclear factor-kappa B (RANK), and RANK ligand (RANKL), suggesting that OGC in liver cancer had similar histogenesis of osteoclastogenesis in bone.11
Chondroid differentiation in sarcomatoid carcinoma or carcinosarcoma of the liver has two issues in diagnosis and pathogenesis: the differential diagnosis of hepatoblastoma and whether it is a true chondrosarcoma versus metaplastic process of tumor cells or stroma. Hepatoblastoma is usually a pediatric tumor, but there are several case reports of adult hepatoblastoma in which embryonal or fetal-type immature hepatocytes and myxofibrous stroma are required for diagnosis.12 Differentiation between chondrosarcoma and metaplastic stroma depends on cellular atypia and the presence of epithelial differentiation in tumor cells embedded within the chondroid matrix. If cellular atypia is sufficient for malignancy without epithelial differentiation, it can be interpreted as chondrosarcoma; in contrast, if the degree of cellular atypism is sufficient for malignancy with epithelial differentiation, it can be interpreted as metaplastic carcinoma and not chondrosarcoma. If neither cellular atypism nor epithelial differentiation is present, it may be the metaplastic process of collagenous stroma.
The prognosis of the sarcomatoid variant of HCC is unfavorable compared with classical HCC, and there are no effective diagnostic tools except for tissue confirmation; thus, several studies have recommended close follow-up and biopsy for tissue confirmation when unusual or atypical features are found in radiological studies.3,13,14
This case was histologically typical but interesting in that it had two distinct tumor cell types on macro- and microscopic findings with a transitional zone and spindle cell components that contained a unique combination of OGC and chondroid differentiation. The patient presented with multiple lung metastases 2 months after resection.
REFERENCES
1. Bosman FT, Carneiro F, Hruban RH, Theise ND. WHO classification of Tumours of the Digestice System. Lyon: IARC; 2010.
2. Yoshida N, Midorikawa Y, Kajiwara T, Yoshida N, Nakayama H, Sugitani M, et al. Hepatocellular carcinoma with sarcomatoid change without anticancer therapies. Case Rep Gastroenterol 2013;7:169-174. 23626518.



3. Wang QB, Cui BK, Weng JM, Wu QL, Qiu JL, Lin XJ. Clinicopathological characteristics and outcome of primary sarcomatoid carcinoma and carcinosarcoma of the liver. J Gastrointest Surg 2012;16:1715-1726. 22767081.


4. Kakizoe S, Kojiro M, Nakashima T. Hepatocellular carcinoma with sarcomatous change. Clinicopathologic and immunohistochemical studies of 14 autopsy cases. Cancer 1987;59:310-316. 2433017.


5. Kojiro M, Sugihara S, Kakizoe S, Nakashima O, Kiyomatsu K. Hepatocellular carcinoma with sarcomatous change: a special reference to the relationship with anticancer therapy. Cancer Chemother Pharmacol 1989;23(Suppl):S4-S8. 2466583.


6. Giunchi F, Vasuri F, Baldin P, Rosini F, Corti B, D'Errico-Grigioni A. Primary liver sarcomatous carcinoma: report of two cases and review of the literature. Pathol Res Pract 2013;209:249-254. 23484778.


7. Fayyazi A, Nolte W, Oestmann JW, Sattler B, Ramadori G, Radzun HJ. Carcinosarcoma of the liver. Histopathology 1998;32:385-387. 9602346.


8. Kubosawa H, Ishige H, Kondo Y, Konno A, Yamamoto T, Nagao K. Hepatocellular carcinoma with rhabdomyoblastic differentiation. Cancer 1988;62:781-786. 2840192.


9. Murata M, Miyoshi Y, Iwao K, Wada H, Shibata K, Tateishi H, et al. Combined hepatocellular/cholangiocellular carcinoma with sarcomatoid features: genetic analysis for histogenesis. Hepatol Res 2001;21:220-227. 11673107.


10. Tanahashi C, Nagae H, Nukaya T, Hasegawa M, Yatabe Y. Combined hepatocellular carcinoma and osteoclast-like giant cell tumor of the liver: possible clue to histogenesis. Pathol Int 2009;59:813-816. 19883433.


11. Ikeda T, Seki S, Maki M, Noguchi N, Kawamura T, Arii S, et al. Hepatocellular carcinoma with osteoclast-like giant cells: possibility of osteoclastogenesis by hepatocyte-derived cells. Pathol Int 2003;53:450-456. 12828610.


12. Fu Y, Kobayashi S, Kushida Y, Saoo K, Haba R, Mori S, et al. Sarcomatoid hepatocellular carcinoma with chondroid variant: case report with immunohistochemical findings. Pathol Int 2000;50:919-922. 11107071.


Figure┬Ā1
Macroscopic picture and scan veiw. The mass consisted of (A) a mixture of two different solid components; a white glistening hard lobulating mass and a red friable sponge-like mass extending into the perihepatic adipose tissue. (B) The white mass showed a central hypocellular stroma and peripheral hypercellular tumor nodules. (C) The sponge-like mass showed some cellular components in blood pools [Hematoxylin-eosin stain, original magnification ├Ś12 (B), (C)]
Figure┬Ā2
Microscopic and immunohistochemical findings. Classical hepatocellular carcinoma component (A) was reactive for pan-cytokeratin (E). Spindle cell component (B) was reactive for vimentin (F), but not pan-cytokeratin (G). Chondroid components consisted of a pink hyalinized collagen matrix and a blue matrix containing viable cells within the lacunae (C). Large multinucleated giant cells having 10-25 small nuclei within the cytoplasm (osteoclast-like giant cells, D) were reactive for CD68 (H) [(A), (B), (C), (D) Hematoxylin-Eosin stain, original magnification ├Ś200 (A), (B), (C), ├Ś400 (D), (E), (F), (G), (H)]



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




